What Gets Stored In A Cookie
This site stores nothing other than an automatically generated session ID in the cookie no other information is captured.
In general, only the information that you provide, or the choices you make while visiting a web site, can be stored in a cookie. For example, the site cannot determine your email name unless you choose to type it. Allowing a website to create a cookie does not give that or any other site access to the rest of your computer, and only the site that created the cookie can read it.
Could Certain Microbes Act As A Trigger That’s The Central Premise Of The Infection Hypothesis
While it had long been known that the virus could infect the brain leading to a dangerous swelling called encephalitis that required immediate treatment this was thought to be a very rare event. In the early 1990s, however, Itzhaki’s examinations of post-mortem tissue revealed that a surprising number of people showed signs of HSV1 in their neural tissue, without having suffered from encephalitis.
Importantly, the virus didn’t seem to be a risk for the people without the APOE4 gene variant, most of whom did not develop dementia. Nor did the presence of APOE4 make much difference to the risk of people without the infection. Instead, it was the combination of the two that proved to be important. Overall, Itzhaki estimates that the two risk factors make it 12 times more likely that someone will develop Alzheimer’s, compared to people without the gene variant or the latent infection in their brain.
As far back as the 1990s, a surprising number of people showed signs of HSV1 in their neural tissue
Itzhaki hypothesised that this was due to repeated reactivation of the latent virus which, during each bout, invades the brain and somehow triggers the production of amyloid beta, until eventually, people start to show the cognitive decline that marks the onset of dementia. “I think there has to be repeated activation and accumulated damage to explain the long-term course of the disease.”
Thinking Outside The Box In Alzheimers Disease: Could Infection Be The Answer
The third speaker was Professor Brian Balin from the Center for Chronic Disorders of Aging at the Philadelphia College of Osteopathic Medicine, and his talk was entitled Thinking outside the box in Alzheimers Disease: Could infection be the answer?
Upon finding infection in the AD brains and evaluating by reverse transcriptase PCR, transcripts for Chlamydia pneumoniae were obtained from frozen tissue samples. Given these results, it may be possible for some organisms found in frozen tissue to be cultured in vitro. To determine if this was possible, homogenates of human brain tissues were cultured with human THP1 monocytes with positive results . The organism was confirmed to be present and preserved in frozen brain tissues by this method. That the organism propagated in vitro intracellularly in the monocytes was confirmed using antibodies specific to Chlamydia pneumoniae and by PCR. Propagation in this manner was required as this bacterial organism is an obligate intracellular pathogen that will only grow in an intracellular environment.
A42 deposits in the CNS at 2 months post-infection following intranasal infection with Chlamydia pneumoniae. Brains were examined by light microscopy for the presence of A42 using a specific anti-A 142 antibody. Mag bars = 20 m .
You May Like: What Is The Difference Between Dementia And Senility
Could Depression Be Caused By An Infection
So does that mean that the microbe is the cause of Alzheimer’s, and amyloid a harmless reaction to it? According to Tanzi it’s not that simple.
Tanzi believes that in many cases of Alzheimer’s, microbes are probably the initial seed that sets off a toxic tumble of molecular dominoes. Early in the disease amyloid protein builds up to fight infection, yet too much of the protein begins to impair function of neurons in the brain. The excess amyloid then causes another protein, called tau, to form tangles, which further harm brain cells.
But as Tanzi explains, the ultimate neurological insult in Alzheimer’s is the body’s reaction to this neurotoxic mess. All the excess protein revs up the immune system, causing inflammation and it’s this inflammation that does the most damage to the Alzheimer’s-afflicted brain.
So what does this say about the future of treatment? Possibly a lot. Tanzi envisions a day when people are screened at, say, 50 years old. “If their brains are riddled with too much amyloid,” he says, “we knock it down a bit with antiviral medications. It’s just like how you are prescribed preventative drugs if your cholesterol is too high.”
Tanzi feels that microbes are just one possible seed for the complex pathology behind Alzheimer’s. Genetics may also play a role, as certain genes produce a type of amyloid more prone to clumping up. He also feels environmental factors like pollution might contribute.
Ulcers, we now know, are caused by a germ.
