Why Such ‘precision Medicine’ May Matter
As theyre based on a specific at-risk population, the teams findings lend support to a treatment approach called precision medicine, which has grown increasingly popular in Alzheimers research. It veers from a one-size-fits-all model and considers individual differences in environment, lifestyle and genetics in drug development and treatment decisions.
The traditional drug development approach for Alzheimers disease has been focusing on one protein, one gene or one cellular pathway, Huang says. The assumption for many years has been that we may find a magic bullet that will fit every Alzheimer’s disease patient.
Now, experts increasingly say the answer to ending Alzheimers probably doesnt lie in a single drug or therapy. Tackling the disease will likely require specific types of treatments, perhaps multiple therapies, including some that may target an individuals unique genetic and disease characteristics much like cancer treatments that are available today, Jean Yuan, M.D., a program director in the NIAs Division of Neuroscience, said in a statement.
A major reason: The disease cant be pinned to one cause, at least in most people. Experts say it’s likely due to a combination of age-related changes in the brain, along with genetic, environmental and lifestyle factors.
The Maddening Saga Of How An Alzheimers Cabal Thwarted Progress Toward A Cure For Decades
The findings raise hopes that aiming not so narrowly at beta-amyloid, and instead at the full cascade of molecular changes, might be more effective. But this hypothesis-free approach can also be a bit unsettling. Sure, it looks like the drug might work for Alzheimers disease. But why would it work? No one knows.
The mechanism of the drug is well-known, but what the authors havent addressed is how that mechanism is related to what they think might happen if they were to give this drug to Alzheimers patients, said Shilpa Kadam, a neurologist at Johns Hopkins University who studies developmental disabilities. She has been closely following a trend of physicians prescribing bumetanide off-label to people with autism, epilepsy, and other brain disorders. But in these instances of drug repurposing, the mechanism for why the drug might provide a benefit is well-understood. Bumetanide reduces water retention by blocking proteins that shuttle salts across cell membranes. Those proteins are also found in neurons, and if those neurons are overexcited as occurs in conditions like epilepsy blocking them can restore balance.
However, bumetanide can also be tricky to use because it can leave patients dehydrated and with their electrolytes off-kilter, said Jeffrey Cummings, director of the Chambers-Grundy Center for Transformative Neuroscience at the University of Nevada Las Vegas, who was not involved in the study.
Can Dementia Be Reversed
For decades, conventional medicine has offered little regarding the treatment of patients suffering from dementia or cognitive decline. Alzheimers disease is now the third-leading cause of death in North America, after heart disease and cancer. But treatment is possible.
It is estimated that dementia will afflict approximately 50 percent of the next generation of senior citizens . Most seniors today feel utterly helpless as they witness the deterioration of their cognitive abilities. Some even accept this as a normal part of aging. But dementia is not a normal part of aging and should not be accepted as such.
And while weve all met cancer survivors, no one has ever met an Alzheimers survivoruntil now.
The ReCODE program was developed over the past decade by Dr. Dale Bredesen, the director of research for neurodegenerative disease at UCLA and the author of The End of Alzheimers: The First Program to Prevent and Reverse Cognitive Decline.
This treatment program is based on addressing the several dozen mechanisms responsible for the expression of Alzheimers disease and is completely in alignment with the principles of integrative and functional medicine.
The ReCODE program is a full-spectrum, functional medicine approach for addressing all the physiological dysfunctions and imbalances that ultimately present as Alzheimers in the patient.
Also Check: How Fast Does Ftd Progress
Type : Inflammatory Or Hot
These are patients with mainly inflammatory symptoms. They have a brain that is figuratively on fire and balance cannot be restored until the inflammation has been brought under control. There have been many similarities observed between adults with this type of Alzheimers and children on the autism spectrumwith some overlap of successful treatment modalities for both.
The End Of Alzheimers Presents A Revolutionary Model To Halt Cognitive Decline

Everyone knows someone who has survived cancer, but no one knows anyone who has survived Alzheimers Disease. Until Now.
