Researchers Find New Hints That Could Explain How The Disease Spreads In Human Brains
Case Western Reserve University researchers studying human prionsmisfolded proteins that cause lethal and incurable diseaseshave, for the first time, identified surface features responsible for the replication of prions in the brain.
The ultimate goal of their research is to design a strategy to stop prion disease in humansand, ultimately, to translate new approaches to work on Alzheimers and other neurodegenerative diseases.
Scientists have yet to discover the exact cause of Alzheimers disease, but largely agree that protein issues play a role in its emergence and progression. Alzheimers disease afflicts more than 6 million people in the U.S., and the Alzheimers Association estimates that their care will cost an estimated $355 billion this year.
Research was done at the Safar Laboratory in the Department of Pathology and the Center for Proteomics and Bioinformatics at Case Western Reserve University School of Medicine, and at Case Western Reserves Center for Synchrotron Bioscience at Brookhaven Laboratories in New York. Jiri Safar, professor of pathology, neurology and neurosciences at Case Western Reserve University School of Medicine, leads the work. The report, Structurally distinct external domains drive replication of major human prions, was published in the June 17 issue of PLOS Pathogens.
The researchers developed a new three-step process to study human prions:
What Is The Underlying Cause Of Prion Disease
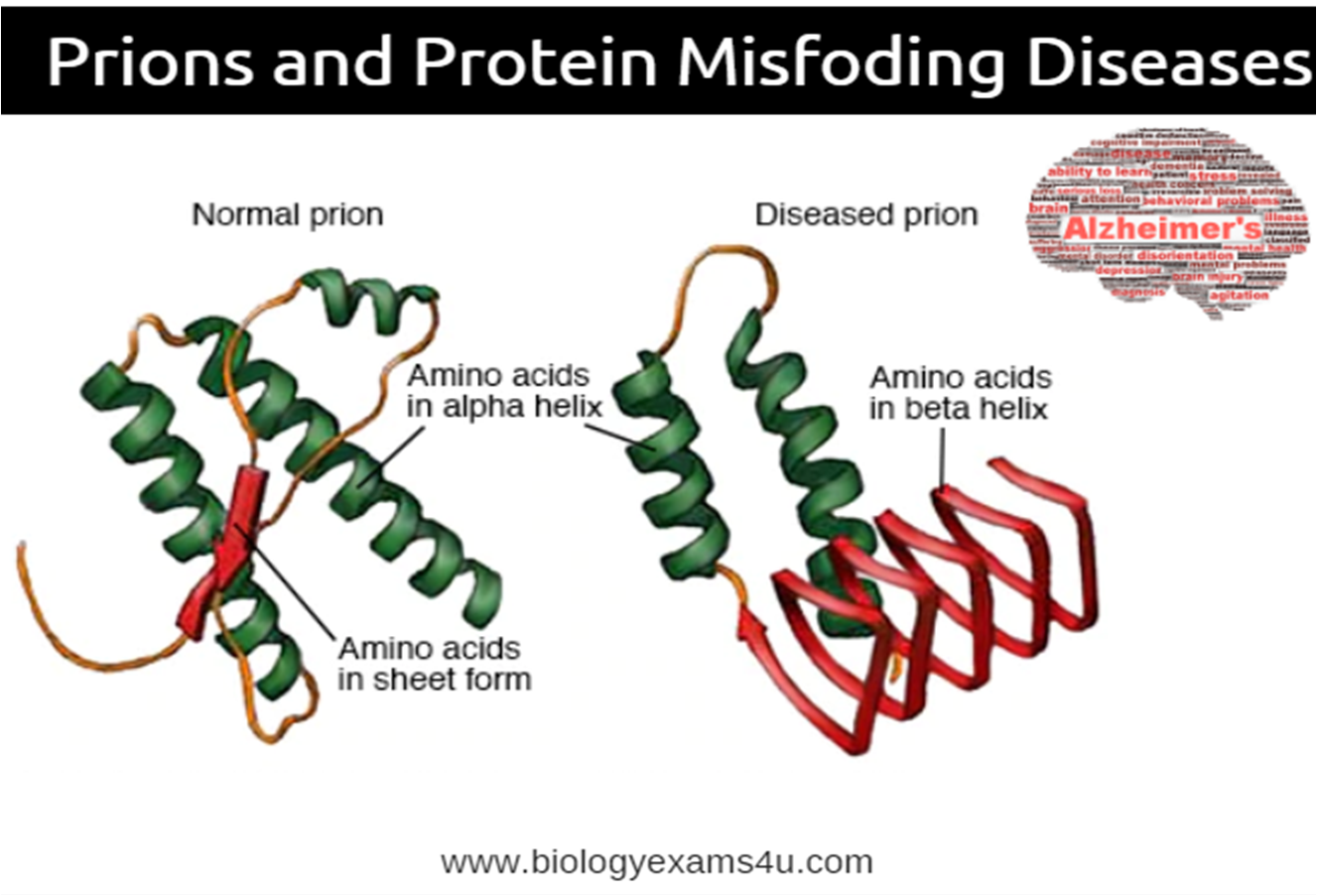
What causes prion disease? Prion diseases occur when normal prion protein, found on the surface of many cells, becomes abnormal and clump in the brain, causing brain damage. This abnormal accumulation of protein in the brain can cause memory impairment, personality changes, and difficulties with movement.
