Trial Begins Of Nasal Vaccine For Alzheimer’s Disease
HealthDay Reporter
WEDNESDAY, Nov. 17, 2021 — The first human clinical trial of a nasal vaccine to slow the progression of Alzheimer’s disease is set to begin after nearly 20 years of research.
This is a “remarkable milestone,” according to Dr. Howard Weiner, co-director of the Ann Romney Center for Neurologic Diseases at Brigham and Women’s Hospital in Boston.
“Over the last two decades, we’ve amassed preclinical evidence suggesting the potential of this nasal vaccine for AD ,” Weiner said in a hospital news release. “If clinical trials in humans show that the vaccine is safe and effective, this could represent a nontoxic treatment for people with Alzheimer’s, and it could also be given early to help prevent Alzheimer’s in people at risk.”
The vaccine features an experimental agent called Protollin that stimulates the immune system. It’s designed to prompt white blood cells in the lymph nodes on the sides and back of the neck to migrate to the brain and clear beta amyloid plaques, a hallmark of Alzheimer’s disease.
According to Dr. Tanuja Chitnis, principal investigator in the trial, “For 20 years, there has been growing evidence that the immune system plays a key role in eliminating beta amyloid. This vaccine harnesses a novel arm of the immune system to treat AD.” Chitnis is a professor of neurology at the hospital.
The trial is funded by I-Mab Biopharma and Jiangsu Nhwa Pharmaceutical, developers and makers of Protollin.
More information
Two Primary Doses + Two Boosters
- This is for people who are over 75, or a resident in a care home. These are the people most at risk from coronavirus. The first vaccines will have worked well for these people, but they become less effective over time. You have to wait three months after your last vaccine to have your third dose.
Your GP or hospital specialist will write to you inviting you to book fourth dose. You will need to bring the letter with you to your appointment. A clinician at the site will check to make sure that you’re eligible for a fourth dose.
If you think you’re eligible for a fourth dose but you do not have a suitable letter, contact your GP or hospital specialist.
If you are offered a fourth dose, you should take it as soon as you can. It will offer extra protection, including against new variants of coronavirus.
It is recommended that the Pfizer or Moderna vaccine is used for booster and fourth dose vaccines, no matter which vaccine brand you had for your first vaccines doses.
Full guidance from the JCVI about third doses for immunosuppressed people can be found on the government website.
Testing On A Mouse Model Of Ad
When the researchers immunized mice with AD-like traits with the cyclized vaccine, they observed a significant reduction in the amount of amyloid plaque present and neuron death, indicating that the vaccine halts the development of the disease, as well as the downstream impaired function.
This led the researchers to ask if memory and cognition were also improved in immunized mice. They employed a method called the Morris water maze, in which mice are placed into a pool of water with a hidden platform. Healthy mice quickly learn where the platform is and can navigate to it, even from different starting positions. Mice with features of AD, on the other hand, always struggle to find the platform, suggesting a memory deficit. When these same mice were immunized with the cyclized vaccine, they were able to quickly navigate to the platform, suggesting the vaccine had prevented cognitive decline and memory loss.
The researchers hope to take the therapeutic antibody and the vaccine through clinical trials.
Recommended Reading: Familial Dementia
Pivotal Stage Of Clinical Development
In the phase 2 trial of the AADvac1, 196 volunteers in their over 60 were recruited from eight countries across Europe. They were selected at random to receive several doses of the Alzheimers vaccine or a placebo over the course of two years.
It led to high levels of antibodies against tau in the treated group, says Novak. AADvac1 shows robust antibody response, excellent safety profile and highly significant impact on neuro-degeneration.
Larger trials are now being planned to replicate the results and further test the clinical efficacy. Benefits are expected include symptomatic relief and even the prevention of Alzheimers. Our trial successfully demonstrated the strengths of AADvac1, a tau vaccine on track to prevent and treat Alzheimers disease, says Michal Fresser, CEO of Axon Neuroscience. The results confirm the disease-modifying effect and support Axons progress toward a pivotal stage of clinical development. In view of the recent approval of amyloid-based therapy, our strong NfL end point results could serve as a surrogate in our forthcoming clinical development to achieve accelerated approval.
Axon currently boasts the single biggest team in the world dedicated exclusively to tau research, with more than 60 scientists and 15 senior scientists. Over the last two decades, it has published a large body of evidence demonstrating pathological tau is the main driver of Alzheimers disease.
SWNS writer Mark Waghorn contributed to this report.