In his book The End Of Alzheimers, Dr. Bredesen unveils his groundbreaking protocol to prevent and reverse Alzheimers disease. The Bredesen Protocol, known as ReCODE, fundamentally changes how we understand cognitive decline.
Relying on 40 years of work and research in medicine, Dr. Bredesen reveals that Alzheimers disease is not one condition, as it is currently treated, but three distinct conditions each impacting the brain differently. The End of Alzheimers outlines 36 metabolic factors that can trigger downsizing in the brain. The protocol shows us how to rebalance these factors using lifestyle modifications like taking B12, eliminating gluten, or improving oral hygiene.
The results? Two years after its release, The End Of Alzheimers, ReCODE, and the Bredesen Protocol continue to change lives, as evidenced by hundreds of testimonials.
David Perlmutter, MD
Dr. Jeffrey Bland
Founder of the Institute for Functional Medicine, the Cleveland Clinic
Sara Gottfried, MD
Leroy Hood, MD, PhD
Awarded the National Medal of Science, presented by President Barack Obama in 2011, and founder of Institute for Systems Biology
ReCODE Participant N.K.
You May Like: Smelling Farts Dementia
How Is Aducanumab Administered
To take the drug, you need an intravenous infusion every four weeks for as long as you live. This means going to a hospital or a clinic where an intravenous tube is inserted, and the monoclonal antibody is slowly infused over several hours. The other monoclonal antibody, Gantenerumab, can be administered below the skin, and one may be able to administer the drug at home however, this technique does require some skill and supplies.
Evaluating The Drug Candidate In Mice
Over the last few decades, Dr. Pamela Maher and colleagues at the Salk Institute have studied a chemical called fisetin, found in fruits and vegetables, which can improve memory. The team developed a version of fisetin called CMS121, which they found to be effective in slowing the loss of brain cells.
In the current study, Maher and the team evaluated the effect of CMS121 on a type of mouse that ages very quickly and develops a disease similar to Alzheimers in humans.
The scientists gave one group of mice daily doses of CMS121. They started this when the mice were 9 months old the equivalent of late middle age in humans and had already begun showing symptoms of declining memory and learning.
They chose this timing of treatment to mirror the time when an individual would typically visit a doctor to receive treatment for cognitive issues.
The other two groups consisted of healthy untreated mice or untreated mice with the disease. After 3 months, when the mice were 12 months old, the team tested their memory and behavior and analyzed genetic and molecular indicators in their brains. They compared the results of the CMS121 group with those of the untreated mice.
Read Also: Senile Dementia Treatments
Promising New Drug Candidate May Reverse Alzheimers
Researchers at the Salk Institute have discovered that a drug candidate that scientists have previously shown to slow aging in brain cells has successfully reversed memory loss in a mouse model of inherited Alzheimers disease.
The new findings appear in the journal Redox Biology.
Alzheimers is a progressive brain disease that destroys brain cells, leading to memory loss and impairments in thinking and behavior. These symptoms are severe enough to restrict an individuals daily life and activities.
According to the National Institute on Aging , Alzheimers disease, the most common form of dementia, is the sixth leading cause of death in the United States. Additionally, they note that studies suggest that more than 5.5 million people in the U.S. may have dementia due to Alzheimers.
Old age is the biggest risk factor, with symptoms usually first appearing in adults in their mid-60s. However, it remains unclear what occurs in the brain at the molecular level with aging that contributes to Alzheimers disease.
Metabolic And Toxic Causes
Several endocrinal disorders and vitamin deficiencies can masquerade as dementia and need to be investigated, especially in young and rapidly progressive dementias. Several toxins can cause RPD. Exposure to heavy metals, such as arsenic, mercury, aluminum, lithium, or lead, can lead to cognitive decline, particularly after acute exposure. Most cases of acute exposures result in florid encephalopathies that progress over hours to days and thus would not be confused with rapidly progressive dementias, which progress over weeks to months. Manganese toxicity, found usually in miners, can present with significant Parkinsonism. Bismuth is a metal used to treat gastrointestinal disorders, principally peptic ulcer disease and diarrhea. Bismuth intoxication, typically caused by overdosing on bismuth-containing products, such as Pepto-Bismol, can cause a disorder mimicking Creutzfeldt-Jakob disease . Patients initially manifest with apathy, mild ataxia, and headaches, which progress to myoclonus, dysarthria, severe confusion, hallucinations , seizures, and, in severe cases, even death. Blood levels of bismuth, greater than 50 mg/L, are considered in the toxic range. The condition usually is reversible however, extremely prolonged use can result in permanent tremors.
