Genetics Of Complex Diseases
The genetics of complex diseases can be best explained by the model of Manolio et al., which was adapted for AD from Lane et al. . Here, the significance of a genetic variant is quantified on the basis of frequency on the one hand and effect of the variant on the other hand. As illustrated, high penetrant alleles have a rather low allele frequency in the population, whereas low susceptibility alleles such as variants found in genome-wide association studies are common in the population.
Risk genes associated with AD. Previously identified genetic variants, represented by the frequency of risk alleles and the strength of the genetic effect. Colors in the legend indicate pathways in which the genes are involved.
Study Reveals How Genetic Changes Lead To Familial Alzheimers Disease
A new mouse model of neurodegenerationImage courtesy of Raymond Kelleher and Jie Shen, Harvard Medical School.
NIH-funded research suggests novel approach for developing drugs for inherited Alzheimers
Mutations in the presenilin-1 gene are the most common cause of inherited, early-onset forms of Alzheimers disease. In a new study, published in Neuron, scientists replaced the normal mouse presenilin-1 gene with Alzheimers-causing forms of the human gene to discover how these genetic changes may lead to the disorder. Their surprising results may transform the way scientists design drugs that target these mutations to treat inherited or familial Alzheimers, a rare form of the disease that affects approximately 1 percent of people with the disorder. The study was partially funded by the National Institute of Neurological Disorders and Stroke , part of the National Institutes of Health.
For decades, it has been unclear exactly how the presenilin mutations cause Alzheimers disease. Presenilin is a component of an important enzyme, gamma secretase, which cuts up amyloid precursor protein into two protein fragments, Abeta40 and Abeta42. Abeta42 is found in plaques, the abnormal accumulations of protein in the brain which are a hallmark of Alzheimers. Numerous studies suggested that presenilin-1 mutations increased activity of gamma-secretase. Investigators have developed drugs that block gamma-secretase, but they have so far failed in clinical trials to halt the disease.
Alzheimer’s Disease: The Genomic Era
AD can be divided into two subtypes: familial AD and sporadic AD . FAD is usually early-onset AD , generally occurring in individuals < 65 years, whereas SAD is mainly late-onset AD , affecting people > 65 years . FAD accounts for less than 5% of disease incidence and is caused by one or more autosomal dominant mutations in the gene that encodes for APP, or in the genes that encode for presenilin-1 or -2 , which are components of the -secretase enzyme that is involved in the cleavage of APP . Although the role of genetics in SAD is less well defined than that in FAD, a heritable component has been recognized, exemplified by the larger monozygotic twin concordance rate for the disease than the dizygotic twin concordance rate .
Figure 8.2. Alzheimer’s disease subtypes and their genetics.
Familial AD accounts for & lt 5% of AD cases and is caused by autosomal dominant mutations in the amyloid precursor protein , PSEN1, or PSEN2 genes. Sporadic AD accounts for & gt 95% of AD cases and many common genetic variants have been associated with this type of disease. SNP, single nucleotide polymorphism.
Joseph B. Martin, in, 1992
Read Also: Dementia Awareness Color
Genetic Factors In Alzheimer’s Disease And Cerebral Amyloid Angiopathy
Familial Alzheimer’s disease is associated with defined mutations in the APP and presenilin genes. Polymorphisms in apolipoprotein E are related to the development of cerebral amyloid angiopathy and sporadic Alzheimer’s disease. ApoE is an amyloid scavenging molecule regulating extracellular concentrations of A through ApoE receptor internalization via the endosomal/lysosomal pathway. ApoEA complexes are transported from the interstitial fluid in the brain into blood through specific ApoE receptors present at the bloodbrain barrier. However, co-localization of ApoE with A in brain parenchyma and in artery walls suggests that ApoEA complexes are also transported with interstitial fluid along perivascular drainage pathways. In this way, ApoE may act as a chaperone molecule for A. ApoE exists in three isoforms the 4 isoform is the most important genetic risk factor for the development of sporadic Alzheimer’s disease and has a strong link with cerebral amyloid angiopathy.