A New Potential Treatment And Vaccine For Alzheimers Disease Has Been Developed By A Team Of Uk And German Scientists Using A Different Approach With Amyloid Beta Protein

According to the NHS, Alzheimers disease is the most common cause of dementia in the UK. Dementia is a group of symptoms affecting the gradual decline in brain function, affecting memory and other mental abilities.
In a collaboration between University of Leicester, the University Medical Center Göttingen and the medical research charity LifeArc, a protein-based vaccine and antibody-based treatment created was found to reduce Alzheimers symptoms in mouse models of the disease.
You May Like: What Color Represents Dementia
New Vaccine For Alzheimers Disease Shows Potential In Mice
A preclinical study has tested a new vaccine for Alzheimers disease. The researchers found that the vaccine, which targets the beta-amyloid protein, was safe and effective in mice.
Alzheimers disease is a progressive neurological condition and the most common cause of dementia.
According to the latest estimates from the Alzheimers Association, 1 in 10 people over the age of 65 years in the U.S. have Alzheimers disease, and experts expect the number of people with the condition to increase as the population ages. By 2050, projections show that 13.8 million people aged 65 years and over will have Alzheimers disease in the U.S.
While groups around the world are working to find an effective treatment for the condition, an alternative approach is to develop a vaccine. Although they are more commonly associated with infectious diseases, vaccines can also prime the body to defend itself against other, noninfectious molecules.
In Alzheimers disease, scientists believe that two processes drive the progression of the disease: the buildup of plaques comprising beta-amyloid proteins between neurons in the brain and tangled knots of the tau protein within neurons.
Since the general understanding is that beta-amyloid initiates the disease process, scientists have tried to develop a vaccine against it. The hope is that the immune system recognizes and destroys the beta-amyloid before it can aggravate the cell damage that the tau protein causes.
Effectiveness In Mouse Models Of Ad
The researchers then assessed the therapeutic effects of the cyclic designer protein and the TAP01_04 antibodies in two mouse models of AD.
Immunization with either the cyclic designer protein or the humanized TAP01_04 antibody reduced the levels of amyloid plaques in the brains of the AD mouse model. This shows that while both approaches do not target plaques directly, they can reduce the formation of these aggregates by targeting amyloid- monomers.
The researchers then examined the impact of these immunization approaches on glucose metabolism in the brain, which AD diminishes. Both immunization approaches were able to rescue this reduction in glucose metabolism in a mouse model of AD.
Health experts characterize AD by memory loss and the loss of neurons in specific brain regions, including those involved in memory.
The researchers found that both the TAPAS vaccine and the humanized TAP01_04 antibody effectively improved performance in a memory task in a mouse model of AD. At the same time, both approaches reduced the loss of neurons in the hippocampus, a brain region that plays a central role in the formation of memories.
These results suggest that both the TAPAS vaccine and the TAP01_04 antibody were effective in reducing brain markers associated with the progression of AD.
Don’t Miss: Alzheimers Awareness Ribbons
Vaccines And Prion Disease
Before I discuss all these claims, I have to answer the question: What are prions? Prion diseases are more properly referred to as transmissible spongiform encephalopathies and represent a rare form of progressive neurodegenerative disorders that can affect both humans and animals. The most famous prion diseases in animals are bovine spongiform encephalopathy , scrapie, and chronic wasting disease in humans, Creutzfeldt-Jakob Disease and kuru. These diseases have a long incubation period and produce in the brain characteristic spongiform changes associated with loss of neurons with no inflammatory response. The causative agents for these diseases are prions. According to the CDC:
The term prions refers to abnormal, pathogenic agents that are transmissible and are able to induce abnormal folding of specific normal cellular proteins called prion proteins that are found most abundantly in the brain. The functions of these normal prion proteins are still not completely understood. The abnormal folding of the prion proteins leads to brain damage and the characteristic signs and symptoms of the disease. Prion diseases are usually rapidly progressive and always fatal.
The BSE inquirys report calls for vaccines to be investigated as a possible route of transmission. But it concedes that this will be hampered by the fact that systematic records of the action taken in response to BSE in respect of individual medical products are lacking.
And:
And:
And:
What Did The Scientists Do
A research team at the University Medical Center Göttingen and University of Leicester collaborated with scientists at the contract research organisation, LifeArc, to develop a new treatment approach for the leading cause of dementia.
Alzheimers disease affects roughly two thirds of all people with dementia in the UK.
