The X Chromosome Is Unique
In women, there is a systematic demand to compensate for SCD by silencing one of the copies of the X chromosome. With two X-chromosomes, women are more prone to inheriting potentially deleterious mutations in X-encoded genes, which, because of Xi, may all be expressed in different cells. The first finding of inactivation of the X chromosome was reported by Lyon, . It was found that one of the X chromosomes, paternal or maternal, was always inactivated, suggesting that an inactivation mechanism only allows active transcription at one X chromosome . This process of X-chromosome inactivation evolved as a mechanism to regulate gene dosage. As a compensation mechanism, it does not affect all genes equally, and those genes that are not affected are known to escape XCI .
Xi acquires several features of heterochromatin, such as hypermethylation, hypercondensation, altered replication patterns , and depletion of acetylated histones . Methylation patterns have been extensively used to determine the inactive chromosome , enabling an analysis of non-random inactivation processes in diseases that are X chromosome-linked . In our published study we suggest that changes in the inactivation patterns of the X-chromosome could have an impact on AD pathogenesis .
S Based On The Conformational Changes Of Single
DGGE is a rapid, commonly used method for mutation detection. The technology is based on the mobility of double-stranded DNA in polyacrylamide gel containing linearly increasing concentrations of denaturing chemicals., SSCP is a simple PCR-based mutation detection method. The mobility of double-stranded PCR fragments depends on the size of the DNA, since the polymorphisms might result in the altered mobility of single-stranded DNA by changing its conformation . The PCR products should be denatured by heat and formamide, followed by neutral polyacrylamide gel electrophoresis.,
The single-strand conformation polymorphism process. After denaturation of the polymerase chain reaction product, the conformation of single-stranded DNA could be different, resulting in altered mobility in polyacrylamide gel.
Abbreviations: dsDNA, double-strand DNA PAGE, polyacrylamide gel electrophoresis WT, wild type.
Brain And The X Chromosome
The X chromosome harbors 3-5% of all the genes in a genome . There has been a debate on how many genes reside on the X chromosome and how many genes are expressed in the brain alone, compared to genes that are X-linked and expressed in the placenta, testes, muscles, and ovary. It is estimated that between 1,100 and 1,500 genes are present on the X chromosome . By using the Mart View software it was found that 1,500 X-linked genes are expressed in the brain, which represent numerous candidate genes that could be responsible for X-linked brain diseases . Many of the proteins expressed from the genes linked to the X chromosome represent channels, receptors, repair, transcription factors, and DNA/RNA binding proteins. Most of these proteins are located in the postsynaptic cleft and postsynaptic density and are regulated through signaling complexes . It is intriguing that even if the X chromosome harbors 3-5% of all the genes, it is responsible for 10% of all diseases with Mendelian inheritance .
Read Also: Does Neil Diamond Have Alzheimer’s
Symptoms Of Alzheimers Disease
Memory loss Repeating things, often forgetting conversations or appointments, routinely misplacing things, eventually forgetting the names of family members and everyday objects.
- Problems with abstract thinking People with Alzheimers may have trouble recognising and dealing with numbers.
- Difficulty finding the right word It may be a challenge for those with Alzheimers to find the right words to express thoughts or even follow conversations. Eventually, reading and writing also are affected.
- Disorientation- People with Alzheimers disease often lose their sense of time and dates, and may find themselves lost in familiar surroundings.
- Loss of judgment Solving everyday problems, such as knowing what to do if food in the oven is burning, becomes increasingly difficult, eventually impossible. Alzheimers is characterized by greater difficulty in doing things that require planning, decision making and judgment.
- Difficulty performing familiar tasks- Once-routine tasks that require sequential steps, such as cooking, become a struggle as the disease progresses. Eventually, people with advanced Alzheimers may forget how to do even the most basic things.
- Changes in personality.
Sex Chromosome Dosage : An Engine Of Stability
The crosstalk that exists between X chromosomes and autologous genes is a relatively new paradigm that has emerged as a result of the biology of sex differences, and gives rise to the question of how SCD shapes the genome function. To explore this, human sex aneuploidies were analyzed from a genome-wide expression dataset by Raznahan et al. where they found a dosage sensitivity of the X-Y chromosome pair resulting in increased expression of genes that decrease X/Y chromosomal dosage . The most interesting finding was that X-linked genes were found to regulate co-expression of networks of autosomal genes that are SCD-sensitive and, in addition to these findings, suggest that the autosomal genes and their corresponding networks are crucial for cellular functions. This highlights the potential of SCD to affect the occurrence of disease.
