Induced Pluripotent Stem Cells
iPSCs were first obtained from mouse fibroblasts in 2006. They are derived in vitro from mature somatic cells, usually adult dermal fibroblasts, by small-molecule therapy or viral vectormediated upregulation of transcription factors. Genetic modification makes them pluripotent and ESC-like in terms of phenotypic and differentiation capacity.
However, the following unresolved questions about the use of iPSCs pose huge obstacles to their clinical application: Teratoma formation, long-term safety and effectiveness, tumorigenicity, immunogenicity, patient genetic defects, optimal reprogramming and so forth.
Genetics Of Ad Pathogenesis
Studies have shown that two typical misfolded proteins accumulate in the brain of patients with AD. The first is A, which is a pathological cleavage product of amyloid precursor protein . The accumulation of A into plaques and smaller oligomers is one of the pathological features of AD. APP mutations have been confirmed to be associated with hereditary familial AD. Familial AD is an early-onset autosomal dominant genetic disease. The age of onset is less than 65 years, but it only accounts for 2% of all AD cases . Many failed clinical trials targeted this pathway directly or indirectly through small-molecule or antibody therapies to reduce A production or promote A clearance. The second misfolded protein in AD is tau, a microtubule-associated protein that aggregates in cells in the form of neurofibrillary tangles. The most closely related pathological feature is AD cognitive decline. However, the vast majority of cases of AD, which do not involve mutations in APP processing pathways, are sporadic, and the age of onset is more than 65 years. For this population, the main predictive factor for AD is the genetic risk factor apolipoprotein E4, in addition to age. APOE4 carriers account for 60%75% of AD cases. Compared with noncarriers, patients with AD and APOE4 are younger.
Can Stem Cell Research Help With Alzheimers
It has long been hoped that stem cell therapies could be used to repair the damage caused by diseases such as Alzheimers.
Although stem cell biology is a relatively new field, promising animal studies are beginning to show how stem cells might be able to treat diseases of the brain.
New research from Professor Eva Feldman at the University of Michigan has been testing this idea.
Her research group placed human neural stem cells into the brains of mice that were showing the symptoms of Alzheimers. The transplanted cells were put into the hippocampus, an area of the brain responsible for memory. The mice were then monitored to look for any changes in their thinking and memory skills.
Also Check: Dementia Awareness Ribbon Color
Intracerebroventricular Injection Of Bm
At 12 months of age, female APP/PS1 and WT mice were implanted with a stainless steel cannula , as described previously. The cannula was placed at 0.4 mm posterior from the bregma, 1 mm right of the midline, and 2.5 mm deep to reach the right ventricle of the brain. At 13 months of age, female APP/PS1 and WT mice were injected with BM-MSCs ) or vehicle through the cannula with a Hamilton syringe at a rate of 1 l/min. After intracerebroventricular injection two times at a 2-week interval, the MWM test was conducted .
To evaluate the distribution of BM-MSCs, we injected WT and APP/PS1 mice at 1314 months old with PKH-labeled BM-MSCs into the right ventricle via the cannula. BM-MSCs were stained with PKH26 , according to the manufacturers protocol. Mice were sacrificed at 1, 14, and 28 days after injection of PKH-labeled BM-MSCs .
A Better Way To Study Disease
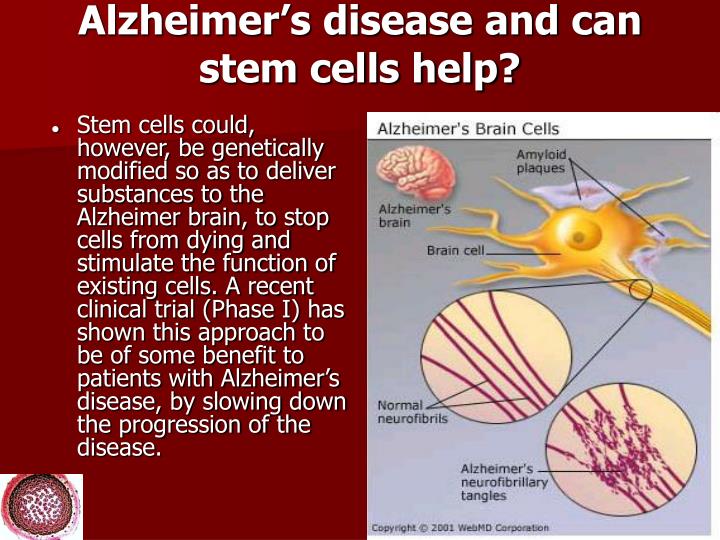
Harvard Stem Cell Institute scientists use a different set of tools to understand disease. With the advent of induced pluripotent stem cell technology the biological reprogramming of mature cells into stem cells it is possible to generate stem cells from the skin of an adult Alzheimers patient and then direct these cells to become different types of brain cells. HSCI investigators can direct patient stem cells to become brain cells both affected and not affected in Alzheimers disease. Our scientists plan to examine the types and amounts of Aß proteins generated by these cell types, and to look at how the different cell types respond to plaques.
