The X Chromosome Is Unique
In women, there is a systematic demand to compensate for SCD by silencing one of the copies of the X chromosome. With two X-chromosomes, women are more prone to inheriting potentially deleterious mutations in X-encoded genes, which, because of Xi, may all be expressed in different cells. The first finding of inactivation of the X chromosome was reported by Lyon, . It was found that one of the X chromosomes, paternal or maternal, was always inactivated, suggesting that an inactivation mechanism only allows active transcription at one X chromosome . This process of X-chromosome inactivation evolved as a mechanism to regulate gene dosage. As a compensation mechanism, it does not affect all genes equally, and those genes that are not affected are known to escape XCI .
Xi acquires several features of heterochromatin, such as hypermethylation, hypercondensation, altered replication patterns , and depletion of acetylated histones . Methylation patterns have been extensively used to determine the inactive chromosome , enabling an analysis of non-random inactivation processes in diseases that are X chromosome-linked . In our published study we suggest that changes in the inactivation patterns of the X-chromosome could have an impact on AD pathogenesis .
What Is Alzheimer’s Disease
Alzheimer’s disease is a brain disorder that causes severe cognitive decline in older people. It’s one of the leading causes of dementia. Dementia is a condition that affects how a person thinks, functions, and behaves. It also mainly affects a person’s memory and how new memories are formed.
There are two forms of the condition. Early-onset Alzheimer’s occurs in people between the ages of 30 and 60. It is also very rare. Late-onset Alzheimer’s is the more common form of the condition.
The condition is named after Dr. Alois Alzheimer, who observed abnormalities in the brain of a woman who had died due to a suspected mental illness. He noticed unusual clumps called amyloid plaques on her brain. This build-up can be observed in the brains of people who have the condition today.
Can Alzheimer’s Disease Be Prevented
Unfortunately, there isn’t much to be done to avoid certain risk factors such as gender and age that increase the risk of developing Alzheimer’s disease.
Research does show that specific lifestyle changes could help. Maintaining a healthy diet, exercising regularly, and cutting down on smoking and drinking alcohol could reduce your risk of developing the disease.
In cases where the disease has already developed, these lifestyle changes could also potentially help slow the disease’s progression.
Read Also: Dementia Ribbon Colour
Genes Associated With Risk In Sporadic Ad
AD2: APOE
Inheritance and clinical features
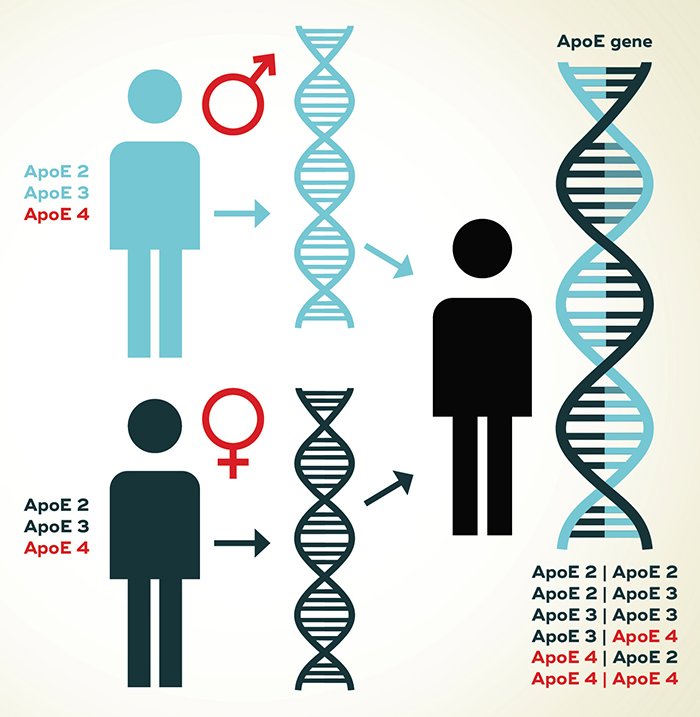
The APOE gene has been associated with both familial late-onset and sporadic late-onset AD in numerous studies of multiple ethnic groups. The APOE4 genotype is associated with higher risk of AD, earlier age of onset of both AD and Down syndrome , and a worse outcome after head trauma and stroke, both in humans and in transgenic mice expressing human APOE4. The frequency of the APOE4 allele varies between ethnic groups, but regardless of ethnic group, APOE4 carriers are more frequently found in controls and APOE4+ carriers are more frequently found in patients with AD.,
Gene location and structure
Apolipoprotein E structure and single nucleotide polymorphisms . The general protein structure of APOE is shown. The 2 SNPs in exon 4 and corresponding protein locations are shown . The 3 APOE 4 alleles are defined by 2 SNPs, rs429358 and rs7412, with 2 defined by nucleotides T-T 3 defined by T-C, and 4 defined by C-C, respectively.