Senile Plaques And Neurofibrillary Tangles
Tau is an essential neuronal microtubule-associated protein that tends to localize to the axon due to its main function of stabilizing the formation of microtubules to provide structural support for the neuron . There are several cellular processes in the brain that are dependent on tau, such as axonal growth and vesicle/organelle transport . The phosphorylation of tau at Ser214 and Thr231 can lead to the disruption of these cellular processes due to the detachment of tau from the microtubule . In a normal brain there is a balance of phosphorylated and non-phosphorylated tau that is regulated by the balance of activity between tau kinases and phosphatases . In an AD patient, the tau kinase/phosphatase balance is impaired, with the balance being skewed towards tau kinases leading to hyperphosphorylated tau . When tau is hyperphosphorylated, it assembles into tangles . When the hyperphosphorylated form of tau is prominent, cellular processes that rely on tau are disrupted and axonal and dendritic transport are compromised. Interestingly, A contributes to phosphorylated tau protein via activation of tau kinases .
Don’t Miss: Alzheimer Awareness Ribbon Color
It’s A Result That Is So Striking It’s Hard To Believe
As interest in the infection hypothesis has grown, scientists have started to investigate whether any other pathogens may trigger a similar response with some intriguing conclusions. A 2017 study suggested that the virus behind shingles and chickenpox can moderately increase the risk of Alzheimer’s disease. There is also evidence that Porphyromonas gingivalis, the bacterium behind gum disease, can trigger the accumulation of amyloid beta, which may explain why poor dental health predicts people’s cognitive decline in old age. Certain fungi may even penetrate the brain and trigger neurodegeneration. If the causal role of these microbes is confirmed, then each finding could inspire new treatments for the disease.
Scientists studying the infection hypothesis have also started making some headway in explaining the physiological mechanisms.
Their explanation centres on the surprising discovery that amyloid beta can act as a kind of microbicide that fights pathogens in the brain. Studies by Fulop and others, for instance, show that the protein can bind to the surface of the herpes simplex virus. This seems to entrap the pathogen with a web of tiny fibres and prevents it from attaching to cells.
Amyloid beta can act as a kind of microbicide that fights pathogens in the brain
Double threat
Is There A Link Thats Still A Big Maybe
Other experts caution that the science is not yet strong enough to prove germs are the cause or, more likely, one of the causes of dementia. And because several of the germs found more frequently in the brains of people with Alzheimer’s are ubiquitous, scientists suspect that if they are involved, they are likely part of a complex interplay of genes, age, inflammation, environmental exposures, head trauma, and metabolic factors.
“It is clear that there are more and more data being accumulated that point to a connection of some kind between viral sequences and Alzheimer’s in the brain,” said Richard Hodes, director of the National Institute on Aging. He added, however, that a correlation between the presence of microbes in the brain and Alzheimer’s pathology does not prove one causes the other.
Eliezer Masliah, director of the NIA’s division of neuroscience, said the agency is funding research into potential treatments from “multiple directions.” It has targeted funding for how the body’s microbiome affects the nervous system and why people with HIV are at higher risk for dementia. Hodes said the NIA will consider whether to make research on germs and Alzheimer’s a higher priority during the next round of funding.
“Infection is the prequel to the amyloid hypothesis, not a replacement,” said Tanzi, who is also known for identifying three genes involved in early-onset Alzheimer’s.
Don’t Miss: Do People With Dementia Dream
Imbalance In Gum Bacteria Linked To Alzheimers Disease Biomarker
Increases in bad bacteria and decreases in good bacteria in gums associated with amyloid beta
Older adults with more harmful than healthy bacteria in their gums are more likely to have evidence for amyloid betaa key biomarker for Alzheimers diseasein their cerebrospinal fluid , according to new research from NYU College of Dentistry and Weill Cornell Medicine. However, this imbalance in oral bacteria was not associated with another Alzheimers biomarker called tau.
The study, published in the journal Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring, adds to the growing evidence of a connection between periodontal disease and Alzheimers. Periodontal diseasewhich affects 70 percent of adults 65 and older, according to CDC estimatesis characterized by chronic and systemic inflammation, with pockets between the teeth and gums enlarging and harboring bacteria.
To our knowledge, this is the first study showing an association between the imbalanced bacterial community found under the gumline and a CSF biomarker of Alzheimers disease in cognitively normal older adults, said Angela Kamer, DDS, PhD, associate professor of periodontology and implant dentistry at NYU College of Dentistry and the studys lead author. The mouth is home to both harmful bacteria that promote inflammation and healthy, protective bacteria. We found that having evidence for brain amyloid was associated with increased harmful and decreased beneficial bacteria.
Press Contact
Infectious Hypothesis Of Alzheimer Disease
- Charles E. Seaks,
Citation: Seaks CE, Wilcock DM Infectious hypothesis of Alzheimer disease. PLoS Pathog 16: e1008596. https://doi.org/10.1371/journal.ppat.1008596
Editor: Rebecca Ellis Dutch, University of Kentucky, UNITED STATES
November 12, 2020
Copyright: © 2020 Seaks, Wilcock. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Funding: The authors have received no funding for this work.
