Are There Harms Or Limitations Of Genetic Testing
Currently no cure or effective preventive therapy is available if a gene mutation is found. A positive test result for a known early-onset familial AD family gene mutation can result in:
- Adverse psychological reaction, family issues/distress
- Confidentiality issues
Insurance discrimination
- Historically, genetic testing in an asymptomatic individual may have affected their ability to obtain life, disability, critical illness, long-term care and/or extended health insurance. However, in 2017 Canada passed the Genetic Information Non-Discrimination Act that protects individuals from the use of a genetic test result to prevent them from obtaining insurance.
A negative test result for a known familial early-onset familial AD gene mutation can result in survivor guilt.
When an individual with no known familial gene mutation has genetic testing, a negative result is not a definitive answer.
For review articles on AD see Alonso Vilatela ME et al., Genetics of Alzheimers disease. Arch Med Res. 2012 43: 622-31 and Goldman JS et al., Genetic counseling and testing for Alzheimer disease: Joint practice guidelines of the American College of Medical Genetics and the National Society of Genetic Counselors. Genet Med 2011 13: 597-605.
Other AD resources:
Patients Carrying One Or Two Apoe4 Alleles
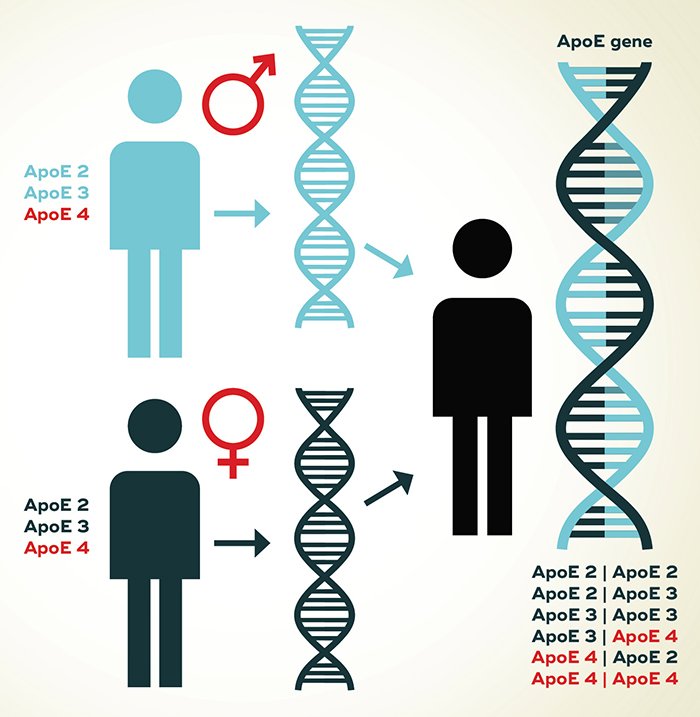
The by far largest single genetic risk factor for LOAD is apolipoprotein E , which has been known since 1993. There are three common isoforms of APOE , which result from polymorphic variation in the gene. APOE4 is associated with higher risk of AD . However, APOE4 cannot be regarded as causal in the development of AD, since it is neither sufficient nor necessary to cause AD. This means, individuals carrying one or two of the APOE4 risk alleles will not certainly develop AD and individuals without APOE4 are not protected. However, its high importance can be explained by its relatively high frequency in the population combined with a relatively high effect strength, which is also illustrated in Fig. . Other known genetic variants either occur many times less frequently or have only minor effect on disease development.
The APOE4 allele has been described to promote amyloid deposition starting already in middle age. PET studies in cognitively healthy individuals in various age groups have shown that APOE4-positive individuals exhibit amyloid deposition significantly earlier than APOE4-negative individuals . Although APOE4 has been known as a risk factor for a very long time, there remains disagreement over the specific mechanisms by which the APOE4 allele increases the risk of AD and age-related cognitive decline.
Which Genes Increase Your Risk For Alzheimers
There are several genes that researchers believe have an impact on your risk of developing Alzheimers. Again, these genes do not determine whether you will actually get Alzheimers, they simply tilt the odds in one direction. The most well-known and well-studied of these risk genes is the APOE-e4 gene its estimated that 40 to 65% of people diagnosed with Alzheimers have this gene.
Everyone inherits one APOE gene from each of their parents, but there are three different alleles of the gene, including e2, e3, and e4. If you inherit one APOE-e4, you have a threefold increased risk for developing Alzheimersinherit two and it increases up to 8 to 10 times .