None of the Food and Drug Administration approved treatments on the market for Alzheimers tackle the underlying cause of the disease. Rather, they attempt to alleviate symptoms associated with the loss of brain cells. Using stem cells, HSCI scientists can reveal potential targets for therapeutic intervention by studying the differences between brain cell types, and between those suffering from the disease and those who are healthy.
Also Check: Senility Vs Dementia
Alzheimers Syndrome And Consequences Of The Disease
Alzheimers syndrome is caused by the progressive death of brain cells. It often results from a certain combination of genetic, behavioural and environmental factors. Scientists do not yet understand entirely what causes this illness, but it is obvious that AD severely damages the brain.
Affected by this disease, the brain loses its cells and connections between them. Along with cells dying, the brain undergoes certain shrinkage. At first, the only symptoms of this process are mild confusion or increasing forgetfulness. However, in time, AD robs a patient of most of the memories. If a person does not undergo serious Alzheimers therapy, he or she starts suffering from disorientation, speaking and writing problems, social withdrawal, anxiety and depression. Patients suffering from this disease lose their ability to think and reason, make judgments and decisions, plan and perform even simple tasks.
Stem Cell Alzheimers Disease Research
Could stem cell Alzheimers Disease research provide a potential path to future treatment or preventative measures?
The goal of todays post is to provide a review of the state of the field in this area including possible approaches and challenges. You can watch a video covering the topic by Dr. Paul Knoepfler below on our stem cell YouTube channel. If you like this or our other videos please subscribe.
You May Like: Bobby Knight Health Update
How Are Stem Cells From Stem Cell Supplement Administered For Alzheimers Disease
Unlike traditional Stem Cell Therapy which can only be applied via injection in the selective hospitals/ clinics, Stem Cell Supplement delivers stem cells through high-tech bio-active softgel capsules which can be shipped in secure packages and delivered right to your home. The lyophilized method employed in softgel capsules produces stem cells which remain biologically active without damaging the effectiveness of the valuable, big, bio-active matter. Being enteric coated, softgel capsules by-pass the stomach and dissolve in the small intestine whereby the stem cells and other active ingredients are fully absorbed by the body. Therefore, to receive the results and benefits of Stem Cell Supplement, all you need to do is to take 1 3 softgel capsules a day at the comfort of your own home.
Alzheimers Stem Cells Treatment At Swiss Medica I Century
Stem cell therapy the key treatment mode used in our clinic is based on the insertion of new adult stem cells into mutilated tissue. Many specialists are sure that this method has the potential to alleviate the suffering of patients with AD. Stem cells have the unique ability to self-renew they stimulate subsequent generating of certain tissues and the whole body.
The method of stem cells in Alzheimers treatment does not involve any donors. To create new beta cells we use the cells contained in the patients bone marrow and/or fat tissue. These mesenchymal stem cells help to repair different tissue types after they are injected into the human body. Having immune-modulatory properties, which are similar to those of cells from different origins, a patients own stem cells help in the regrowth of healthy brain tissue.
This method allows our specialists to repair the damage caused to people with AD in the progressive stages of the disease. Numerous scientific researches prove that stem cells make a great contribution to rebuilding damaged nerve fibers. Additionally, because mesenchymal stem cells are a part of patients body naturally, there is no risk of rejection, side effects or allergic reactions.
Read Also: 7th Stage Of Alzheimer’s
How Do Scientists Get Stem Cells To Specialize Into Different Cell Types
One of the biggest hurdles in any stem cell-based therapy is coaxing stem cells to become a single cell type. The vital process of maturing stem cells from one state to another type is called differentiation.Guiding stem cells to become a particular cell type has been fraught with difficulty. For example, stem cells growing in a developing embryo receive a carefully choreographed series of signals from the surrounding tissue. To create the same effect in the lab, researchers have to try and mimic those signals. Add the signals in the wrong order or the wrong dose and the developing cells may choose to remain immatureor become the wrong cell typeMany decades of research has uncovered many of the signals needed to properly differentiate cells. Other signals are still unknown. Many CIRM-funded researchers are attempting to differentiate very pure populations of mature cell types that can accelerate therapies.
Find Out More:
Isolation Of Exosomes From The Cm Of Bm
When BM-MSCs reached 90% confluency after three passages in 15-cm dishes , the cells were washed with PBS, and the conventional medium was replaced with 10 ml FBS-free alpha-minimal essential medium. After incubation for 24 h, the total culture medium was collected. MagCapture Exosome Isolation Kit PS was used to isolate exosomes from the medium, according to the manufacturers protocol. Exosomes were then labeled with PKH , according to the manufacturers protocol. As a control, exosomes isolated from PBS using MagCapture Exosome Isolation Kit PS were labeled with PKH.