Competing interests: The authors have declared that no competing interests exist.
Read Also: What Color Ribbon Is Alzheimer’s
Nature Portfolio Microbiology Community
Copy the link
A amyloid fibrils were produced in the brain and bound to invading pathogens, eventually encapsulating them in a “prison”.
This has never been show in humans and whether or not infection could really be a clinical cause of Alzheimer’s Disease remains to be seen. At the very least though, inflammation pathways could now be investigated as new targets for the treatment/prevention of Alzheimer’s.
Setting Your Browser To Accept Cookies
There are many reasons why a cookie could not be set correctly. Below are the most common reasons:
- You have cookies disabled in your browser. You need to reset your browser to accept cookies or to ask you if you want to accept cookies.
- Your browser asks you whether you want to accept cookies and you declined. To accept cookies from this site, use the Back button and accept the cookie.
- Your browser does not support cookies. Try a different browser if you suspect this.
- The date on your computer is in the past. If your computer’s clock shows a date before 1 Jan 1970, the browser will automatically forget the cookie. To fix this, set the correct time and date on your computer.
- You have installed an application that monitors or blocks cookies from being set. You must disable the application while logging in or check with your system administrator.
Recommended Reading: Can Cause Sores Rashes Dementia Or Blindness
Antibiotics In The Frame
The worst offenders for causing protein clumping in the worms were two bacterial species called Klebsiellapneumoniae and Pseudomonasaeruginosa.
Interestingly, research has linked these species to an increased risk of protein conformational diseases, such as Parkinsons.
In their paper, the researchers note the increasing prevalence of antibiotic resistance in these species in recent years.
They speculate that antibiotic treatments may, therefore, have the perverse effect of encouraging the growth of these antibiotic-resistant bad bacteria while decreasing the abundance of good bacteria.
In support of this hypothesis, a recent population-based study found that a history of antibiotic therapy is associated with an increased risk of Parkinsons.
Gum Disease And Alzheimers
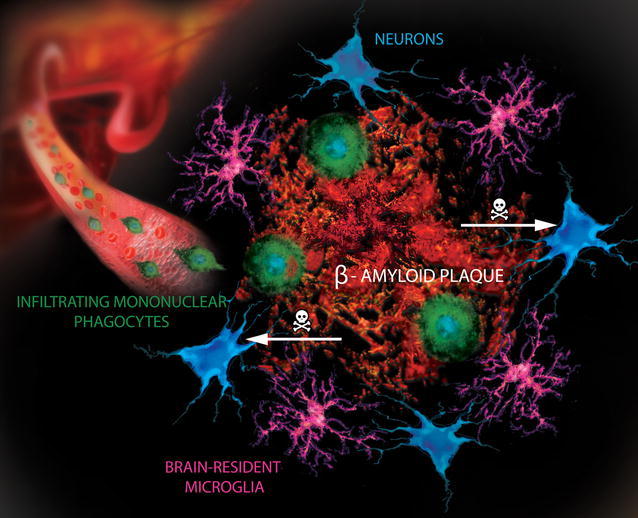
Periodontitis, commonly known as gum disease, affects about 40 percent of American adults 30 years or older. Its also a risk factor for cognitive decline and Alzheimers. The bacteria that is the main culprit of the disease, Porphyromonas gingivalis, is present in the brains of more than 90 percent of people with Alzheimers.
Recent findings suggest that the bacteria may trigger an antibacterial response from beta-amyloid as well as impair the function of tau, a protein thought to play a role in regulating the communication between brain cells.
In a study published this month, researchers observed that, after they infected human brain neurons with Porphyromonas gingivalis bacteria, the bacteria reproduced and created toxins called gingipains. According to Stephen Dominy, an author of the study and the co-founder and chief scientific officer of biopharmaceutical company Cortexyme, a quarter of the brain cells died subsequently and the surviving neurons developed pathologies linked to Alzheimers, such as the breaking down of tau.
Dominy and other researchers also found in a study last year that Porphyromonas gingivalis may stimulate the production of beta-amyloid 42 peptide, a type of beta-amyloid. The peptides killed the bacteria, he explained, by binding onto and damaging its membrane.
And, the theory is supported by a growing body of research to suggest that beta-amyloid may be a natural defense mechanism of the brain.