On the other hand, if you inherit the APOE-e2 version, your Alzheimers risk goes down. The most common APOE allelee3is believed to have no effect at all on the risk of Alzheimers. There are many other factors, including socioeconomic determinants of health, associated with the expression of the APOE gene, including race, says Dr. Noble: We know from many studies that e4 seems to have a higher risk of expressing Alzheimers in non-Hispanic white people, while it appears to have a lower impact on those who are non-Hispanic Black people, he explains.
Genes associated with inherited Alzheimers
How do genetics affect treatment?
Should I get tested for APOE-e4?
The bottom line
Don’t Miss: What Color Is The Alzheimer’s Ribbon
Sex Chromosome Dosage : An Engine Of Stability
The crosstalk that exists between X chromosomes and autologous genes is a relatively new paradigm that has emerged as a result of the biology of sex differences, and gives rise to the question of how SCD shapes the genome function. To explore this, human sex aneuploidies were analyzed from a genome-wide expression dataset by Raznahan et al. where they found a dosage sensitivity of the X-Y chromosome pair resulting in increased expression of genes that decrease X/Y chromosomal dosage . The most interesting finding was that X-linked genes were found to regulate co-expression of networks of autosomal genes that are SCD-sensitive and, in addition to these findings, suggest that the autosomal genes and their corresponding networks are crucial for cellular functions. This highlights the potential of SCD to affect the occurrence of disease.
The most common aneuploidy in AD is XO mosaicism . In respect to SCD and the XO status, Raznahan et al. have demonstrated up-regulation of the protein networks, noncoding RNA metabolism, suppression of the cell cycle, changes in regulation of DNA/chromatin organization, glycolysis, and response to stress . Changes in these collective networks through XO and supernumerary XXY, and XXYY syndromes may enhance the risk of AD .
Figure 2 X chromosome instability, Sex Chromosome Dosage, Topological changes of Chromosomes, and its possible role in AD.
Dementia And Down Syndrome
People with Down syndrome are born with an extra piece of DNA. This means they also have an extra copy of the APP gene. This leads to the build-up of amyloid plaques in the brain, which play a role in the development of Alzheimers disease. While not everyone with Down syndrome will go on to develop symptoms of Alzheimers, most people with the condition over the age of 40 will have amyloid build-up. It is estimated that about 50% of people with Down syndrome develop symptoms like memory loss, usually in their 50s and 60s.
To find out more about Down syndrome and dementia you can contact Downs Syndrome Association helpline on 0333 1212 300 or visit their website www.downs-syndrome.org.uk.
Read Also: What Color Represents Alzheimer’s
Genes Which May Influence Alzheimers Disease
Having a close relative with the Alzheimers disease is not evidence of a genetic link. People who are influenced by risk factor genes are only at a slightly increased risk in developing the disease than the average population.
The most important gene discovered to date is the Apolipoprotein E gene, which is found in chromosome 19. This gene occurs in three forms in humans: types 2, 3 and 4. Every person in the world carries two Apolipoprotein genes: they can be the same type , or a mixture of two types . What has been found is that people with at least one type 4 and especially those with two, such as 4,4, are at an increased risk of developing Alzheimers disease earlier in life than those with the other types of Apolipoprotein E. Nevertheless half of the people aged 85 who have 2 copies of apolipoprotein E 4 do not have symptoms of Alzheimers disease at that age.
People with type 2, especially 2,2, appear to be protected against developing Alzheimers disease, until much later in life. Researchers do not understand why this is so, and there is much research underway to find out why.
The type of Apolipoprotein does not mean definitely that Alzheimers disease will or will not occur. Indeed it is known that some people can reach 90 with type 4 and not develop dementia, whereas others with type 2 can develop dementia much earlier in life. What this means is that the type of Apolipoprotein a person has, is not enough on its own to cause Alzheimers disease.
The Genetics Of Alzheimer’s Disease
In order to understand the genetics of Alzheimer’s disease , it is first necessary to understand the basics of genetics. Most cells of the body have 46 chromosomes, grouped into 23 pairs. One member of each chromosome pair is inherited from each parent. Each chromosome is composed of deoxyribonucleic acid , the genetic material that provides instructions for how the body grows and functions . Genes are segments of DNA found on each of our chromosomes. Each gene provides the instructions for a specific protein with a specific function. Certain variations within the genes may lead to differences in individual traits . Other variations in gene sequence may be disease-causing. When a gene variant causes disease, it is often referred to as a mutation.