Prpc: A Novel Therapeutic Target For Ad
PrPC has been identified as a major player in mediating the toxicity of A oligomers that leads to synaptic loss and cognitive impairment in AD. Therefore, targeting PrPC, its interaction with A oligomers, or downstream mediators can be considered the new line of choice for therapeutic development for treatment of Alzheimer’s disease.
Genetic ablation of PrPC in mice rescues the neurotoxic phenotypes of A oligomers . It might be reasonable to speculate that using shRNA or siRNA to knock down the expression of PrPC may represent a therapeutic approach for AD, though little has been done in this regard. Nevertheless, knocking down PrPC will also affect other functions of PrPC, causing various complications. For example, PrPC reduces A production by inhibiting BACE1 activity, and has a protective role in AD . This can be jeopardized by PrPC knockdown.
Screening for small molecules that could efficiently target either the A oligomer/PrPC interaction or the downstream mediators may represent a promising avenue for therapeutic development.
The Fyn kinase has been found to be activated upon binding of A oligomers with PrPC, which then initiate downstream signaling to mediate A toxicity, for example, activation of Fyn kinase lead to hyperphosphorylation of tau . Targeting Fyn kinase or other A/PrPC downstream mediators, for example by genetic engineering, RNAi, or small molecule modulators, may also be of therapeutic value.
Recommended Reading: How To Move A Parent With Dementia To Assisted Living
Vaccines And Alzheimers Disease: A Brief History
One of the earliest antivaccine claims that I ever dealt with was a rather specific claim about the influenza vaccine. I first encountered it when Bill Maher parroted it in an interview with Larry King on Larry King Live, way back in December 2005. I think its useful to recount what Maher said in this exchange:
MAHER: Im not into western medicine. That to me is a complete scare tactic. It just shows you, you can
KING: You mean you dont get a you dont get a flu shot?
MAHER: A flu shot is the worst thing you can do.
KING: Why?
MAHER: Because its got its got mercury.
KING: It prevents flu.
MAHER: It doesnt prevent. First of all, thats
KING: I havent had the flu in 25 years since Ive been taking a flu shot.
MAHER: Well, I hate to tell you, Larry, but if you have a flu shot for more than five years in a row, theres ten times the likelihood that youll get Alzheimers disease. I would stop getting your
KING: What did you say?
MAHER: That went better in rehearsal but it was still good. Absolutely, no the defense against disease is to have a strong immune system. A flu shot just compromises your immune system.
A chiropractor. Of course, the presentation at NVIC had to be recorded by a chiropractor:
This is an old claim, dating back 24 years!
And heres a more recent addition since 2005, dated 2006:
But what about prion disease? And what are prion diseases, anyway?
Altered Levels Of Prpc Involved In Ad Pathology
PrPC play an important role in the pathogenesis of AD. Some pathological evidence indicates that PrPC deposits often accompany A plaques in AD . The importance of the association between PrPC and A is greatly strengthened when it was demonstrated that PrPC was the receptor for the high affinity to A42 oligomers on cells .