Read Also: What Color Ribbon Is Alzheimer’s
The Role Of Herpes Simplex Virus Type 1 In Alzheimers Disease
In the 2017 IAGG symposium held in San Francisco, the first speaker was Professor Ruth Itzhaki from the Universities of Oxford and Manchester, and her talk was entitled, The role of herpes simplex virus type 1 in Alzheimers disease .
Dr. Itzhakis laboratory first discovered in 1991 that HSV1 DNA is present in a high proportion of the brains of both AD patients and elderly normal subjects . Subsequently, six other groups detected HSV1 in human brain . The fact that HSV1 is present in elderly normal people as well as AD patients does not preclude a viral role. Most viruses infect far more people than they affect: genetic factors can determine who is asymptomatic and who suffers disease. Indeed, found that HSV1 DNA in the brains of carriers of an apolipoprotein E-4 allele confers a high risk of developing AD. A study by another group confirmed the HSV1-APOE-4 association in AD, and work on HSV1-infected APOE-transgenic mice has shown that APOE-4 animals display a greater viral load, and a greater potential for viral damage . Significantly, APOE-4 is a risk also for cold sores , which are usually caused by HSV1 in the peripheral nervous system, suggesting that the damage caused by HSV1 is greater, or that repair is lesser, in APOE-4 carriers.
Quantification of HSV1 proteins, -amyloid and abnormal tau phosphorylation in HSV1-infected cells after acyclovir treatment .
Gut Metabolites And A Clearance
Found in the ileum and the colon, gut microbiomes produce biologically active short-chain fatty acids which can cross the bloodâbrain barrier , by fermenting the fibrous foods. The experimental studies have indicated that the gut metabolites can increase inflammation, tau and Aβ aggregations in CNS, which has been validated by meta-analysis .
Gut bacteria secrete more than 100 metabolites, but effects of the most on the pathogenesis of AD have not been elucidated. Valeric acid and isovaleric acid, isobutyric acid and butyric acid, propionic acid, acetic acid and formic acid have been studied and have been found to affect the pathogenesis of AD by disturbing the activations of microglia and astrocyte, help to reduce inflammation, and aggregations of Aβ and tau .
Recommended Reading: What Is The Difference Between Dementia And Senility
Oral Bacteria May Be Responsible For Alzheimers Disease
Alzheimers disease is the leading cause of dementia. It progressively worsens multiple aspects of health over time, from short-term memory loss to behavioral changes to loss of bodily functions. The actual cause of Alzheimers is currently unknown. One widely-accepted hypothesis proposes that Alzheimers is caused by the accumulation of misfolded proteins in the brain. Unfortunately, many drugs targeting misfolded proteins perform poorly in clinical trials, hinting that this hypothesis might be wrong. Misfolded proteins might be another side effect, not the cause.
Researchers recently published a new line of evidence supporting a hypothesis that Alzheimers might be a result of an infection by oral bacteria P. gingivalis. The bacteria produces toxins called gingipains that are found to accumulate in the brain of Alzheimers patients. The gingipains degrade human proteins, giving rise to the infamous misfolded proteins. The researchers also developed chemical compounds that could neutralize gingipains. Mice injected with gingipains developed degenerate brain cells, while mice that were pretreated with neutralizing compounds beforehand maintained healthy brain cells.
Managing Correspondent: Veerasak Srisuknimit
Microbes Take The Stage
The study that spurred last week’s headlines was published on Jan. 23 in the journal Science Advances. In this study, researchers suggested that Porphyromonas gingivalis, the bacteria that cause a common type of gum disease,may also play a role in Alzheimer’s. The researchers found that people with Alzheimer’s had these bacteria in their brains.
The majority of the study was conducted in mice, however. In those animals, the team showed that the bacteria were able to travel from the mouth to the brain, where they could inflict damage on brain cells and increase the production of the beta-amyloid proteins that cause the telltale plaques of Alzheimer’s. What’s more, the researchers found that they could stop this damage in mouse brains by targeting toxic enzymes produced by the bacteria.
Patira noted that while the new study is “well-done it was done in mice, and mice’s brains are similar to but not the same.” Many studies looking at late-stage Alzheimer’s have been conducted on mice some were successful, some not. But even the successful ones, one after the other, failed when they reached the human stage, Patira said.
In any case, while staying skeptical that the new findings will hold true in humans, she said that the results are “promising.”
The microbes most commonly suspected of playing a role in Alzheimer’s are herpesviruses.
But even if a virus plays a role in the development of the disease, it’s likely not the only factor.
Don’t Miss: What Color Ribbon Is Alzheimer’s