Gene function and expression
The mechanisms that govern apoE toxicity in brain tissue are not fully understood. Some proposed mechanisms include isoform-specific toxicity, APOE4-mediated amyloid aggregation, and APOE4-mediated tau hyperphosphorylation.
In addition, individuals carrying APOE4 have higher amyloid and tangle pathology and an increase in mitochondrial damage compared to those carrying other APOE polymorphisms.
Genetic variation
Sex Chromosome Dosage : An Engine Of Stability
The crosstalk that exists between X chromosomes and autologous genes is a relatively new paradigm that has emerged as a result of the biology of sex differences, and gives rise to the question of how SCD shapes the genome function. To explore this, human sex aneuploidies were analyzed from a genome-wide expression dataset by Raznahan et al. where they found a dosage sensitivity of the X-Y chromosome pair resulting in increased expression of genes that decrease X/Y chromosomal dosage . The most interesting finding was that X-linked genes were found to regulate co-expression of networks of autosomal genes that are SCD-sensitive and, in addition to these findings, suggest that the autosomal genes and their corresponding networks are crucial for cellular functions. This highlights the potential of SCD to affect the occurrence of disease.
The most common aneuploidy in AD is XO mosaicism . In respect to SCD and the XO status, Raznahan et al. have demonstrated up-regulation of the protein networks, noncoding RNA metabolism, suppression of the cell cycle, changes in regulation of DNA/chromatin organization, glycolysis, and response to stress . Changes in these collective networks through XO and supernumerary XXY, and XXYY syndromes may enhance the risk of AD .
Figure 2 X chromosome instability, Sex Chromosome Dosage, Topological changes of Chromosomes, and its possible role in AD.
Recommended Reading: Ribbon Color For Dementia
How Is Alzheimer’s Disease Treated
There is currently no cure for Alzheimer’s disease. However, scientists have developed treatments that can reduce the severity of some of the condition’s debilitating symptoms.
Treating Alzheimer’s disease effectively typically involves a combination of medication and therapy. Specific lifestyle changes are also recommended to help a person with the condition live a more fully functioning life.
Female Chromosomes Offer Resilience To Alzheimers
Study Reveals the Second X Chromosome Confers Protection
Women with Alzheimers live longer than men with the disease, and scientists at UC San Francisco now have evidence from research in both humans and mice that this is because they have genetic protection from the ravages of the disease.
The new study offers a first look at how sex chromosomes affect vulnerability to Alzheimers. And it helps explain why women survive longer and with less severe symptoms than men during early stages of the disease, even when they have comparable levels of toxic Alzheimers proteins in their brains.
This finding challenges a long-standing dogma that women are more vulnerable to Alzheimers, said Dena Dubal, MD, PhD, associate professor of neurology at UCSF and senior author of the study, published Aug. 26, 2020, in Science Translational Medicine. Dubal is the David Coulter Endowed Chair in Aging and Neurodegenerative Disease, and a member of the UCSF Weill Institute for Neurosciences. More women than men have Alzheimers because they survive to older ages, when risk is highest. But they also survive with the disease for longer.