The most common aneuploidy in AD is XO mosaicism . In respect to SCD and the XO status, Raznahan et al. have demonstrated up-regulation of the protein networks, noncoding RNA metabolism, suppression of the cell cycle, changes in regulation of DNA/chromatin organization, glycolysis, and response to stress . Changes in these collective networks through XO and supernumerary XXY, and XXYY syndromes may enhance the risk of AD .
Figure 2 X chromosome instability, Sex Chromosome Dosage, Topological changes of Chromosomes, and its possible role in AD.
You May Like: Shampoos That Cause Alzheimer’s Disease
S Of Detecting Mutation
PCR-based methods can be performed for monitoring the mutations in the AD risk factor genes . Genomic DNA can be extracted from total blood, buffy coat , bone marrow, or cell cultures, using a specific extraction kit. DNA should be amplified by specific primers, designed for the AD risk-factor genes such as APP, PSEN1, PSEN2, and APOE.,, Several mutation detection methods have been developed, such as restriction fragment length polymorphism , single-strand conformation polymorphism , denaturing gradient gel electrophoresis , temperature gradient gel electrophoresis , and heteroduplex analysis. RFLP is based on the recognition of a specific cleavage site and can be used for genetic mapping and linkage analysis. To identify the polymorphisms in the PCR products, the amplicons should be sequenced.
Polymerase chain reaction -based genetic methods.
Abbreviations: DGGE, denaturing gradient gel electrophoresis RFLP, restriction fragment length polymorphism TGGE, temperature gradient gel electrophoresis SSCP, single-strand conformation polymorphism.
How Is Alzheimers Disease Treated
Medical management can improve quality of life for individuals living with Alzheimers disease and for their caregivers. There is currently no known cure for Alzheimers disease. Treatment addresses several areas:
- Helping people maintain brain health.
- Managing behavioral symptoms.
- Slowing or delaying symptoms of the disease.
Don’t Miss: Difference Between Senility And Dementia
Alzheimers Disease Diagnostic Criteria
A patient suspected to have AD should undergo several tests, including neurological examination, magnetic resonance imaging for neurons, laboratory examinations such as vitamin B12, and other tests besides the medical and family history of the patients . Vitamin B12 deficiency has been long known for its association with neurologic problems and increasing risks of AD, according to some studies. A special marker of vit. B12 deficiency is elevated homocysteine levels, which can cause brain damage by oxidative stress, increasing calcium influx and apoptosis. Diagnoses of vit. B12 deficiency can be done by measuring serum vit. B12 level alongside complete blood count and serum homocysteine levels tests .
In 2011, The National Institute on AgingAlzheimers Association made several changes and updated the 1984 NINCDS-ADRDA criteria for higher specificity and sensitivity in the diagnosis of Alzheimers disease. The newly proposed criteria include probable and possible AD dementia for the use in clinical settings and probable or possible AD dementia with pathophysiological evidence for research purposes, in addition to clinical biomarkers. There are two categories of Alzheimers disease biomarkers: markers of brain amyloid such as positron emission tomography and cerebrospinal fluid , and markers of neuronal injury like cerebrospinal fluid tau, fluorodeoxyglucose for metabolic activity, and magnetic resonance imaging for atrophy measurement .
Support For Family And Friends
Currently, many people living with Alzheimers disease are cared for at home by family members. Caregiving can have positive aspects for the caregiver as well as the person being cared for. It may bring personal fulfillment to the caregiver, such as satisfaction from helping a family member or friend, and lead to the development of new skills and improved family relationships.
Although most people willingly provide care to their loved ones and friends, caring for a person with Alzheimers disease at home can be a difficult task and may become overwhelming at times. Each day brings new challenges as the caregiver copes with changing levels of ability and new patterns of behavior. As the disease gets worse, people living with Alzheimers disease often need more intensive care.
You can find more information about caring for yourself and access a helpful care planning form.
Also Check: What Color Ribbon Is For Alzheimer Disease
Bgrowth Hormone Response To Growth Hormone
Results of several studies addressing this point show varied results: superimposable responses of GH to GHRH than responses of GH to GHRH in controls a blunted GH to GHRH response in AD patients higher GH concentrations in the morning and a greater increase of GH to GHRH in AD patients than in controls . More recent studies demonstrate the reduced release of GH to GHRH in AD patients . In general, investigations of GH-releasing stimulation tests, especially of GHRH in AD, are equivocal and, in some cases, contradictory. Factors that may contribute to the inconsistent findings include the variability of GHRH stimulated among control groups, the age and sex of patients, the lack of uniformity in test procedures, and the variability and the lack of reproducibility of the GHRH test either in controls or in AD patients .
