Viral Infections Could Promote Neurodegeneration
- Date:
- DZNE – German Center for Neurodegenerative Diseases
- Summary:
- Some viral diseases could possibly contribute to neurodegeneration. Researchers found that certain viral molecules facilitated intercellular spreading of protein aggregates that are hallmarks of brain diseases like Alzheimer’s. These findings may provide clues how acute or chronic viral infections could contribute to neurodegeneration.
Some viral diseases could possibly contribute to neurodegeneration. DZNE researchers report this in the scientific journal Nature Communications. Their assessment is based on laboratory experiments in which they were able to show that certain viral molecules facilitate intercellular spreading of protein aggregates that are hallmarks of brain diseases like Alzheimer’s. These findings may provide clues how acute or chronic viral infections could contribute to neurodegeneration.
Experiments with Cell Cultures
Ligands Facilitate Aggregate Spreading
Potential Effects on Neurodegeneration
Story Source:
Epidemiology And Symptoms Of Covid
COVID-19 has spread worldwide and was declared as a pandemic by the World Health Organization in March 2020. By March 24, 2021, a total of 123 902 242 confirmed cases of COVID-19 worldwide, including 2 727 837 deaths, have been recorded by the WHO . To date, there have been 85 countries, territories and areas that have had more than 100 000 cases each, with the United States having the largest number of confirmed cases.
SARS-CoV-2 belongs to the family Coronaviridae and is an enveloped non-segmented, single-stranded, positive-sense RNA virus. SARS-CoV-2 can spread rapidly through various routes including droplets, aerosol, and fomite . Moreover, fecal-oral and fecal-aerosol transmissions are suspected potential routes of SARS-CoV-2, as high viral loads have been detected in patients feces-related specimens . The majority of COVID-19 cases develop a lower respiratory tract infection, which leads to the high rate of viral transmission in densely populated areas or hospitals . The median age of COVID-19 patients was reported to be 59years , and more than half of them were males . People with low immune function, particularly the elderly and those with renal and hepatic dysfunction, are at a high risk of SARS-CoV-2 infection .
Does Alzheimer’s Make The Brain More Susceptible To Infection
The blood-brain barrier protects the brain by controlling what substances can pass from the blood into brain tissue. In Alzheimer’s disease, the blood-brain barrier is damaged, particularly in the brain region affected by Alzheimer’s.
Evidence suggests that inflammation, the Alzheimer’s hallmark amyloid protein and the ApoE4 gene, which are all linked to Alzheimer’s disease, can contribute to the breakdown of the blood-brain barrier. Once it has been weakened, bacteria, viruses, and other harmful substances can get through into the brain more easily. This may explain why certain viruses and bacteria, such as herpes and spirochetes, are more common in the brains of people with Alzheimer’s.
You May Like: Taking Aricept Without Alzheimer’s
Microbial Pathogenesis In Alzheimers Disease Grant
Intriguing evidence suggests Alzheimers disease may have a link to infectious diseases or a microbial mechanism. Could this be the missing link to lead to a cure for Alzheimers disease? To advance research that could shed light, and provide hope to the more than 5.7 Americans and 47 million people worldwide living with the disease, the IDSA Foundation established the Alzheimers Research Grant now known as the Microbial Pathogenesis in Alzheimers Disease Grant, in 2018 to foster further investigation. These grants support research that suggest evidence of an infectious agent or microbial community is correlated to Alzheimers disease and promotes novel research in the field of microbial triggers for Alzheimers disease.
Applications are now closed for the 2021 grant cycle. 2021 grant applicants and awardees will be notified of their application status in December 2021.
Therapeutic Implications Of The Infection Hypothesis

It would be very optimistic to imagine that an antibiotic therapy may be administered and cure, or at least prevent progression of AD. It is clear, however that no antibiotic regimen has been developed to cure periodontitis which has a similar implication of chronic infectious microorganisms and biofilms. However, accepting that senile plaques have biofilm components opens avenues to different possibilities to attack the integrity of the biofilm. Small molecules, vaccines and other means could be developed to target the biofilms or the microorganisms that created them. It can be also imagined that by recognizing the infectious etiology of AD, we will be able to discover early markers of the disease and we could develop new prevention trials targeting new molecules. It is clear that A should no longer be considered as the best or only promising target for the prevention and treatment of AD.