You May Like: Bob Knight Health
The Role Of Genetics: Will I Get Alzheimers Disease
Scientists are still trying to determine the underlying causes of Alzheimers disease. The hope is that one day we will be able to stop the disease from progressing or perhaps even prevent it altogether. To date, researchers have identified a few genes that play an important role in Alzheimers. Some of these genes are simply risk factors for Alzheimers disease. Other genes are hereditary and will cause Alzheimers disease to develop.
There are two types of Alzheimers disease: early-onset Alzheimers disease and late-onset Alzheimers disease. Early-onset Alzheimers disease is rare, occurring in people age 60 and younger. This represents less than 5% of all people with Alzheimers. One type of early-onset Alzheimers disease is known as autosomal dominant Alzheimers disease or early-onset familial Alzheimers disease . This is even more uncommon, affecting less than 1% of all people with Alzheimers. What makes this type of early-onset Alzheimers disease so unusual is that it is caused by a hereditary genetic mutation to one of three genes PSEN1, PSEN2, or APP.
A recent study examined data from 4 large, observational studies of adults ages 60 and older. The study reported the association between various APOE genotypes and the risk of developing mild cognitive impairment or dementia due to Alzheimers by age 85 as follows:
- No family history of dementia
- Being male
Factors that may increase a persons risk of developing Alzheimers include:
Showing Benefit Of Second X Chromosome
To get closer to this causal question, the scientists performed experiments in mice. First, they looked inside the brains of female mice and confirmed that both copies of Kdm6a were actively transcribing RNA to make protein. Female mice had significantly higher levels of this protein in a brain region called the hippocampus, which is critical to learning and memory and gets damaged early in Alzheimers.
Then they bred mice which model human Alzheimers by producing toxic amyloid beta in their brains, so their male offspring produced amyloid and also carried two X chromosomes like females.
With a second X chromosome, the male mice did better on cognitive tests, and they also lived longer, despite the toxic proteins in their brains. To be sure it was the second X that provided protection, rather than the absence of a Y chromosome, scientists deleted the second X from female Alzheimers mice. And just like males, these female mice were more cognitively impaired and died faster.
In further tests, when the scientists exposed neurons from male and female mouse brains to increasing doses of amyloid beta, the male neurons died faster. But this difference was eliminated when the scientists used a gene editing technique to reduce Kdm6a protein levels in the neurons from females brains and increased it in the neurons from male mouse brains.
Recommended Reading: Did Reagan Have Alzheimers
Expression In Various Cell Types
The expression of genes was assigned to specific cell classes of the adult brain, as described previously. Briefly, middle temporal gyrus single-nucleus transcriptomes from the Allen Brain Atlas dataset were used to annotate and select six main cell classes using Seurat 3.1.1 : glutamatergic neurons, GABAergic neurons, astrocytes, oligodendrocytes, microglia and endothelial cells. Enrichment analyses were performed by using the mean gene expression per nucleus for each cell type relative to the total expression summed across cell types as a quantitative covariate in a MAGMA gene property analysis.
Key Points About Early
-
Alzheimer disease commonly affects older people, but early-onset Alzheimer disease can affect people in their 30s or 40s.
-
It affects memory, thinking, and behavior.
-
Although there is no known cure, early diagnosis and treatment can lead to better quality of life.
-
Stay healthy with a good diet and regular exercise.
-
Avoid alcohol and other substances that may affect memory, thinking, and behavior.
You May Like: Where Does Bobby Knight Live
Can Genetic Testing Determine Alzheimers Disease Risk
Having a family history of Alzheimers disease might make you concerned about developing it yourself at an older age. In that case, you may want to undergo genetic testing to know about the probability of developing Alzheimers.
Multiple researchers have highlighted the link between an increased Alzheimers risk and a persons genetics. Scientific findings show that individuals with first-degree relatives that have had Alzheimers are at a greater risk of developing the disease. The risk multiplies further if a person has more than one first-degree relative with Alzheimers.