Reported data suggest a regulatory influence of PrPC expression in the pathological process of AD. The altered expression of PrPC in aging and the development of AD are associated with disease progression, and it has been observed that PrPC is decreased in the hippocampus and temporal cortex in aging and sporadic AD but not in familial AD, suggesting that PrPC expression reduced reflects a main mechanism of disease and is not merely a minor consequence of other AD-associated changes . In a study by Velayos et al. , there was a tendency for a lower expression of PrPC in AD patients than in healthy patients, which indicated that existing PrPC expression may play a protective role in AD.
In addition, other studies have focused on PrPC expression level alterations in advanced stages of AD, mainly stage Braak III to VI, most likely due to neuronal loss. Vergara et al. demonstrated that, in AD patients with Braak stages IVI, PrPC protein expression in the brain increases in the early stages of AD and peaks at approximately stage III. Thereafter, PrPC expression decreases until the manifestation of clinical symptoms in both cases .
Recommended Reading: Do People With Dementia Know They Have It
What Causes Prion Disease
Prion diseases occur when normal prion protein, found on the surface of many cells, becomes abnormal and clump in the brain, causing brain damage. This abnormal accumulation of protein in the brain can cause memory impairment, personality changes, and difficulties with movement. Experts still don’t know a lot about prion diseases, but unfortunately, these disorders are generally fatal.
Infectious Proteins Known As Prions Are Involved With Most Forms Of Neurodegenerative Disease The Global Epidemic Has More To Do With The Prion Contagion Than Age
Dr. Stanley Prusiner, an American neuroscientist from the University of California at San Francisco, earned a Nobel Prize in 1997 for discovering and characterizing deadly;prions;and;prion disease. Prusiner claims that all TSEs, including Alzheimers disease, are caused by prions.;Prions are a deadly and unstoppable form of protein. They migrate, mutate, multiply and kill with unparalleled efficiency.
President Obama awarded Prusiner the National Medal of Science in 2010 to recognize the importance of his research. According to Prusiners science, TSEs are a spectrum disease. Creutzfeldt-Jakob disease, which is extremely aggressive and extremely transmissible, is at the extreme end of the spectrum. Unfortunately, Prusiners science is being ignored and we are facing a public health disaster because of the negligence.
Neurologists dont know where along the prion spectrum the disease becomes transmissible. The entire spectrum could represent a transmissible disease. Unfortunately, neurologists are not warning these patients and their caregivers about the risks of exposure. Even those with CJD;are not quarantined now. They are sent home, where they can infect friends, family, caregivers, clinics, dental offices, restaurants and entire communities.
The brain diseases caused by prions include Alzheimers, Parkinsons, Huntingtons, amyotrophic lateral sclerosis , and other disorders known as frontotemporal dementias, said Prusiner.
Read Also: Is There A Support Group For Dementia
Molecular Consequences Of The Prpc/ao Interaction In Ad
The molecular and cellular consequences of the PrPC-A oligomer interaction are dependent on raft-based complexes. The integrity of cholesterol-rich lipid rafts is critical for the interaction between A42 with PrPC. It has been demonstrated that the PrPC-mediated toxicity of A oligomers and the activation of downstream pathways require lipid rafts . GPI-anchored PrPC is localized to the cholesterol-rich lipid raft microdomains of the plasma membrane . Cholesterol depletion disrupts these rafts with PrPC being redistributed into nonraft regions of the membrane . A study revealed that the disruption of the rafts causes a significant reduction in A oligomer binding to cells and prevents the activation of Fyn kinase .
A growing body of evidence suggests that PrPC mediates downstream intracellular processes through many different receptors, including the metabotropic glutamate receptors mGluR1 and mGluR5, the 7 nicotinic acetylcholine receptor, the kainite receptor GluR6/7, and AMPA receptor subunits GluA1 and GluA2 . These studies indicate that PrPC functions as an extracellular scaffolding protein that is able to organize multiprotein complexes that mediate intracellular signal transduction at the cell surface.
Thus, further investigations have been made to expound these complex and their downstream pathways to prevent neurotoxic consequences .
Prion Contamination Disease Unstoppable
If you think that you and your family are immune to the surging epidemic of neurodegenerative disease, think again. Neurodegenerative disease,;including;Alzheimers disease,;is;the fastest-growing cause of death in the world.;Its getting worse every day thanks to mismanagement and misinformation.