Using two types of mice with features of Alzheimers disease, the scientists looked at the effect of injecting an antibody and engineered vaccine they developed.
The treatments target a shortened form of the hallmark Alzheimers protein, amyloid, which other Alzheimers drugs in development have not targeted.
You May Like: Bobby Knight Health
Development Timeline Of Alzheimers Vaccine
On September 29, IMM announced that it had licensed MultiTEP, the universal vaccine platform technology it has been developing, to Nuravax.
As with the flu or coronavirus vaccines, the technology is intended to elicit an extremely high immune system response to an invader in this case, amyloid plaque and tau tangles. It is based on the widely adopted theory that pathological amyloid-beta and tau are major drivers of AD pathology.
Elderly immune systems are characterized by a deficiency of naïve T helper cells but are replete with memory T helper cells. Thus, Nuravax is designing the vaccine to stimulate these T helper cells to interact with B cells in order to produce a large number of antibodies. These antibodies would then prevent amyloid plaques and tau tangles from forming, delaying or halting the disease.
The Alzheimers vaccine design uses 12 protein segments from vaccines that have been administered to the general population during their lifespans, as well as previous infections. This is expected to generate titers of antibodies high enough to clear the blood-brain barrier.
So, why is an Alzheimers vaccine the right approach to this disease? Because the disease pathology begins much earlier than one’s twilight years, Agadjanyan said.
He went on to compare the role of prophylaxis to that of monoclonal antibody treatments.
Featured Jobs on BioSpace
A Vaccine For Alzheimer’s
Researchers May Be Getting Closer to Vaccine for Brain Disorder
July 19, 2006 — After the widely publicized failure of a potential vaccine for Alzheimer’s diseaseAlzheimer’s disease in 2002, new hope may be on the horizon.
According to research presented Wednesday at the 10th International Conference on Alzheimer’s Disease and Related Disorders in Madrid, Spain, researchers are already making great strides in developing a vaccine for this progressive brain disorder.
Affecting about 4.5 million Americans, Alzheimer’s disease gradually destroys a person’s memory and ability to learn, reason, make judgments, communicate, and carry out daily activities, according to the Alzheimer’s Association.
Alzheimer’s disease is a result of damage to nerve cells in the brain. Plaques of a protein called beta amyloid contribute to the damage and death of brain cells. The antibody therapy tested in this study targeted beta-amyloid protein and amyloid plaques.
The older trial was halted in 2002 when 6% of participants developed a dangerous brain inflammation called encephalitisencephalitis some also developed brain shrinkage. But two new approaches toward vaccines seem to avoid such problems, explains John C. Morris, MD, director of the Alzheimer’s Disease Research Center of Washington University in St. Louis.
Also Check: Bob Knight Alzheimer’s
Dantiamyloid Therapies And Caa
Recent clinical as well as experimental studies with A immunotherapies for AD have demonstrated that CAA has important implications for the future of antiamyloid therapies. Experimental studies with AD model mice indicated that A immunotherapies can induce CAA-related cerebral hemorrhage, although long-term A immunotherapies could decrease CAA.114,115,186189
In clinical trials with A42 immunization for AD , immunized patients showed significantly higher levels of CAA and cortical microhemorrhages and microvascular lesions compared with unimmunized AD controls, although the longest living had virtually complete absence of both plaques and CAA.116 A CAA-related macrohemorrhage was reported in an AD patient in the phase 2a AN1792 trial.190 The findings suggest that A immunization results in solubilization of plaque A42, which flows out of the brain via the perivascular pathway, causing an increase of CAA and CAA-related hemorrhages. Eventually, this flow also results in A clearance from the cerebral vasculature.
Hyun Yang, Dong Seok Kim, in, 2015
Covid Vaccines Confirmed To Cause Parkinsons Dementia Symptoms
Monday, July 26, 2021 by: Ethan Huff
Former National Institutes of Health contract scientist and Classen Immunotherapies proprietor J. Bart Classen has published a paper warning that there is strong potential for Wuhan coronavirus mRNA vaccines to trigger Parkinsons, dementia and a variety of other prion and chronic diseases.