Don’t Miss: Alzheimers Ribbon Color
How Are The Stem Cells Collected
We use only umbilical cord stem cells that are derived exclusively from umbilical cord donations. The umbilical cord stem cells are collected after informed consent has been given by the parent, or parents, and only after the delivery of the baby.The collection follows strict ethical protocols ensuring the stem cells are from safe, reliable sources using a non-invasive, simple and painless procedure.Once collected, the cord blood is then screened for disease. Once the cells are collected, our lab uses the most advanced technology to isolate, cultivate, expand and test the mesenchymal stem cells. The entire process takes three weeks to ensure the lab produces stemcells that are safe and efficacious.
Why Does It Take So Long To Create New Stem Cell Therapies
Stem cells hold the potential to treat a wide range of diseases. However, the path from the lab to the clinic is a long one. Before testing those cells in a human disease, researchers must grow the right cell type, find a way to test those cells, and make sure the cells are safe in animals before moving to human trials.Find Out More:Hans Keirstead talks about hurdles in developing a new therapy
Don’t Miss: Can Prevagen Help Dementia
Causes Of Alzheimers Disease
A combination of genetic, lifestyle and environmental factors may cause the onset of Alzheimerâs. The exact cause of the disease is currently unknown, but scientists do know that the emergence of Alzheimerâs coincides with the brainâs inability to function normally.
Alzheimerâs may also be characterized by an increase in amyloid and tau protein buildup within the brain though this is a symptom of Alzheimerâs, it has not been proven to be the cause of the disease.
The Aduhelm Case & Development Of Stem Cell Therapy For Alzheimers
A recent development this summer illustrates the broader challenges of trying to develop drugs for Alzheimers. This article in the Washington Post does a nice job covering the news that the FDA allowed the first accelerated approval of an Alzheimers drug, Aduhelm, prompting controversy including over the underlying cause of the disease.
Historically, the amyloid hypothesis held that plaques, or clumps of amyloid beta, are the major causes of AD. However, researchers are now mostly concurring that the connection between amyloid beta and AD is more complex. Though Aduhelm has been shown to remove plaque and slow down cognitive decline, the controversy is centered on its fast approval even after an FDA advisory panel concluded there was a lack of sufficient clinical evidence. Several experts have opposing views on the FDAs decision for this new drug, but some think that Aduhelm may provide a modest clinical benefit to certain patient subgroups.
There seems to be a growing consensus that only targeting amyloid plaques will not be adequate in the treatment of AD. We will have to see how the stem cell clinical research in this area proceeds to know if a stem cell approach might provide additional concrete options.
Also Check: Does Diet Coke Cause Alzheimer’s
Stem Cell Trials In Humans
Inconsistencies in preclinical studies have prevented several potential stem cell therapies from transitioning to human clinical trials. By contrast, evidence for the safety and efficacy of MSC-based therapies in animal models, combined with ease of handling and isolation, has supported the approval of several human clinical trials.
Details of ongoing trials are summarised in Table . While many of these employ an intravenous infusion administration route, one trial will assess the safety and efficacy of intraventricular MSC injection via an Ommaya reservoir system. Umbilical cord blood-derived MSCs remain a common cell choice, although key differences exist with regards to cell number, dose number, and dose schedule. Two separate trials, both currently undergoing recruitment, will utilise alternative MSC sources. One trial will assess the safety and efficacy of autologous adipose-derived stromal vascular fraction cells acquired from patient liposuction. Another study will utilise ischaemia-tolerant allogeneic human bone marrow-derived MSCs. Grown under hypoxic conditions to more closely resemble the physiological environment of the CNS, these MSCs express higher levels of angiogenic growth factors, including VEGF and angiopoietin, and show enhanced migratory activity .
Table 2 Ongoing stem cell trials in humans with Alzheimers disease
What Is Unique About Receiving Stem Cell Therapy Through Trustem Cell Therapy For Alzheimers Disease
-
Our focus is safety, efficacy, and patient-centric care when providing access to superior stem cell therapy.
-
We utilize only board certified surgeons, physicians and accredited clinicians to provide care for patients.
-
Laboratory protocols are developed and refined by our PhD Neuroscientist.
-
A clinical team with expertise in practicing cellularbased medicine.
-
Accredited Surgical Centers for enhanced procedural and patient safety
-
Targeted administration methods that direct stem cells toward specificareas of injury which include the joints, brain, spine, lungs, etc
-
Skilled Patient Advocates who are trained to provide truthful, realistic expectations resulting from stem cell therapy. We do not make outlandish promises of cures or inaccurate claims related to improvement rates.