Read Also: What Is The Difference Between Dementia And Senility
Role Of Neuroinflammation And The Innate Immune System
Neuroinflammation is considered a hallmark of AD . The infection hypothesis provides the stimulus for this neuroinflammation. It also sheds light on two other fundamental issues: Neuroinflammation may not be entirely the consequence of A deposition but rather may itself cause A deposition as a protective response toward microbial challenge and It is not the case that A is exclusively a harmful molecule that aggregates to form plaque, but it is also a basic element of the innate immune defense system and thus a beneficial molecule as well, at least under certain conditions . Ultimately, as reactivations of infections become more frequent and chronic production of A increases, its antimicrobial effect may be blunted by loss of active A through recruitment to plaque formation inflammation becomes chronic and ultimately deposition proceeds and results in senile plaque formation . The deposition of plaque may be the initiator of the inflammatory process, maintain it and finally destroy the neighboring neurons. This process becomes visible clinically when a threshold is crossed.
Inflammation Linked To Alzheimer’s Disease Development
- Date:
- Memorial Sloan Kettering Cancer Center
- Summary:
- Scientists have discovered a direct link between the immune response to viruses and bacteria and the development of plaques in the brain that characterize Alzheimer’s disease.
Alzheimer’s disease is a neurodegenerative condition that is characterized by the buildup of clumps of beta-amyloid protein in the brain. Exactly what causes these clumps, known as plaques, and what role they play in disease progression is an active area of research important for developing prevention and treatment strategies.
Recent studies have found that beta-amyloid has antiviral and antimicrobial properties, suggesting a possible link between the immune response against infections and the development of Alzheimer’s disease.
Chemical biologists at the Sloan Kettering Institute have now discovered clear evidence of this link: A protein called IFITM3 that is involved in the immune response to pathogens also plays a key role in the accumulation of beta-amyloid in plaques.
“We’ve known that the immune system plays a role in Alzheimer’s disease — for example, it helps to clean up beta-amyloid plaques in the brain,” says Yue-Ming Li, a chemical biologist at SKI. “But this is the first direct evidence that immune response contributes to the production of beta-amyloid plaques — the defining feature of Alzheimer’s disease.”
Mounting Evidence for a New Hypothesis
Story Source:
Journal Reference:
Cite This Page:
Recommended Reading: Dementia Awareness Ribbon Color
Concerns About Previously Healthy People
The three new studies, presented at the annual Alzheimer’s Association International Conference, being held in Denver, each looked at different aspects of the link between COVID-19 and brain issues. None of the studies has yet been peer reviewed, the standard for scientific research, so their findings are preliminary.
One study, from Argentina, included nearly 300 people over 60. Interviewed three to six months after their initial infection, more than half reported lingering problems with forgetfulness and about a quarter still had speech or organizational challenges.
People with continued smell loss were more likely to have these issues, though there was no connection seen between long-term effects and the seriousness of someone’s initial infection.
Another study, done by researchers at New York University Langone Health, looked at 310 hospitalized COVID-19 patients over 60 for changes and cell death in their brains. About half had neurological symptoms, particularly confusion.
The patients with symptoms had higher blood levels of markers often associated with brain damage and Alzheimer’s.
Sperling said she found the results concerning, because they may suggest “there is a direct effect of COVID-19 on the brain that is potentially accelerating Alzheimer’s disease. That of course, is what we all worry about.”
Could Alzheimers Disease Stem From Infection
Could it be that Alzheimers disease stems from the toxic remnants of the brains attempt to fight of infection? Provocative new research by a team of investigators at Harvard leads to this startling hypothesis, which could explain the origins of plaque, the mysterious hard little balls that pockmark the brains of people with Alzheimers. The study, published in the journal Science Translational Medicine, posits that infections, including ones that are too mild to elicits symptoms, may produce a fierce reaction that leaves debris in the brain, causing Alzheimers. If it holds up, the hypothesis has major implications for preventing and treating AD.
The Harvard researchers reported the following scenario. A virus, fungus or bacterium gets into the brain passing through a membrane the blood-brain barrier that becomes leaky as people age. The brains defense system rushes in to stop the invader my making a sticky cage out of proteins, called beta amyloid. The microbe, like a fly in a spider web, becomes trapped in the cage and dies. What is left behind is the cage, a plaque that is the pathological hallmark of AD.
