Am I At Risk For Alzheimer’s
Whether we have seen early signs in ourselves or not, many of us want to know what our chances of getting this disease may be. Research has shown a number of possible factors that can impact your chances of getting Alzheimer’s disease, although none of these are a cause in and of themselves.
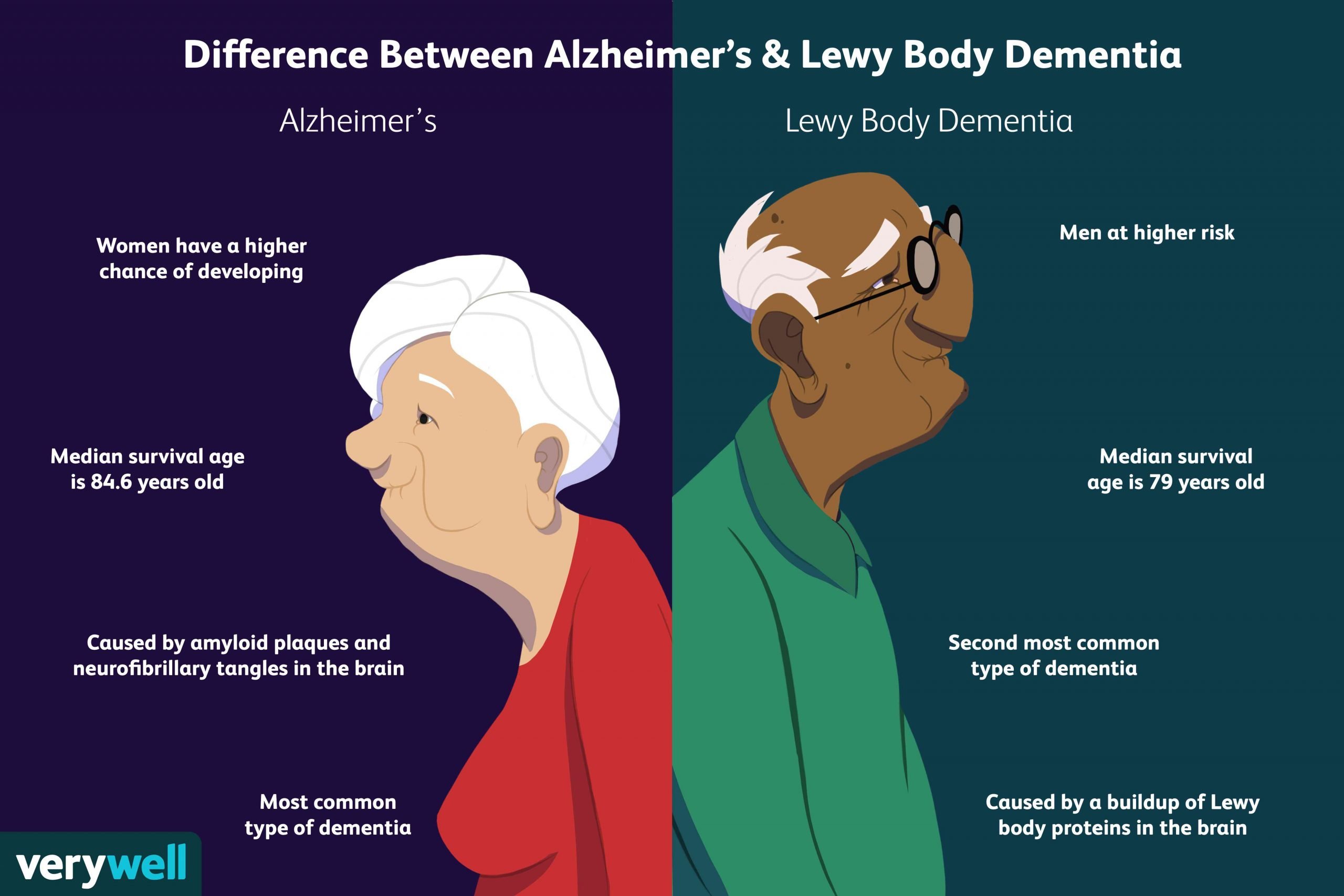
Some ages are more at risk
Old age is one of the most obvious risk factors. The vast majority of people develop the disease after the age of 65, and once you reach 65, your risk of getting Alzheimer’s doubles every five years. But Alzheimer’s doesn’t only affect people over 65 it has been known to affect people half that age, although this is much rarer.