S.B. Prusiner, in, 2018
How Much Do These Risk Factors Change The Risk
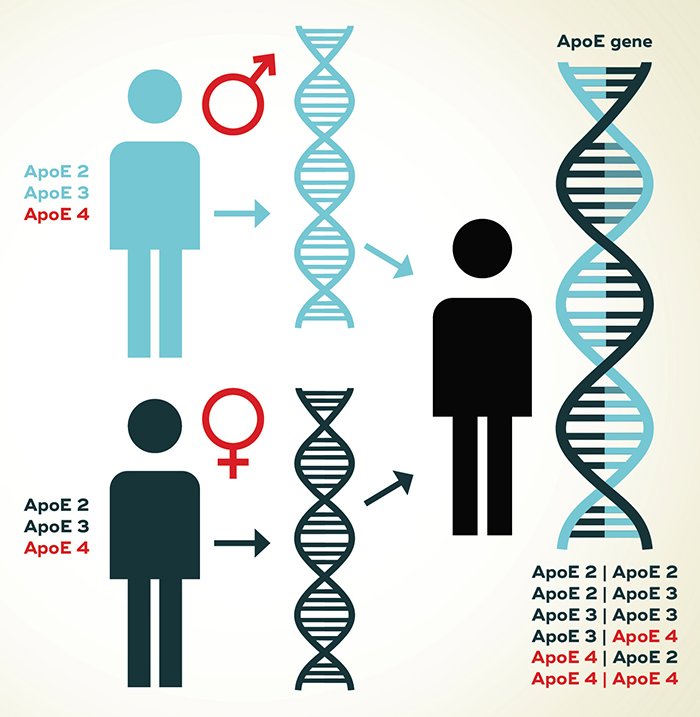
Lets take the example of APOE. APOE is a gene that comes in a number of different variants , APOE e2, APOE e3 and APOE e4, and you receive one allele from each parent. If you have one APOE e4 allele, you have a higher risk of developing Alzheimers disease. If you have two APOE e4 alleles your risk is even higher, but its still not a certainty, this is reflected in their position in the diagram above. It might help to understand this in simple numbers.
According to one study3 if we take 100 men aged 85, we would expect 10 to develop AD during their lifetime. The figure for women is slightly higher, 14 out of 100. However, if we take a group of 100 men who have one APOE e4 allele and one APOE e3 allele we would expect 23 of them to develop AD. Again its slightly higher for women, at 30 out of 100. The numbers are even higher for people who have two copies of the APOE e4 allele , but having these genes still doesnt completely determine whether you develop Alzheimers disease or not. Having two APOE e4 alleles isnt all that common, between 10% and 30% of the population may have this particular genotype4,5. The other variants of APOE dont increase your risk, in fact studies suggest the APOE e2 allele is slightly protective.
Also Check: What Is Senility
Study Design And Participants
We conducted a retrospective cohort study of families with histories suggestive of ADAD, which were referred to the Dementia Research Centre at University College London’s Institute of Neurology from clinical and research centers across the United Kingdom and Ireland between July 1, 1987, and September 2, 2019. We used clinical and genetic data from these families . Inclusion criteria for the study were a family history suggestive of ADAD and known age at symptom onset. Exclusion criteria were a neurodegenerative condition other than ADAD, unknown age at symptom onset, unknown year of birth, and no information on last year of contact with the center.
PSEN1 and APP: characteristics of our cohort
Genetic Risk Scores For Alzheimer Disease May Have Implications For Parkinson Disease
The findings from the study indicate that higher genetic risk score for Alzheimer disease are associated with cognitive declines in patients with Parkinson disease , while higher genetic risk scores for PD signify an increased risk of PD, albeit with slower clinical progression after diagnosis.
Researchers from a new study have found that genetic risk scores and cerebral spinal fluid biomarkers of Alzheimer disease have implications for outcomes of patients with Parkinson disease .
The findings from the study, , indicate that higher GRS for AD are associated with cognitive declines in patients with PD, while higher genetic risk scores for PD signify an increased risk of PD, albeit with slower clinical progression after diagnosis.
Coexisting Alzheimers disease pathology is observed in approximately 30% of autopsy-confirmed Parkinsons disease patients, and mixed AD pathology is associated with more -synuclein -positive Lewy body disease pathology and faster cognitive decline, explained the researchers. Accordingly, multiple studies have focused on predicting AD pathology using cerebrospinal fluid biomarkers, such as phosphorylated-tau, total-tau, and -amyloid , or apolipoprotein E 4 allele in PD However, the contribution of CSF biomarkers on clinical progression in PD patients is conflicting.