So far, the researchers have confirmed their hypothesis in neurons growing in petri dishes, as well as in yeast, roundworms, fruit flies, and mice. Work remains to be done to determine if humans might experience a similar outcome the funding for a stage 1 clinical trial is in place to commence the study shortly.
The answers, they reported, were yes and yes.
Recommended Reading: What’s The Difference Between Dementia And Alzheimer’s And Senility
The Alzheimer Disease Field And Infectious Hypothesis
The answer likely stems from the composition of the AD field. Though expanding and diversifying, most AD researchers are not microbiologists or virologists. Typically, they are neuroscientists, biochemists, neuropathologists, neuropsychologists, and pharmacologists. There is a distinct lack of overlap between the disciplines that is sometimes hard to bridge in terms of forming collaborations or productive dialogue. The number of individuals who work on AD and identify as a microbiologist or virologist is extremely small.
The longstanding support for A research is overwhelming, despite high failure rates for therapeutics centered on A modulation . NIH funding for AD research as of this articles writing sits at $2.3 billion, a funding level reflective of the imminent health concern AD represents. Of the NIH AD funding, a negligible amount is used to investigate pathogens in AD . However, the recent announcement of special interest by the National Institute on Aging for grants examining a potential connection between pathogens and AD represents an important step in increased funding in this area .
For those with greater interest in specific aspects of the infectious hypothesis, we would direct you to more comprehensive reviews written by Dr. Ruth Itzhaki, who frequently publishes scientific reviews on the topic.
Other Infections Linked To Dementia
Scientists have been investigating links between infection and dementia for years, said Avindra Nath, M.D., clinical director of the National Institute of Neurological Disorders and Stroke at the National Institutes of Health . Research shows just one episode of an infection you get hospitalized with pneumonia, for example can accelerate cognitive decline, especially if you’re already predisposed to memory problems.
“In general, we know that as you get older and get any kind of disease, even a urinary tract infection, it can bring out an underlying dementia, Nath said.
Respiratory viruses in particular seem to pack a powerful neurological punch.
“It’s possible I’d even say it’s probable that having COVID-19 will increase your risk of dementia.
After the severe acute respiratory syndrome outbreak in 2002 and the Middle East respiratory syndrome outbreak in 2012 both syndromes are caused by coronaviruses 1 in 5 recovered patients reported memory impairment, according to a review in The Lancet Psychiatry.
Before that, scientists studying the 1918 influenza pandemic noted an increase in cognitive decline, psychosis and Parkinson’s disease in those who had survived.
The neurological effects of COVID-19 range from mild symptoms such as headache and loss of taste and smell to more severe symptoms such as dizziness, confusion, seizures and stroke.
Also Check: Alzheimers Awareness Ribbons
A Whole New Hypothesis
When science converges from multiple independent laboratories like this, it is very compelling, says Casey Lynch of Cortexyme, a pharmaceutical firm in San Francisco.
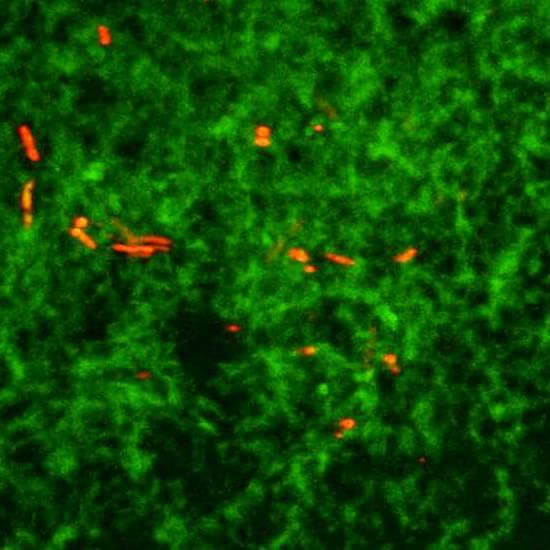
Now researchers from Cortexyme and several universities have reported finding the two toxic enzymes that P. gingivalis uses to feed on human tissue in 99 and 96 per cent of 54 human Alzheimers brain samples taken from the hippocampus a brain area important for memory . These protein-degrading enzymes are called gingipains, and they were found in higher levels in brain tissue that also had more tau fragments and thus more cognitive decline.