You May Like: Alzheimer Awareness Ribbon
Stem Cells And Dementia
Stem cells are “building block” cells. They can develop into many different cell types, including brain or nerve cells.
Scientists have taken skin cells from people with certain types of dementia, such as Alzheimer’s disease, and “reprogrammed” them into stem cells in the lab. They’ve then triggered these stem cells to become brain cells.
These brain cells can also be used to test potential treatments at a very early stage.
Can Alzheimer’s Be Cured
P. Murali Doraiswamy discusses recent breakthroughs in diagnosing Alzheimer’s disease and what everyone can do to postpone the onset of memory loss.
Are you a scientist? Have you recently read a peer-reviewed paper that you want to write about? Then contact Mind Matters editor Jonah Lehrer, the science writer behind the blog The Frontal Cortex and the book Proust Was a Neuroscientist. His latest book isHow We Decide.
Read Also: Purple Ribbon Alzheimers
Common Medicines Have Reversed Alzheimers In Mice
A study has managed to reverse the symptoms of Alzheimers disease in mice by administering drugs currently used to treat hypertension and inflammation in humans.
In this study, scientists at IRB Barcelona led by Dr. Aloy have characterized three stages of Alzheimers disease, namely initial, intermediate, and advanced.
For each of these stages, they have analyzed the behavior of the animals, studied the effects on the brain , and performed a molecular analysis to measure gene expression and protein levels.
The approach adopted has allowed them to describe the development of the disease at a level of detail hitherto unknown and also compare it with healthy aging.
What we have observed is that, although Alzheimers disease shares some features of accelerated ageing, it is also affected by totally different ageing processes, says Dr. Aloy. This disease is caused by the abnormal accumulation of certain proteins, and we have seen that, in some cases, this is not caused by overproduction but by an error in their removal, he adds.
Aduhelm: What To Know About The New Alzheimers Drug
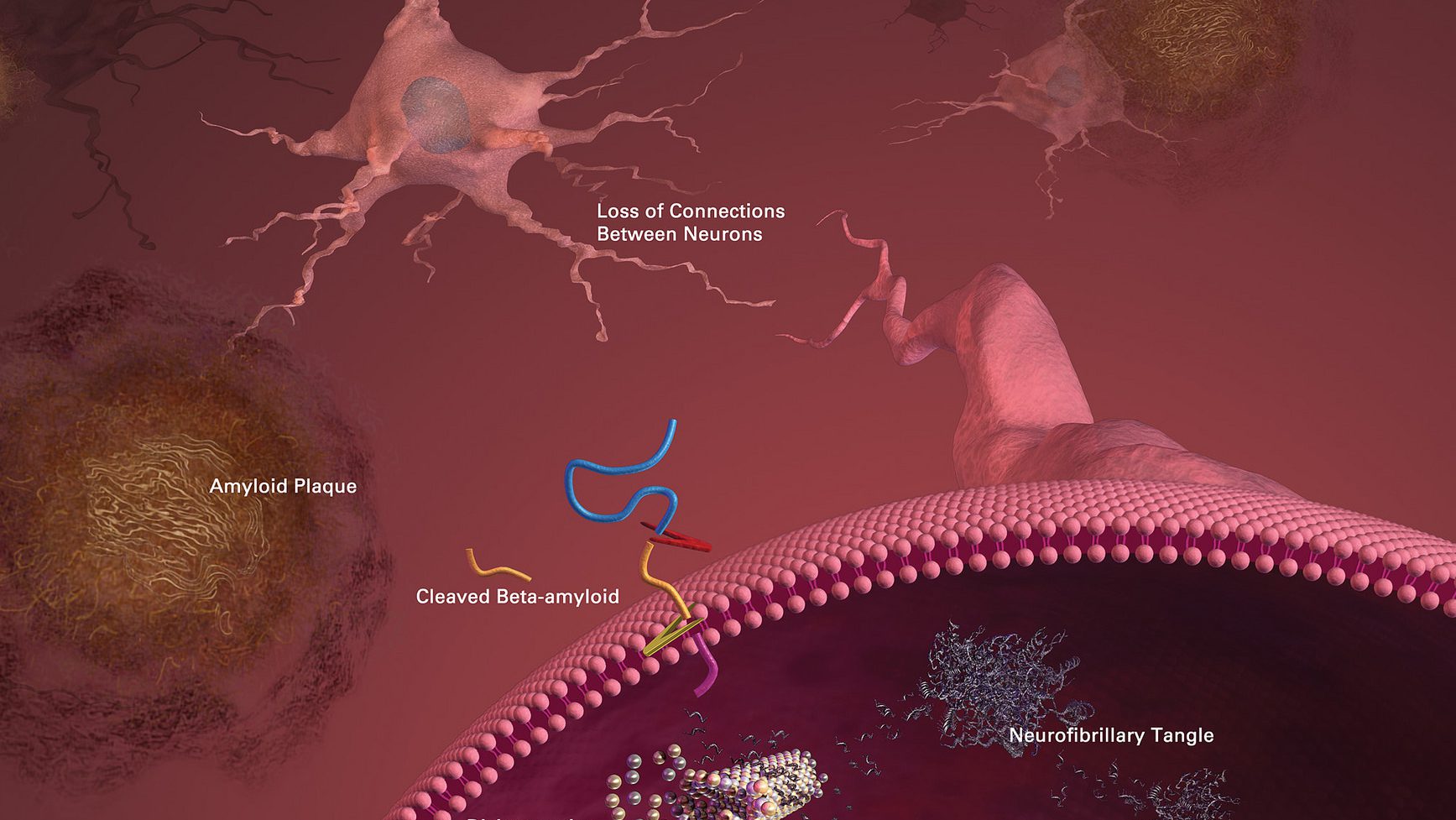