Alzheimer’s is hereditary
A family history will also increase your risk of getting the disease. The risk increases even more if you have multiple family members who have suffered from the disease.
Whilst this may be due to the hereditary genetic factors we will look at in more depth later, there may be other factors at play. These could include environmental factors that impact both yourself and your family.
Gender predisposition
Gender is another significant risk factor. The first discovery of the disease back in 1906 was in a woman, and about twice as many women as men over 65 have Alzheimer’s. This may be in part to the fact that women have a longer lifespan or may even possibly be linked to menopause.
Genetic factors of Alzheimer’s
Other risk factors
Ethics Approval And Consent To Participate
The study was approved by the Medical Ethical Committee of the Erasmus Medical Center Rotterdam. Brain autopsy was conducted by the Netherlands Brain Bank at the designated premises of the VU Medical Center in Amsterdam . Ethical approval for the NBB procedures and forms was given by the Medical Ethics Committee of the VU Medical Center. Written informed consent for the use of tissues, clinical, and neuropathological data was obtained from all participants or their legal representatives, according to the Code of conduct for Brain Banking and Declaration of Helsinki.
Molecular And Neuropathologic Phenotypes Associated With A Polygenic Risk Score
We identified endophenotypes associated with the AD-PRS to aid in understanding the biological processes affected by genetic risk for Alzheimer’s dementia. We examined a wide range of possible effects in several categories of endophenotypes, which included 3 clinical diagnoses, 6 cognitive decline measures, 8 lifestyle and personality traits, 14 medical conditions, 13 brain pathologies, and 5 types of omics measurements from the DLPFC. Details of these endophenotypes, such as their sub-components and methods of acquisition are described in the Methods section and in the Supplementary Table 2. In total, with pathological, cognitive and behavioral factors, 590 variables were tested for the association with the AD-PRS. In our tests, we accounted for sex, age, education, and the first three principal components of genotype. We adopted p-value of 0.05/590 as a Bonferroni-corrected significance threshold .
Figure 1. Association between AD-PRS and clinical phenotypes, neuropathologies, and molecular signatures. A dash line represents Bonferroni-corrected p-value at 0.05.
Read Also: Can Aricept Make Memory Worse
The Five Percent: Strong Genetic Determinants Of Alzheimers
There are a few genes that almost certainly will lead to the development of Alzheimers. These genes are responsible for about five percent of all cases. All these genetic perpetrators happen to increase the production of amyloid plaques a key Alzheimers biomarker early in life:
In most people, these genes will lead to an early-onset Alzheimers between a persons late 30s or early 60s. But do these early-onset genes tell us about the underlying cause of Alzheimers at large? Scientists still arent quite sure.
The tricky part is: There are plenty of people who do develop Alzheimers, but whodont develop this key biomarker of amyloid plaques. Meanwhile, some people with amyloid plaques dont ever develop the disease, making the link between amyloid and Alzheimers tenuous.Some scientists believe that the biology of early-onset Alzheimers may involve a different mechanism, other than amyloid, altogether.
So, while there are a few genes that almost certainly will lead to the development of Alzheimers, unfortunately that doesnt solve the mystery of whats behind the other 95 percent of cases.