The team also found genetic material from P. gingivalis in the cerebral cortex a region involved in conceptual thinking in all three Alzheimers brains they looked for it in.
This is the first report showing P. gingivalis DNA in human brains, and the associated gingipains co-localising with plaques, says Sim Singhrao at the University of Central Lancashire, UK, who wasnt involved in the study. Her team has previously found that P. gingivalisactively invades the brains of mice with gum infections.
The Porphyromonas gingivalis bacteria that can cause gum disease
A. Dowsett, Public Health England/Science Photo Library
When the team gave P. gingivalis gum disease to mice, it led to brain infection, amyloid production, tangles of tau protein and neural damage in the regions and nerves normally affected by Alzheimers. This suggests causation, says Lynch.
Gum Disease And Alzheimers
Periodontitis, commonly known as gum disease, affects about 40 percent of American adults 30 years or older. Its also a risk factor for cognitive decline and Alzheimers. The bacteria that is the main culprit of the disease, Porphyromonas gingivalis, is present in the brains of more than 90 percent of people with Alzheimers.
Recent findings suggest that the bacteria may trigger an antibacterial response from beta-amyloid as well as impair the function of tau, a protein thought to play a role in regulating the communication between brain cells.
In a study published this month, researchers observed that, after they infected human brain neurons with Porphyromonas gingivalis bacteria, the bacteria reproduced and created toxins called gingipains. According to Stephen Dominy, an author of the study and the co-founder and chief scientific officer of biopharmaceutical company Cortexyme, a quarter of the brain cells died subsequently and the surviving neurons developed pathologies linked to Alzheimers, such as the breaking down of tau.
Dominy and other researchers also found in a study last year that Porphyromonas gingivalis may stimulate the production of beta-amyloid 42 peptide, a type of beta-amyloid. The peptides killed the bacteria, he explained, by binding onto and damaging its membrane.
And, the theory is supported by a growing body of research to suggest that beta-amyloid may be a natural defense mechanism of the brain.
Recommended Reading: Dementia Alzheimer’s Elephant Tattoo
Do Infectious Organisms Play A Role In Alzheimer’s
Complete the form below and we will email you a PDF version of“Do Infectious Organisms Play a Role in Alzheimer’s?”
First Name*Would you like to receive further email communication from Technology Networks?Technology Networks Ltd. needs the contact information you provide to us to contact you about our products and services. You may unsubscribe from these communications at any time. For information on how to unsubscribe, as well as our privacy practices and commitment to protecting your privacy, check out our Privacy Policy
Tips For Family Members
What should a patient, or a patients family, do to make sure that infections are not overlooked in someone with suspected Alzheimers disease or another dementia? First and foremost, make sure to express your concern to the clinicians who are caring for someone with an altered mental state. Also, it is crucial to ensure that the patients history is known by his or her health care providers. This is especially important if the cognitive changes are recent, or developed rapidly, or were associated with a known medical illness or known exposure to an infectious disease. Identifying an infection can require special tests of blood or cerebrospinal fluid that are not routinely ordered in many treatment settings. Awareness of a disease history or of an exposure may lead to further assessment and earlier identification of an infection even when typical medical symptoms such as fever, cough, or pain with urination are subtle or absent.
You May Like: Do People With Dementia Dream
How May All These Theories Be Reconciled
The existing data suggest that AD results from a progressive accumulation of noxious inflammatory processes or events in the brain fuelled by multiple infectious agents that colonize/infect the body. Local neuroinflammation may continue at a low level throughout life with little negative effect. However, when exacerbated by reactivation of infections combined with other insults , including age-related insults like increasing amounts of senescent cells, the acute inflammatory response results in unbalanced production of cytotoxic mediators, such as TNF, which, accompanied by immunosenescence/inflamm-aging, becomes difficult to control or stop . Microbial metabolites may not only fuel neuroinflammation, but also contribute to senile plaque formation when their biofilm components are integrated into plaque. The enhanced neuroinflammatory process damages neurons and alters the . These mediators also induce peripheral inflammation and then return to further stimulate local neuroinflammation . This progressive pro-inflammatory situation is exacerbated with age, creating a vicious cycle of local and systemic inflammatory responses leading to activation of cytotoxic microglia, unbalanced cytokine production, A accumulation and irreversible brain damage.