Don’t Miss: Farts And Dementia
Advantages Of Stem Cell Therapy At Swiss Medica Clinic
Adult Autologous Stem Cell Therapy at Swiss Medica I century is the innovative method of treatment used for a number of diseases. The absence of rejection, side effects and immune reactions are explained by the relationship between the organism and its own mesenchymal stem cells. This is why a patient cannot be infected by transmissible illnesses. As adult stem cells are mature enough, the clients in our clinic are not in danger of oncological complications.
During the therapy process, no general anaesthesia is needed. Our specialists take small amounts of body tissue in order to reach the stem cells, placed in either bone marrow or abdominal area, which are both easily accessible. After this simple process is completed, stem cells are separated from the tissue and activated. Finally, they are returned to the patients body using a simple IV dip. All of this is done within only a couple of hours. The procedure is completely safe, quick, simple and painless. As for the results of stem cell Alzheimers treatment, they are generally very successful.
At Swiss Medica clinic, we take care of every patient and we always aspire to demonstrate the best results of our investigations and work. The team of highly qualified specialists reinforced with the newest equipment, provide our clients with safe and effective treatment, proper aftercare and further medical help.
The advantages of our treatment system:
Cost Of Stem Cell Treatments
The cost of the initial treatment ranges from $5,000 to $10,000. The range in cost is dependent on the complexity of delivering the cells back to you. For example, spine conditions require multiple physicians to deliver the cells back to your body and this requires an increase in cost as multiple doctors are involved in the procedure. For many people the initial treatment procedure is all that is needed however, for some conditions, subsequent treatments may be required and these are done at a reduced fee. Stem Cell Therapy procedures for Alzheimers patients are done at both our Dallas and Fort Worth practice locations.
Recommended Reading: Alzheimer Ribbon Color
Types Of Stem Cells: Characteristics Benefits And Limitations
SCs are undifferentiated cells that have the ability of selfrenewal to replicate rapidly and continuously, as well as giving rise to new SCs and specialized cells under appropriate conditions. Asymmetric mitosis is the process that allows a SC to obtain two intrinsically different daughter cells. Cell fate-determining factors are provided extrinsically or inherently to SCs in a polarized manner. By coordinating the cell axis polarity, the daughters of the SCs acquire distinct fates: self-renew or commit to differentiation . The types of SCs vary according to their differentiation potential , which reduces at each step. This means that a unipotent SC cannot differentiate into as many cell types as a pluripotent SC. The zygotes are totipotent cells, the most developmentally expansive cells. Yet they are rarely considered an SCs in mammals because they cleave into blastomeres of equal developmental potency for at most three cell divisions, and therefore manifest minimal self-renewal potential . Below, we will describe and discuss the characteristics and the advantages and disadvantages of the three main types of SCs.
How Do Stem Cells Help Alzheimer’s
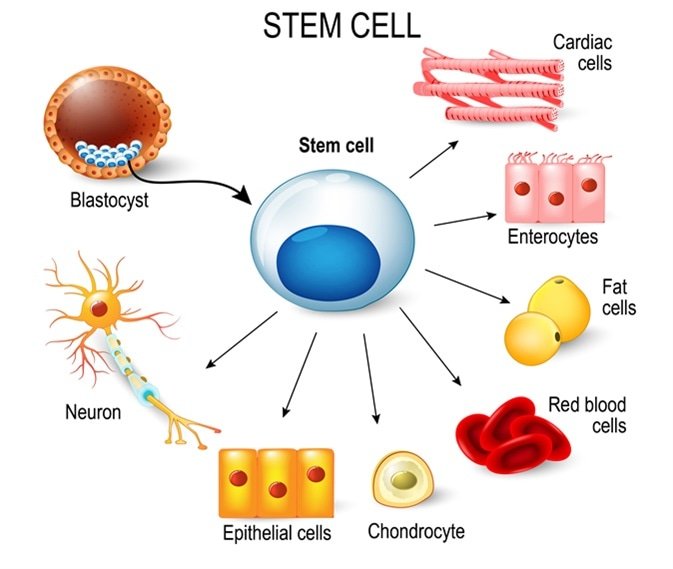
Mesenchymal Stem Cells or stromal stem cells can differentiate into many different types of cells within the body, including bone cells, cartilage, muscle cells, and even neural cells. They are primarily found in the bone marrow of every person and remain dormant until called upon to promote healing within the body. They age as we age, and their number and effectiveness decreases over the years.
According to Sung S. Choi and colleagues in a 2014 study titled Alzheimerâs Disease and Stem Cell Therapy
âStem cells have therapeutic effects using regeneration and substitution of cells and tissues themselves. The therapeutic strategy of stem cells has two directions. One is to induce the activation of endogenous stem cell, and the other is to regenerate the injured cell or tissues through stem cell transplantationâ
Don’t Miss: Does Prevagen Help Dementia