Expression In Various Cell Types
The expression of genes was assigned to specific cell classes of the adult brain, as described previously. Briefly, middle temporal gyrus single-nucleus transcriptomes from the Allen Brain Atlas dataset were used to annotate and select six main cell classes using Seurat 3.1.1 : glutamatergic neurons, GABAergic neurons, astrocytes, oligodendrocytes, microglia and endothelial cells. Enrichment analyses were performed by using the mean gene expression per nucleus for each cell type relative to the total expression summed across cell types as a quantitative covariate in a MAGMA gene property analysis.
You May Like: Did Reagan Have Alzheimer’s In Office
Genes Associated With Risk In Sporadic Ad
AD2: APOE
Inheritance and clinical features
The APOE gene has been associated with both familial late-onset and sporadic late-onset AD in numerous studies of multiple ethnic groups. The APOE4 genotype is associated with higher risk of AD, earlier age of onset of both AD and Down syndrome , and a worse outcome after head trauma and stroke, both in humans and in transgenic mice expressing human APOE4. The frequency of the APOE4 allele varies between ethnic groups, but regardless of ethnic group, APOE4 carriers are more frequently found in controls and APOE4+ carriers are more frequently found in patients with AD.,
Gene location and structure
Apolipoprotein E structure and single nucleotide polymorphisms . The general protein structure of APOE is shown. The 2 SNPs in exon 4 and corresponding protein locations are shown . The 3 APOE 4 alleles are defined by 2 SNPs, rs429358 and rs7412, with 2 defined by nucleotides T-T 3 defined by T-C, and 4 defined by C-C, respectively.
Gene function and expression
The mechanisms that govern apoE toxicity in brain tissue are not fully understood. Some proposed mechanisms include isoform-specific toxicity, APOE4-mediated amyloid aggregation, and APOE4-mediated tau hyperphosphorylation.
In addition, individuals carrying APOE4 have higher amyloid and tangle pathology and an increase in mitochondrial damage compared to those carrying other APOE polymorphisms.
Genetic variation
Cause #: Genetic Mutations
Alzheimers disease isnt fully understood. Scientists believe that for most people, the disease has genetic, lifestyle, and environmental factors. All these factors may work together to create the right conditions for the disease to take root.
There is a hereditary component to Alzheimers. People whose parents or siblings have the disease are at a slightly higher risk of developing the condition. However, were still a long way from understanding the genetic mutations that lead to the actual development of the disease.
Read Also: What’s The Difference Between Dementia And Alzheimer’s And Senility
What Kind Of Dementia Is Hereditary
Dr. Rx
Just because your family member had dementia does not mean you will also get it. Although you are at higher risk, there are also a number of things you can do to lower your risk. Dont smoke, get regular exercise, eat healthfully, manage health issues , maintain regular social connection, and do activities that challenge your brain . Dr. Wegner
Researchers have identified several genes that increase the riskof Alzheimers, such as the APOE-e4 gene. An estimated 40% to 65% of those diagnosed with Alzheimers have this gene.
Vascular dementia is less likely to be influenced by genes. It is more likely linked to risk factors for heart disease like high blood pressure, being overweight, having a sedentary lifestyle, or diabetes.
About one in three of frontotemporal disorders may be hereditary.
Dementia And Down Syndrome
People with Down syndrome are born with an extra piece of DNA. This means they also have an extra copy of the APP gene. This leads to the build-up of amyloid plaques in the brain, which play a role in the development of Alzheimers disease. While not everyone with Down syndrome will go on to develop symptoms of Alzheimers, most people with the condition over the age of 40 will have amyloid build-up. It is estimated that about 50% of people with Down syndrome develop symptoms like memory loss, usually in their 50s and 60s.
To find out more about Down syndrome and dementia you can contact Downs Syndrome Association helpline on 0333 1212 300 or visit their website www.downs-syndrome.org.uk.