The researchers relied on data from 400 patients with de novo PD and 195 healthy controls from the Parkinsons Progression Marker Initiative database.
Reference
Recommended Reading: Dementia Ribbon Color
The Genetics Of Alzheimer’s Disease
In order to understand the genetics of Alzheimer’s disease , it is first necessary to understand the basics of genetics. Most cells of the body have 46 chromosomes, grouped into 23 pairs. One member of each chromosome pair is inherited from each parent. Each chromosome is composed of deoxyribonucleic acid , the genetic material that provides instructions for how the body grows and functions . Genes are segments of DNA found on each of our chromosomes. Each gene provides the instructions for a specific protein with a specific function. Certain variations within the genes may lead to differences in individual traits . Other variations in gene sequence may be disease-causing. When a gene variant causes disease, it is often referred to as a mutation.
Many studies have been carried out to identify gene variants associated with an increased risk for developing AD. Two different types of variants have been identified: disease-causing mutations in individuals with early-onset familial AD and susceptibility variants in individuals with late-onset AD. Those with early-onset familial AD are likely to develop AD before the age of 65 and are likely to have family members who also have early-onset AD. These individuals account for less than 5% of all AD cases. Those with late-onset AD account for the vast majority of AD cases. Those with late-onset AD may or may not have similarly affected family members.
Npcs Have The Capacity To Make Electrically
We wished to establish that our NPCs were capable of making mature neurons that had electrical signaling properties similar to primary neurons. Thus, we recorded electrophysiogical properties from 375 cells that had been neuronally differentiated 35 to 55 days from control iPSC line 7889O and 87 cells from PS1 iPSC line 8446B. Cells differentiated into mature neurons as shown by the presence of fast inward currents due to the Na+ channels opening after depolarization with a series of 10 mV voltage steps from â90 mV and their ability to produce action potentials upon stimulation with a depolarizing current, as shown for 7889O and 8446B .
Action potentials and normal spontaneous Ca2+ transients are present in neurons differentiated from control line 7889O and 8446.Aβ42/Aβ40 Ratio is Elevated in PSEN1 Cells.
Also Check: Alzheimer Awareness Ribbon
Genes And Vascular Dementia
Vascular dementia is caused when blood flow to the brain is reduced, damaging nerve cells. This can happen as a result of a stroke or damage to blood vessels deep in the brain. The majority of cases of vascular dementia are not caused by faulty genes.
We may carry genes that affect our risk of stroke, heart disease or other diseases that may contribute to vascular dementia. However, lifestyle factors such as smoking, lack of exercise, obesity, drinking alcohol over the recommended limits, and an unbalanced diet can also affect our risk.
There are rare genetic disorders that can cause vascular dementia by damaging blood vessels in the brain. One is called CADASIL and can be passed down through families. CADASIL only affects around 1,000 people in the UK.
Gene Expression Profiling Comparison For Control Versus Ps1 Npcs
Having established that we could successfully model the molecular pathology of FAD in our PSEN1 NPCs, we further defined their gene expression via global gene expression studies. The purpose of this approach was threefold: 1) Further characterization of PSEN1 NPCs, including additional analysis of their neurogenic potential 2) Identification of molecules that might have a developmental and/or amyloid-independent role in the pathogenesis of FAD 3) Attempt to find molecules that might also be misregulated in late-onset AD. Thus, we performed GEP experiments on both undifferentiated and 14-day neuronally differentiated control and PSEN1 NPCs using the Illumina HumanHT-12-14 BeadChip platform. The results were analyzed using Genome Studio software, and genes were considered differentially expressed if DiffScores were greater than 13 or less than -13 .
Gene Expression Profiling of Control vs. PS1 NPCs/Early Neurons.
You May Like: Can I Buy Jelly Drops For Dementia
How Can I Reduce My Risk Of Dementia
For the vast majority of people, our genes are only one factor affecting our risk of dementia. There are many other factors involved, such as age and lifestyle. While we cannot change our age or genes, research has found that up to a third of all cases of dementia could be avoided through lifestyle changes.
There are simple things we can do that may help lower our risk:
- Do not smoke.
- Drink fewer than 14 units of alcohol per week.
- Control high blood pressure.
- Keep cholesterol at a healthy level.
- Keep active and exercise regularly.
- Maintain a healthy weight.
- Eat a healthy balanced diet.