Table II. Different Patterns of Response of GH to Its Stimuli in Alzheimer’s Disease Patients
| 1. Higher GH concentrations in the morning |
| 2. Superimposable GH response to GHRH than GH response to GHRH in controls |
| 3. Blunted GH response to GHRH in Alzheimer’s disease patients |
| 4. Higher increase of GH after GHRH in Alzheimer’s disease patients |
| 5. Blunted GH response to clonidine in Alzheimer’s disease patients, with higher levels of aggression |
Leon E. Rosenberg, Diane Drobnis Rosenberg, in, 2012
Genetic Causes Of Dementia
One rare form of Alzheimers disease is passed from generation to generation. This is called Familial Alzheimers disease . If a parent has a mutated gene that causes FAD, each child has a 50% chance of inheriting it. The presence of the gene means that the person will eventually develop Alzheimers disease, usually in their 40s or 50s. This form of Alzheimers disease affects an extremely small number of people probably no more than 100 at any given time among the whole population of Australia.
Three genes have been identified which, if mutated in certain ways, will cause FAD. These are called presenilin 1 , presenilin 2 and the amyloid precursor protein gene on chromosome 21.
Also Check: Is Reading Good For Dementia
Unraveling The Genes Implicated In Alzheimer’s Disease
This article is mentioned in:
Abstract
Introduction
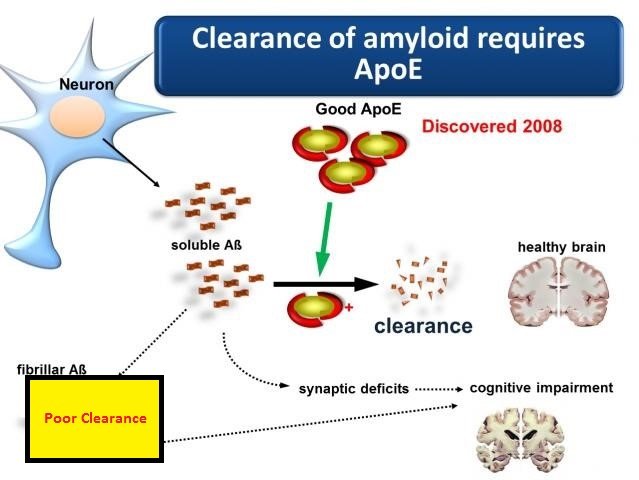
Alzheimer’s disease , the most common form ofdementia among older adults. It is a progressive neurodegenerativedisorder, which has been widely recognized as worldwide challengefor society and health-care providers. Clinically, it is defined byprogressive loss of cognitive functions, ultimately leading todementia and mortality .Neuropathologically, AD is characterized by the aggregation anddeposition of misfolded proteins, in particular aggregatedamyloid- peptide in the form of extracellular senile plaques and hyperphosphorylated tau protein in the formof intracellular neurofibrillary tangles . Early onset AD affects < 1% ofall AD cases with autosomal dominant inheritance. Late onset AD characterized by a genetically complex and high hereditarypattern of inheritance, is the most common form of the disease withan age of onset > 65 years .Evidence indicates that the major factor of AD pathogenesis is dueto abnormal aggregation and clearance of A by apolipoprotein E. However, other potential mechanisms, including modulation ofneurotoxicity and tau phosphorylation by Apo E, synaptic plasticityand neuro-inflammation have not been eliminated .
Associated and susceptibility genes ofEOAD
Table I. |
AD, Alzheimer’sdisease APP, amyloid- precursor protein A, amyloid-.
APP
PSEN1 and PSEN2
ATP binding cassette subfamily Amember 2
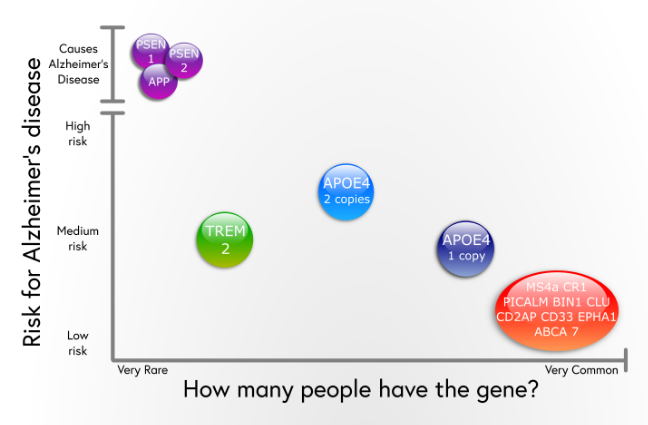
Alzheimers Disease Risk Factors
5.2.1. Aging
The most important risk factor in AD is aging. Younger individuals rarely have this disease, and most AD cases have a late onset that starts after 65 years of age . Aging is a complex and irreversible process that occurs through multiple organs and cell systems with a reduction in the brain volume and weight, a loss of synapses, and ventricles enlargement in specific areas accompanied by SP deposition and NFT. Moreover, several conditions might emerge during aging such as glucose hypometabolism, cholesterol dyshomeostasis, mitochondria dysfunction, depression, and cognitive decline. These changes also appear in normal aging, which makes it difficult to distinguish the cases in early AD . AD can be divided based on age of onset into early-onset AD , the rare form with around 16% of cases, in which most of them are familial AD characterized by having more than one member in more than one generation with AD, and ranges from 3060 or 65 years. The second type is the late-onset AD , which is more common with age of onset above 65 years. Both types may occur in people who have a family with a positive history of AD and families with a late-onset disease .