You May Like: Alzheimers Awareness Symbol
The Genetic Landscape Of Eoad Includes Ftd
Besides pathogenic variants in APP, PSEN1, and SORL1, we discovered a deleterious GRN frameshift variant in one family. The variant is known to cause FTD and has not been associated with clinical AD, although one family was described with profound AD neuropathology . Although FTD-GRN patients may show coexistent AD pathology , certain GRN variants have been suggested as the direct cause of AD through several proposed mechanisms , indicating shared pathways between AD and progranulin . As such, we propose that the identified variant possibly in concert with an AD polygenic risk profile plays a causal role in this family and should be reported in genetic counseling. It would be of interest to evaluate PRS in a larger cohort of GRN carriers presenting with an AD phenotype. In a clinical setting, we recommend to examine an extended panel of dementia genes in familial EOAD, when APP, PSEN1, and PSEN2 are tested negative, as was suggested before . Following a family history of PD and ALS, we also screened genes related to these disorders in a few families. As this did not yield any candidate variants, it remains unclear whether such genes should be standardly examined in AD.
Genetic Testing And Counseling
In contrast, EOFAD, with an age at onset before 60 to 65 years old, has an autosomal dominant mode of inheritance in which 20% to 70% of cases are estimated to have a PSEN1 mutation, 10% to 15% of cases are estimated to have an APP mutation, and PSEN2 mutations are rare., Indeed, approximately 60% of patients with EOAD have another known affected family member. The remaining 40% of patients with EOAD may lack a family history because of an early death of a parent, failure to recognize the disorder in family members, or, very rarely, a de novo mutation. If the parent of a patient with EOAD has a mutant allele, then the risk to the patients sibling of inheriting the mutant allele is 50%. The child of a patient with EOAD who carries a mutation has a 50% chance of transmitting the mutant allele to each of their children.
In this article, we have reviewed the genetics of AD. Further molecular genetic investigations should clarify the roles of additional known genes in the pathogenesis of both common sporadic as well as rare familial forms of AD. Already, investigations of the normal and aberrant functions of A protein and ApoE has provided insight into the underlying mechanisms for AD. Such research will continue to provide new strategies for therapeutic interventions.
Read Also: Dementia Ribbon Tattoo
What Are Risk Factors
- Risk factors are aspects of your lifestyle, environment and genetic background that increase the likelihood of getting a disease.
- Risk factors on their own are not causes of a disease. Rather, risk factors represent an increased chance, but not a certainty, that dementia will develop.
- Similarly, having little or no exposure to risk factors does not necessarily protect a person from developing dementia.
There are some risk factors that can be changed, and some that cannot â read on to know which are which!
Read about risk factors for dementia in our downloadable, print-friendly infosheet.
This sheet also contains strategies and lifestyle changes that can help you reduce your risk of developing dementia.
What Role Do Our Genes Play In Dementia
As dementia is so common, many of us will have a relative living with the condition but this does not mean we will develop it too.
Dementia is caused by diseases that affect the brain, such as Alzheimers disease. The likelihood of developing dementia will usually depend on a complex mix of factors like our age, medical history and lifestyle, as well as our genes. Most cases of dementia are not directly caused by genes we inherit from our parents.
Recommended Reading: Alzheimer’s Lack Of Neurotransmitter
Clinical Demographics Of Included Families
With the pooled genetic data of 4840 patients, we constructed 36 families comprising at least two patients . Seven pedigrees were previously clinically established and used to validate our two methods for determining relatedness. Both approaches were able to define first- and second-degree relationships and were fully concordant.
For each family, clinical and genetic findings are summarized in Table . All included families consisted of at least one patient with AD before the age of 70. The average age at onset of all affected individuals was 63 years. A lumbar puncture was performed in 32/36 probands, confirming decreased concentrations of -amyloid in cerebrospinal fluid . Occasionally, relatives had been diagnosed with other phenotypes on the dementia spectrum . Four families included an unaffected relative aged > 75. Detailed information on the individual level is provided in Additional file .