F.D.A. approval.In June 2021, the Food and Drug Administration approved Aduhelm, the first new Alzheimers medication in nearly two decades. The approval was made despite opposition from many experts, who cited data showing it wasnt clear whether the drug works and it had serious safety risks.
A controversial drug.The F.D.A.s independent advisory committee and a council of senior agency officials voted overwhelmingly that data failed to conclusively show that Aduhelm slowed cognitive decline. Still, the agency granted approval for the drug but required Biogen, the drugs manufacturer, to conduct a new clinical trial that will take several years.
Growing concerns.Questions about the approval, and whether the F.D.A. worked too closely with Biogen, have prompted investigations by two powerful congressional committees, the Health and Human Services departments inspector general and other agencies. Major medical centers, including the Cleveland Clinic, have declined to offer Aduhelm.
Medicare coverage.Citing Aduhelm’s uncertain benefit and significant safety risks, Medicare officials announced that the federal insurance program would limit coverage of the drug to participants in clinical trials. A powerful Alzheimer’s advocacy group had pushed relentlessly for broad access.
Some experts said the F.D.A.s quick reversal was a sign that it had mishandled its initial review and was now ending up closer to where it should have started.
You May Like: Where To Buy Jelly Drops For Dementia Patients
Inside Project Onyx: How Biogen Used An Fda Back Channel To Win Approval Of Its Polarizing Alzheimers Drug
Huangs team then scanned a database of 1,300 drugs to find ones that could flip those altered genes back to a healthy state. Among the top five hits was bumetanide, a powerful diuretic first approved by the FDA in 1983.
They tested its effect first on a line of mice engineered to have two copies of the human APOE4 gene, causing them to develop memory problems and other cognitive deficits around 15 months of age the equivalent of 60 in human years. Bumetanide treatments significantly boosted how well these mice performed on various cognitive and spatial memory tests.