5.2.2. Genetics
Herein, we discuss the strong genetic risk factors in AD.
-
Amyloid Precursor Protein
-
Presenilin-1 and Presenilin-2
-
Apolipoprotein E
-
ATP Binding Cassette Transporter A1
-
Clusterin Gene and Bridging Integrator 1
-
Evolutionarily Conserved Signaling Intermediate in Toll pathway
-
Estrogen Receptor Gene
-
Diet
You May Like: What Color Ribbon Is Alzheimer’s
How Do Genes Work
Genes are the basic building blocks that direct almost every aspect of how youâre built and how you work. Theyâre the blueprint that tells your body what color your eyes should be or if youâre likely to get some kinds of diseases.
You get your genes from your parents. They come grouped in long strands of DNA called chromosomes. Every healthy person is born with 46 chromosomes in 23 pairs. Usually, you get one chromosome in each pair from each parent.
Dementia Caused By A Complex Disease
Nearly all cases of dementia are the result of a complex disease. In these cases, genes may increase the risk of developing dementia, but they dont cause it directly.
What causes complex disease dementias?
When dementia is the result of a complex disease, it is likely to be caused by a combination of risk factors. These include:
- non-genetic factors for example, members of the same family may all smoke or have an unhealthy diet, which are both risk factors for dementia.
- genetic factors a person may inherit the same dementia risk variants as other members of their family. This could include variants in genes such as APOE.
These factors are often shared by members of the same family. This is why many people have some family history of dementia for example, they may have a parent who developed dementia in their 90s and a brother who developed dementia in his 70s. This is not the same as familial dementia.
Is it possible to directly inherit dementia when it’s caused by a complex disease?
No. People who are related to each other by birth are more likely to have the same risk variants as each other. However, risk variants for dementia do not directly cause dementia . This means that its not possible to directly inherit dementia through risk variants.
However, a person who has dementia risk variants is at higher risk of developing dementia than someone who does not have risk variants. Despite this higher risk, they still might not develop dementia.
Recommended Reading: Color For Alzheimers
Dementia Caused By A Single
Of the four most common types of dementia, frontotemporal dementia is most likely to be caused by a single-gene change.
Is dementia different when it’s caused by a single-gene change?
Yes, it can be different in some ways.
- It often develops earlier in life. For example, an affected person might get dementia in their 40s or 50s rather than in their 70s or above. This is known as young-onset dementia.
- It tends to run strongly in families, with several close relatives who develop the same type of dementia. For example, a person with a single-gene dementia may have a grandparent, a parent and a brother who all had frontotemporal dementia . This is why dementia caused by a single-gene disease is sometimes known as familial dementia.
Can single-gene dementias be inherited directly?
Yes. If a single-gene dementia is present in a family, it is quite likely that a person from that family will develop dementia:
- If one of the parents carries the changed gene, each child has a 1 in 2 chance of inheriting it.
- If one of the children carries the changed gene, any brothers or sisters they have has a 1 in 2 chance of carrying it as well.
Alzheimers Disease Affects Women Differently
Compared with men, women are more likely to develop Alzheimers disease. Women account for nearly two-thirds of Americans affected by it.
On the other hand, Alzheimers disease tends to progress more quickly in men. They tend to experience more rapid cognitive decline and die sooner.
Past studies have found that sex-related differences in hormones, immune function, and energy metabolism may help account for these gaps.
The new study on KDM6A adds another piece to the puzzle, highlighting the role that non-hormone-related genes on sex chromosomes may play.
The Alzheimers Association held a think tank in 2015 to explore the biology that may contribute to sex differences in Alzheimers disease, Heather M. Snyder, PhD, the Alzheimers Associations vice president of medical and scientific engagement, told Healthline.
One of the outstanding questions from that think tank was that we did not yet have the tools to fully evaluate the impact of the X or Y chromosome.
She added that the new study is helping to start to address some important scientific questions by using emerging technologies to look at the complexity of the X chromosome.
Don’t Miss: Can Lewy Body Dementia Be Inherited