Infectious proteins known as prions are involved with most forms of neurodegenerative disease in mammals. Prion disease is known in neurology as;transmissible spongiform encephalopathy;. The operative word is transmissible.
It appears that a variety of factors can contribute to neurodegeneration, including genetics, neurotoxins and head trauma. Once the neurodegeneration begins, the cause is the least of our concerns. At this stage, the misinformation and mismanagement are fanning the flames of a global epidemic. Each victim of prion disease becomes an incubator and distributor of the prion pathogen. Prions are now the X factor within a public health disaster.
The global epidemic has more to do with the prion contagion than age. In fact, it appears that the autism epidemic involves neurotoxins and protein accumulation in the brain. The biggest difference between Alzheimers disease and autism could be age. The pathology is different because of the differences between a neurodevelopmental brain and a neurodegenerative one when exposed to the same protein-based neurotoxin.
Recommended Reading: Is Nursing Home Care For Dementia Tax Deductible
Causes And Risk Factors
Prion diseases are transmissible in certain circumstances, but they are not infectious in the usual way. So they are not spread by airborne droplets, blood or sexual contact.
Contact with someone with CJD does not lead to increased risk of developing the conditions no special precautions are required.
The infectious agent is thought to be a prion, an abnormal form of a protein called PrP, which in its natural form occurs in the brain and parts of the body of humans.
Unlike bacteria and viruses, prions are not inactivated by heat, ultraviolet light or other standard sterilisation procedures. Normal Prp can convert into abnormal PrP, named PrPres, which leads to disease.
Scjd Induces H In Hu Brnaggs
Untreated Hu BrnAggs produced large bands of tau . Exposure to a normal brain homogenate produced smaller bands and 2 very weak bands . Exposure to sCJD produced bands between 40 and 50 kDa and strong bands of phosphorylated tau .
In another preliminary study, we made BrnAggs from transgenic mice expressing human mutated tau. These mouse tau BrnAggs spontaneously form small numbers of neurons containing H in nerve cell bodies and dendrites. When Mo BrnAggs were exposed to an A42-containing homogenate from a transgenic 2576 mouse, the number of neurons containing abnormal tau increased approximately 2-fold. In contrast, when tau BrnAggs were exposed to a brain homogenate from a wild-type mouse infected with Rocky Mountain Laboratories scrapie prions containing PrP Sc , the number of neurons and their processes containing abnormal tau increased 10-fold. These results were reproduced in triplicate. The findings argue that exposure to A42 and PrP Sc increases the levels of H inclusions in tau BrnAggs. We did not use normal age-matched human brain homogenate controls in these studies, which is why we classified them as preliminary .
Don’t Miss: What Is The Difference Between Alzheimer’s And Parkinson’s
Is Parkinsons Disease A Prion Disease
Overexpression of -synuclein alone can induce a PD syndrome in animals and humans. It is thus possible that PD is a prion disorder resulting from increased production and/or impaired clearance of proteins such as -synuclein, leading to misfolding and the formation of toxic oligomers, aggregates, and cell death.
Exposure Of Brnaggs To Prions
After 15 days in culture, Mo BrnAggs were exposed for 10 days to Rocky Mountain Laboratories prions derived from scrapie-infected CD1 mouse brains. After 41 days in culture, Mo BrnAggs were harvested and analyzed by immunohistochemistry. Paraffin-embedded sections were stained for phosphorylated tau with mouse anti-tau13 antibody , mouse anti-tauCP13 antibody , or the secondary antibody goat anti-mouse Alexa Fluor 488 . Three to 4 stacks of approximately 40 serial sections were captured by confocal microscopy . Whole BrnAggs were examined throughout by a fluorescence microscope with a 40 × lens .
For experiments with Hu BrnAggs, the cells were exposed to a human brain homogenate containing either sCJD made from the thalamus of sCJD cases or normal brain after 2 days in culture. Treatments were continued for up to 20 days in culture. Human BrnAggs were harvested after 35 days. Twenty BrnAggs were pooled and analyzed for PrP Sc by Western blot analysis using anti-prion protein HuM-P horseradish peroxidase antibody, A142 specific antibody , and anti-tau oligomeric antibody overnight at room temperature.