They then repeated the experiment in another line of mice, who in addition to APOE4 genes also carried the human gene for APP a protein that when broken down becomes beta-amyloid. These mice typically develop plaques at 6 months of age. But bumetanide treatments delivered at 10 months shrank these plaques and restored healthier brain function. Taken together, the results suggest that bumetanide is improving symptoms without specifically targeting plaques.
There are many cellular and molecular changes in Alzheimers disease patients besides plaques, but we usually dont talk about them, said Huang. These results suggest that in order to treat Alzheimers we should probably not target only one or two but multiple genes and multiple pathways involved in the disease.
S To Reverse Dementia And Memory Loss As You Age
Recently, I spoke on a panel for PBS TV at the American Association of Retired Persons convention in Boston. The topic was dementia.
There was a woman with mild cognitive impairment on the panel. Her condition is sort of like pre-Alzheimer’s disease. Everyone on the panel — including the Harvard neurologist — agreed that memory loss is NOT a normal part of aging. The sad part was that the panel didn’t have much to offer people in the way of prevention. Their only solution was just a very bad and pretty ineffective selection of drugs with lots of side effects.
But there is another way to think about brain aging. The brain responds to all the same insults as the rest of the body — stress, poor diet, toxins, lack of exercise or sleep, nutritional deficiencies, and more. All we have to do is give the brain a tune-up and we can see miracles. In today’s blog I will give you nine tips that will allow you to do that. But first, let’s look a little more closely at the magnitude of this problem.
Dementia on the Rise
Dementia is a big problem and growing every day. Ten percent of 65-year olds, 25 percent of 75-year olds, and 50 percent of 85-year olds will get Alzheimer’s disease — at a cost of $60 billion a year to society. Worse, the number of people with Alzheimer’s is predicted to triple in the next few decades. It is now the seventh leading cause of death.
I’d like to illustrate this through the story of one of my patients who had a diagnosis of dementia.
Don’t Miss: Purple Ribbon Alzheimer’s
Identifying Who’s At Risk Of Dementia
Experts know that damage to the brain caused by Alzheimer’s disease can start many years before symptoms appear. If people at risk of Alzheimer’s could be identified at an early stage, it is hoped that treatments could be offered that would slow down or even stop the disease.
A major study, called PREVENT, concentrates on people in their 40s and 50s to identify those who are at greater risk of developing Alzheimer’s . It aims to understand what is happening in their brains before symptoms appear.
Specialised brain scans, known as PET scans, have been developed to study two proteins in the brains of those with Alzheimer’s disease. The aim is to increase the understanding of the disease process, and also to identify those people who will benefit most from new drug treatments.
Although PET scans are sometimes used to help with a dementia diagnosis, these highly specialised scans are usually only available as part of clinical trials.
A number of different trials are now under way in people who are currently well but are at increased risk of Alzheimer’s disease.
Small Trial Shows Memory Loss From Alzheimer’s Disease Can Be Reversed
A small clinical trial of 10 patients with early Alzheimers disease has shown that the memory loss and cognitive impairment can be reversed.
Not only were improvements sustained, but some patients returned to work, regained their ability to speak different languages, and showed an increase in brain matter volume after just a few months.
“All of these patients had either well-defined mild cognitive impairment, subjective cognitive impairment, or had been diagnosed with Alzheimers disease before beginning the program,” says one of the team, Dale Bredesen, University of California, Los Angeles. “Follow up testing showed some of the patients going from abnormal to normal.”
The study investigated the effects of a new kind of personalised treatment on the cognitive abilities of 10 patients who were experiencing age-related decline.
The treatment – called metabolic enhancement for neurodegeneration, or MEND – is based on 36 different factors, including changes in diet, exercise, and sleeping habits, plus the integration of certain drugs, vitamins, and brain stimulation therapy to their regular routine.
These lifestyle changes and treatments were sustained for five to 24 months, and the team from UCLA and the Buck Institute for Research on Ageing in California reports that many of the patients showed real, life-altering improvements as a result.
The team has split the 10 patients up into individual case studies, which you can read about in their open access paper.
Also Check: Alzheimer’s Purple Color Code