Read Also: Andrea Mitchell Drunk
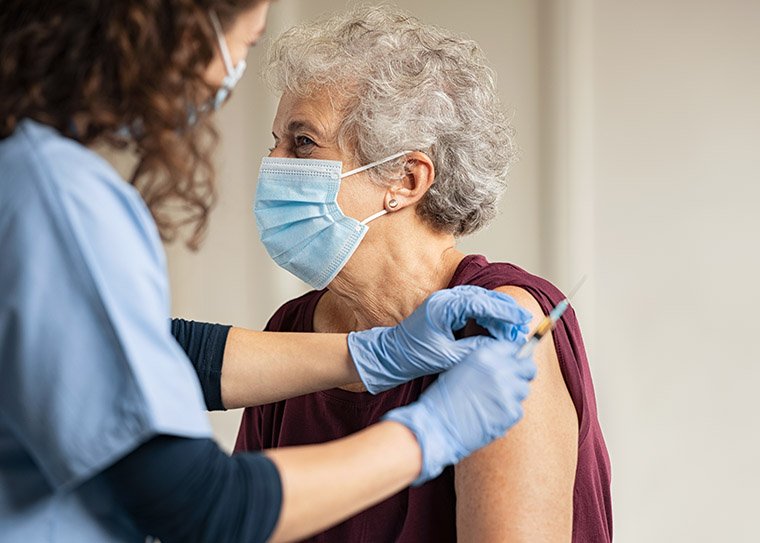
Boston Hospital Launches First Human Trial Of Nasal Vaccine For Alzheimers Disease
Researchers hope vaccine could offer a safe and effective way to prevent or slow down progression of AD.
Brigham and Womens Hospital in Boston is set to begin a clinical trial that will test the safety and efficacy of a new vaccine delivered nasally that has been developed to prevent and slow the progression of Alzheimers disease .
The launch is a remarkable milestone, said Howard L. Weiner, MD, codirector of the Ann Romney Center for Neurologic Diseases at the Brigham, in a press release. Over the last two decades, weve amassed preclinical evidence suggesting the potential of this nasal vaccine for AD. If clinical trials in humans show that the vaccine is safe and effective, this could represent a nontoxic treatment for people with Alzheimers, and it could also be given early to help prevent Alzheimers in people at risk, he said.
An estimated 55 million people live with dementia, which is a general term for impaired ability to remember, think or make decisions beyond what might be expected from the usual consequences of growing old, according to the World Health Organization . Alzheimers disease is the most common form of dementia, and may account for 60 to 70 percent of cases.
Phase 1 Trial Will Test The Safety And Tolerability Of Vaccine
The trial contains 16 participants between 60 and 85 years of age with early, symptomatic AD. All the participants are in good general health with no disease expected to interfere with the study and have had an amyloid-positive PET scan.
Each of the subjects will get two doses of the nasal vaccine, with the second dose being delivered a week after the first. This trials primary objective is to find out if the vaccine is safe and well tolerated, and the impact of nasal Protollin on participants immune response, including its effects on white blood cells, will also be measured.
This vaccine goes to show the immune system is important for so many things outside of infection, such as neurological diseases, cancers, heart disease, and the list goes on, says Purvi Parikh, MD, a clinical assistant professor at NYU Grossman School of Medicine and immunologist at NYU Langone, both in New York City. Dr. Parikh is not involved in the clinical trial.
Its exciting that we can train our immune system with vaccines to fight these disease in an effective way, she says.
Recommended Reading: Bob Knight Health
New Vaccine Formulation Shows Promise For Alzheimer’s Target
AC Immune CEO Andrea Pfeifer pictured above. Photo courtesy of AC Immune.
Switzerland-based AC Immune SAannounced positive interim results from its ongoing Phase Ib/IIa clinical trial of ACI-35.030 for Alzheimers disease. The vaccine showed a potent antigen-specific antibody response against phosphorylated tau in 100% of older patients with early Alzheimers.
AC Immune is working in collaboration withof Johnson & Johnson.
ACI-35.030 is a potent liposomal anti-pTau vaccine. It is engineered to elicit antibodies against phosphorylated pathological Tau protein, which is associated with Alzheimers disease. Two abnormal proteins in the brain are linked to Alzheimers, beta-amyloid and pTau. Generally, although not exclusively, beta-amyloid is seen earlier and pTau is seen later in the disease. ACI-35.030 is designed to decrease and help the clearance of aggregates of Tau.
The data demonstrated anti-tau IgG response preferentially targeted pTau in all patients, with 100% response after the first injection at both the lowest and second highest dosages. Very high anti-pTau IgG levels were observed after injection and the IgM response was also observed in all patients for both doses. The drug was safe and well tolerated with no clinically relevant safety issues. At this time, it plans to advance to the third and highest dosing levels, per study protocol.