Table 1 Summary of the clinical characteristics and genetic findings of the 36 families evaluated in this study
Determine Pedigrees By Genetic Data
We assessed family relationships using two independent methods. First, available SNP arrays were pooled for an identical-by-descent segment-based method to classify degrees of relatedness using PLINK 1.9 . Second, using the exome sequencing data of all three cohorts, we calculated a kinship coefficient between all pairs of samples using the dataset-VCF file with common variants only , based on the method of Manichaikul et al. . Previous studies demonstrated that the algorithm reliably infers up to third-degree relationships. Both methods were compared for possible discrepancies and were fully concordant. All identified kinships were checked and confirmed with previously clinically determined pedigrees, when available.
Also Check: Progressive Aphasia Dementia
The Recipe Book: Apoe4 And Other Risk Genes
The human genome is like a book of recipes, with each of our 20,000 or so genes serving as a guide to make a specific dish. In some peoples genomes, slight variations in these recipes exist.
Thanks to advances in sequencing technology, we can read the recipe book. So far, however, we do not fully understand each section. Our cells first copy the instructions within the master recipe book before passing on these instructions to their sous chefs who make the dish .
To carry the recipe metaphor further, foods like pufferfish, shark meat, or even rhubarb need to be specially prepared so that they do not poison the eater. Similarly, a small change in the genetic code could make what should be a regular protein damaging or cause it to disrupt an important process.
By far,ApoE4 is the most prominent example of a recipe gone wrong genetic risk factor. Carrying one copy of the gene is known to increase the risk of developing Alzheimers, at least in white populations, up to three times, while carrying two copies may increase the risk up to twelve times. If the chefs instructions to the sous chef are wrong, then the final dish wont come out right in some cases like with ApoE4 the mistake sometimes ruins the dish. However, knowing this risk factor hasnt led to any treatments focused on the gene.
Genetic Testing Of Apoe And Prs In Clinical Practice
APOE genotyping is not incorporated in current risk predictions, because it is neither diagnostic nor sufficiently predictive, and at present has no clinical consequences . These guidelines will likely not change until effective treatments become available. Nevertheless, given the fact that half of the families studied here were affected by APOE-4 homozygosity, in some instances accompanied by a potentially relevant VUS, a pertinent question is at which point the risk is sufficiently high to include these factors in genetic risk modeling and counseling. Importantly, APOE genotyping is currently used in clinical trials and is available via direct-to-consumer genetic testing . Previous studies suggest that genetic risk disclosure of APOE can be safely performed in a clinical setting, provided that appropriate and standardized counseling protocols are applied . On the other hand, the risk of misunderstanding or overinterpretation is an important issue, which must be carefully weighed against the possible personal utility of disclosure for the individual patient . At any rate, we urge APOE genotyping in a research setting to further clarify its impact and interplay with other factors.
Recommended Reading: Dementia Ribbon Color
Dementia Caused By A Complex Disease
Nearly all cases of dementia are the result of a complex disease. In these cases, genes may increase the risk of developing dementia, but they dont cause it directly.
What causes complex disease dementias?
When dementia is the result of a complex disease, it is likely to be caused by a combination of risk factors. These include:
- non-genetic factors for example, members of the same family may all smoke or have an unhealthy diet, which are both risk factors for dementia.
- genetic factors a person may inherit the same dementia risk variants as other members of their family. This could include variants in genes such as APOE.
These factors are often shared by members of the same family. This is why many people have some family history of dementia for example, they may have a parent who developed dementia in their 90s and a brother who developed dementia in his 70s. This is not the same as familial dementia.
Is it possible to directly inherit dementia when it’s caused by a complex disease?
No. People who are related to each other by birth are more likely to have the same risk variants as each other. However, risk variants for dementia do not directly cause dementia . This means that its not possible to directly inherit dementia through risk variants.
However, a person who has dementia risk variants is at higher risk of developing dementia than someone who does not have risk variants. Despite this higher risk, they still might not develop dementia.