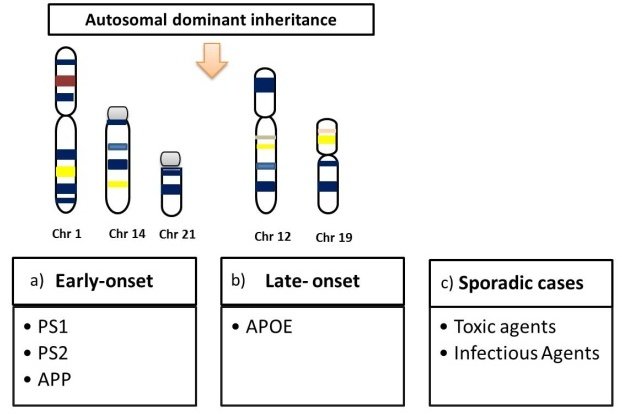
Many studies have been carried out to identify gene variants associated with an increased risk for developing AD. Two different types of variants have been identified: disease-causing mutations in individuals with early-onset familial AD and susceptibility variants in individuals with late-onset AD. Those with early-onset familial AD are likely to develop AD before the age of 65 and are likely to have family members who also have early-onset AD. These individuals account for less than 5% of all AD cases. Those with late-onset AD account for the vast majority of AD cases. Those with late-onset AD may or may not have similarly affected family members.
Don’t Miss: Colors For Alzheimer’s Awareness
Alzheimers Risk Factors You Can Change
Even if you have no family history or genetic risk of Alzheimers, other health issues, such as brain injury and cardiovascular disease, can increase your chances of having dementia.
For instance, your odds of having the disease increase if you suffer from:
Are Genetic Forms Of Alzheimers Disease Still Being Researched
Research into genetic forms of Alzheimers disease and other inherited dementias is still very active in many places around the world.
Many people from Alzheimer’s affected families have generously volunteered to take part in research studies, and this has given us great insight into the causes of different dementias.
In 2008, an international consortium named Dominantly Inherited Alzheimer Network or DIAN was set up to study genetic Alzheimer’s disease to try and understand more about its causes and the earliest signs.
Recommended Reading: Alzheimer’s Association Colors
General Definition And Overview
Alzheimers disease is a type of dementia that gradually destroys brain cells, affecting a persons memory and their ability to learn, make judgments, communicate and carry out basic daily activities. Alzheimers disease is characterized by a gradual decline that generally progresses through three stages: early, middle and late stage disease. These three stages are distinguished by their general features, which tend to progress gradually throughout the course of the disease. Alzheimers disease is not inevitable in people with Down syndrome. While all people with Down syndrome are at risk, many adults with Down syndrome will not manifest the changes of Alzheimers disease in their lifetime. Although risk increases with each decade of life, at no point does it come close to reaching 100%. This is why it is especially important to be careful and thoughtful about assigning this diagnosis before looking at all other possible causes for why changes are taking place with aging. Estimates show that Alzheimers disease affects about 30% of people with Down syndrome in their 50s. By their 60s, this number comes closer to 50%.
Exploring The Role Of Kdm6a
The authors of the new study suspected the KDM6A gene may help account for the differences observed in mice with one versus two X chromosomes.
When mice have two X chromosomes, most of the genes on the second X chromosome are inactivated. But KDM6A is one of a small group of genes that remains active on both chromosomes.
After reviewing a public dataset of gene expression studies, the researchers found that about 14 percent of people carry a particularly active variant of the KDM6A gene.
This variant of the gene wasnt associated with lower risk of developing Alzheimers disease, but it was linked to slower cognitive decline in people who have the disease.
When the researchers engineered male mice with Alzheimers disease to produce more KDM6A protein than usual, they found the mice performed much better than average on cognitive tests.
Read Also: Alzheimer’s Aphasia
The Genetics Of Alzheimers Disease
Robert C. Barber
1Department of Pharmacology and Neuroscience, University of North Texas Health Science Center, 3500 Camp Bowie Boulevard, Fort Worth, TX 76107, USA
Abstract
Alzheimers disease is a progressive, neurodegenerative disease that represents a growing global health crisis. Two major forms of the disease exist: early onset and late onset . Early onset Alzheimers is rare, accounting for less than 5% of disease burden. It is inherited in Mendelian dominant fashion and is caused by mutations in three genes . Late onset Alzheimers is common among individuals over 65 years of age. Heritability of this form of the disease is high , but the etiology is driven by a combination of genetic and environmental factors. A large number of genes have been implicated in the development of late onset Alzheimers. Examples that have been confirmed by multiple studies include ABCA7, APOE, BIN1, CD2AP, CD33, CLU, CR1, EPHA1, MS4A4A/MS4A4E/MS4A6E, PICALM, and SORL1. Despite tremendous progress over the past three decades, roughly half of the heritability for the late onset of the disease remains unidentified. Finding the remaining genetic factors that contribute to the development of late onset Alzheimers disease holds the potential to provide novel targets for treatment and prevention, leading to the development of effective strategies to combat this devastating disease.