You May Like: How Long Does Uti Dementia Last
Concluding Remarks And Perspectives
Although there are conflicting reports regarding the function of PrPC as a cell surface receptor to mediate the deleterious effects of A oligomers in AD, there are no ambiguities for two end points: high affinity binding of A oligomers to PrPC , and high synaptic toxicity of A oligomers . The challenge remaining for scientists is to make the two ends meet.
Strittmatter group’s finding that PrPC acts as the receptor for mediating A oligomer neurotoxicity has at least opened a new direction towards understanding the molecular mechanism that connect A oligomers and their toxic effects. The research discoveries of his team have apparently been supported by multiple studies from other groups, though there have been conflicting results reported. Sophisticated studies using advanced animal models and optimized experimental conditions are needed to elucidate the precise role of PrPC in mediating A oligomer neurotoxicity, or to identify other potential cell surface receptors and signaling networks that make the two ends toxin and toxicity meet.
If the role of PrPC in A oligomer-mediated pathogenic process turns out to be substantial, it would be of interest to seek potential co-receptors or to examine whether other A-mediated signaling pathways are also PrPC-dependent, making a complete prion connection network of AD.
Do Vegans Get Alzheimers
Recent research suggests that Alzheimers disease, like heart disease and strokes, is linked to the saturated fat, cholesterol, and toxins found in meat and dairy products. Studies have shown that people who eat meat and dairy products have a greater risk of developing Alzheimers disease than do vegetarians.
Recommended Reading: Are Jigsaw Puzzles Good For Dementia
Scientists Develop Test To Identify Alzheimers Parkinsons And Prion Diseases
Researchers may have developed a new test that can easily identify prion diseasesa group of diseases that includes Mad Cow diseasein a new study. The test may also have implications for identifying beta-amyloid proteins in Alzheimers disease.
According to Johns Hopkins Medicine, prions are a type of protein that causes other proteins in the brain to fold, leading to certain rare diseases like Creutzfeldt-Jakob disease .
Its also possible that prions are linked to Alzheimers disease. Some recent scientific discussions have centered around the idea that both beta-amyloid and tau proteins are prions.
Prion diseases are rare, but there are many mis-folded protein-based diseases, which affect millions of people, that are also very poorly understood, Ronald Zuckermann, senior scientist at Berkeley Labs Molecular Foundry and a researcher on the study, said in a news release.
The new method was outlined by researchers at Lawrence Berkeley National Laboratory in a study published in PLOS One, and singles out prion proteins in body fluid samples. The researchers tested this in animals, and concluded they were one step closer to establishing a noninvasive prion detection test in humans.
Its like Velcro, Michael Connolly, senior scientific engineer at Berkeley Labs Molecular Foundry, said in the news release, explaining that misfolded proteins have binds that attach to peptide beads.
Research Now Shows That Amyloid Plaques And Tau Tangles From Diseased Brains Can Infect Healthy Brain Tissue The Infectivity Of Brain Tissue Is Just The Tip Of The Iceberg
Prions invade the entire body, but they do their damage in the brain. Prions are in the bodily fluids and cell tissue of victims, including blood, saliva, mucus, urine and feces. Common household items become biohazards, including utensils and dishes. A cough or sneeze sends prions into the air. Prions are a real-world version of Pandoras Box. Prions are unstoppable. Prion disease is always fatal. This is an extremely important issue that demands the truth from government and industryespecially the medical world.
Prions migrate, mutate, multiply and kill with unparalleled efficiency. Containing prions and preventing their spread is the key to avoiding a public health disaster. Unfortunately, the floodgates have been wide open for years.
Prion disease also is known as transmissible spongiform encephalopathy .;The operative word is transmissible. Prusiner;claims that all forms of TSE are caused by infectious prions. TSE is a spectrum disease that varies in severity and symptoms. The diagnosis depends on which region of the brain is impacted first and by what prion mutation. Few cases are identical in terms of symptoms and diagnoses.
Prions are such a formidable threat that the U.S. governmentenacted the Bioterrorism Preparedness and Response Act of 2002, which includeda provision to halt research on prions in all but two laboratories.
You May Like: Is Weight Loss A Symptom Of Dementia