You can find more detailed information about how you can reduce your risk of dementia here.
Qpcr Validation Of Psen1 Npc Differentially
We postulated that most positively and negatively regulated genes in our GEP experiments would be the most potentially relevant to AD. There were 34 upregulated genes and 6 downregulated genes that were attractive candidates for further validation. We further pruned the upregulated list to 23 genes by eliminating 11 genes that were not expressed in control NPCs at statistically detectable levels. This reduced the potential of a rare minority cell type skewing the data.
We analyzed the remaining 29 genes in two independent differentiation experiments utilizing our 8 core cell lines in biological triplicates for each experiment . The average expression of each of the four controls lines was compared against the average expression of the four PSEN1 lines . Ten upregulated genes and four downregulated genes were validated by this method . Four additional genes showed an upregulation trend in PSEN1 NPCs . We then looked at the expression of three targets in more detail: NLRP2, ASB9 and NDP.
Don’t Miss: Alzheimer’s Awareness Color
Information About Genetic Testing
Having a test to look for a faulty gene that causes dementia is only appropriate for a very small number of people. This is because inherited dementia is rare.
If you are worried that you have a strong history family of early-onset Alzheimers disease or frontotemporal dementia, you can speak to your doctor about this.
Not all gene mutations that cause dementia have been identified, meaning that some families may have many affected members, but no mutation can be found. Therefore, a negative test result cannot always rule out a genetic cause of a disease.
If a test is appropriate, your doctor should be able to refer you to a genetic counsellor or specialist. This could be a cognitive neurologist or memory clinic psychiatrist. They will discuss with you the pros and cons of taking a test and what will be involved. They will also tell you where the results will be kept, who they will be shared with, and what the next steps would be. For people found to have a genetic mutation that causes dementia, these discussions will also cover the options available if you are considering starting a family.
To find out more about genetic testing and what support is available you can visit www.raredementiasupport.org or call 020 3325 0828. Leave a message and you will be referred to the most appropriate team member.
Definition Of Associated Loci
A region of ±500kb was defined around each variant with a stage I P value below 1×105. These regions were then merged to define nonoverlapping regions. The region corresponding to the APOE locus was excluded. We then used the PLINK clumping procedure to define independent hits in each region. An iterative clumping procedure was applied to all variants with a stage I P value below 1×105, starting with the variant with the lowest P value . Variants with a stage I P value below 1×105, located within 500kb of this index variant and in LD with the index variant were assigned to the index variants clump. The clumping procedure was then applied until all the variants had been clumped. LD in the EADB-TOPMed dataset was computed using high-quality imputed genotypes.
Read Also: Difference Between Dementia And Senility
Genetic Testing For Alzheimers Disease
A blood test can tell which APOE gene you have, but the results canât predict whether youâll get Alzheimerâs. Doctors use these tests mostly for research purposes. The test can tell them who has certain risk factors so they can watch for brain changes in case the disease develops.
Doctors donât typically recommend genetic testing for late-onset Alzheimerâs because the results can be confusing and cause emotional distress. If youâre showing symptoms or have a family history, your doctor may recommend testing to help diagnose early-onset Alzheimerâs. Doctors can usually diagnose Alzheimerâs without a genetic test.
Cause #: Past Head Trauma
The Alzheimers Association states that scientists have found a link between traumatic brain injury and a greater risk of dementia. After a traumatic injury, your brain creates large amounts of beta amyloid. This is the same protein that develops into the damaging plaques that are a hallmark of Alzheimers.
Theres one difference: After a traumatic brain injury, beta amyloid, although present, doesnt clump into plaques. However, the damage may increase the risk of them doing so later in life.
Don’t Miss: Did Ronald Reagan Have Alzheimer In Office
Can Vascular Dementia Be Inherited
In most cases, vascular dementia itself is not inherited. However, the underlying health issues that sometimes contribute to this condition, such as high blood pressure or diabetes, may be passed on from one generation to another.
Other than in a few, very rare cases, parents cannot pass on vascular dementia to their children. However, a parent may pass certain genes that increase the risk of developing vascular dementia.
The sort of genes that increase the risk of vascular dementia are often the same ones that increase the risk of high blood pressure, diabetes, heart disease and stroke.
For this reason, having a healthy lifestyle, such as eating well and staying physically active, are probably more important for preventing vascular dementia than they are in Alzheimer’s disease.