1. Introduction
2. Familial Alzheimers Disease
2.1. Amyloid Precursor Protein
2.2. Presenilins 1 and 2
Common Genetic Factors In Both Depression And Alzheimers Disease
Complete the form below and we will email you a PDF version of“Common Genetic Factors in Both Depression and Alzheimers Disease”
First Name*Would you like to receive further email communication from Technology Networks?Technology Networks Ltd. needs the contact information you provide to us to contact you about our products and services. You may unsubscribe from these communications at any time. For information on how to unsubscribe, as well as our privacy practices and commitment to protecting your privacy, check out our Privacy Policy
Epidemiological data have long linked depression with Alzheimers disease , a neurodegenerative disease characterized by progressive dementia that affects nearly 6 million Americans.
Now, a new study identifies common genetic factors in both depression and AD. Importantly, the researchers found that depression played a causal role in AD development, and those with worse depression experienced a faster decline in memory. The study appears in Biological Psychiatry, published by Elsevier.
Co-senior author Aliza Wingo, MD, of Emory University School of Medicine, Atlanta, USA, said of the work, It raises the possibility that there are genes that contribute to both illnesses. While the shared genetic basis is small, the findings suggest a potential causal role of depression on dementia.
Also Check: Alzheimer Ribbon Color
People Affected By Dementia Are Often Concerned About Whether The Condition Can Be Passed Along In Families
Here we discuss the role of heredity in Alzheimers disease and other forms of dementia.
Dementia is the term used to describe the symptoms of a large group of illnesses which cause a progressive decline in a persons functioning. It is a broad term to describe a loss of memory, intellect, rationality, social skills and what would be considered normal emotional reactions. Alzheimers disease is the most common form of dementia, accounting for 50% to 70% of all cases of dementia. It occurs relatively frequently in older people, regardless of family history. For females aged 65 to 69 years dementia affects 1 person in 80 compared to 1 person in 60 for males. For both males and females aged 85 and over the rate is approximately 1 person in 4.
Areas Of Future Research
According to the National Institute on Aging, here are some questions to answer in future research into the association between Alzheimers and genetics:
- How genetics interact with lifestyle factors to affect Alzheimers risk
- How the disease process begins
- Why Alzheimers is the most common cause of dementia symptoms
- What the difference in Alzheimers between various racial groups and sexes
- How to prevent Alzheimers
- What treatments are possible and effective
- Who is most at risk for Alzheimers, and why
Don’t Miss: Can Prevagen Help Dementia
The Genetics Of Alzheimer’s Disease And Frontotemporal Disorders
The Genetics of Alzheimer’s Disease
Most cases of Alzheimers disease-type dementia result from a mix of genetic, environmental and lifestyle factors. Overall, 65 year olds have a 10 percent risk of developing symptoms. In other words, 1 in 10 people over age 65 will develop it, and those who reach 85 or older, the rate rises to 1 in 3. That level of risk varies in subtle ways across individuals. About 20 gene variants are known to confer low levels of increased risk for late-onset Alzheimers disease, while others endow resilience.
Some cases of Alzheimer’s disease, and other neurodegenerative diseases such as frontotemporal dementia, are caused by a genetic mutation, or permanent change in one or more specific genes. About 3% of Alzheimer cases are familial, or caused by single mutations inherited from a parent. Familial Alzheimer’s disease usually onsets before age 65. Read on to learn about the genetic differences between early- and late-onset Alzheimer’s disease, and frontotemporal degeneration .
Identifying genetic variants may help researchers find the most effective ways to treat or prevent diseases such as Alzheimer’s or frontotemporal degeneration in an individual. This approach, called precision medicine, takes into account individual variability in genes, environment, and lifestyle for each person.
Late-Onset Alzheimers Disease Genetics
* Adapted from the National Institute on Aging: Alzheimer’s Education and Referral Center
More information sources:
Can Genes Cause Dementia
Around 1 in 4 people aged 55 years and over has a close birth relative with dementia. Find out what part genes play in dementia and how genetics can affect the risk of developing the condition.
It is well known that children can take after their parents for example, in the way they look. This is partly because many of the key characteristics of a person are passed down from parents to children in their genes.
Also Check: Alzheimer’s Awareness Ribbon