Although scientific advancements, such as genetic testing, can conveniently detect hereditary diseases, the question remains, would it even be helpful to determine an individuals risk of developing Alzheimers via this technique? Before we know the answer to this question, we must know about the genes and genetics of Alzheimers disease.
How Can I Reduce My Risk Of Dementia
For the vast majority of people, our genes are only one factor affecting our risk of dementia. There are many other factors involved, such as age and lifestyle. While we cannot change our age or genes, research has found that up to a third of all cases of dementia could be avoided through lifestyle changes.
There are simple things we can do that may help lower our risk:
- Do not smoke.
- Drink fewer than 14 units of alcohol per week.
- Control high blood pressure.
- Keep cholesterol at a healthy level.
- Keep active and exercise regularly.
- Maintain a healthy weight.
- Eat a healthy balanced diet.
You can find more detailed information about how you can reduce your risk of dementia here.
Also Check: Does Diet Coke Cause Dementia
Patients Carrying One Or Two Apoe4 Alleles
The by far largest single genetic risk factor for LOAD is apolipoprotein E , which has been known since 1993. There are three common isoforms of APOE , which result from polymorphic variation in the gene. APOE4 is associated with higher risk of AD . However, APOE4 cannot be regarded as causal in the development of AD, since it is neither sufficient nor necessary to cause AD. This means, individuals carrying one or two of the APOE4 risk alleles will not certainly develop AD and individuals without APOE4 are not protected. However, its high importance can be explained by its relatively high frequency in the population combined with a relatively high effect strength, which is also illustrated in Fig. . Other known genetic variants either occur many times less frequently or have only minor effect on disease development.
The APOE4 allele has been described to promote amyloid deposition starting already in middle age. PET studies in cognitively healthy individuals in various age groups have shown that APOE4-positive individuals exhibit amyloid deposition significantly earlier than APOE4-negative individuals . Although APOE4 has been known as a risk factor for a very long time, there remains disagreement over the specific mechanisms by which the APOE4 allele increases the risk of AD and age-related cognitive decline.
How Can Genes Cause Disease
Genes often play a role in the development of diseases. This can happen in two main ways.
Single-gene diseases
Sometimes, a gene changes and this causes a disease. This is known as a single-gene disease because it is caused by a change in just one gene. Single-gene diseases are often serious, but they are rare.
It is possible to directly inherit a single-gene disease. This means that if a child inherits the same changed gene that a parent has, they are very likely in some cases almost certain to develop the same disease.
Complex or multiple-gene diseases
Complex diseases are less straightforward. They develop through the interaction between several different factors. These are known as risk factors for that disease. They include:
- the persons environment or lifestyle for example, their diet and whether they smoke or not
- multiple gene variants known as risk variants because they increase the persons risk of developing the disease.
It isnt possible to inherit a complex disease in the same way as a single-gene disease. This is because complex diseases are caused by multiple factors acting together.
However, a person with a family history of a complex disease will often have an increased chance of developing it. This is because they are likely to share risk factors with other birth relatives. Despite this increased chance, the person still might never develop the disease.
Also Check: Alzheimer’s Purple Color Code
Dementia And Down Syndrome
People with Down syndrome are born with an extra piece of DNA. This means they also have an extra copy of the APP gene. This leads to the build-up of amyloid plaques in the brain, which play a role in the development of Alzheimers disease. While not everyone with Down syndrome will go on to develop symptoms of Alzheimers, most people with the condition over the age of 40 will have amyloid build-up. It is estimated that about 50% of people with Down syndrome develop symptoms like memory loss, usually in their 50s and 60s.
To find out more about Down syndrome and dementia you can contact Downs Syndrome Association helpline on 0333 1212 300 or visit their website www.downs-syndrome.org.uk.
Genes Associated With Autosomal Dominant Ad
Although several hundred families carry one of the following mutations, they account for less than 1% of cases.
AD1: Amyloid precursor protein
Inheritance and clinical features
In the 1980s, Kang and colleagues purified both plaque and vascular amyloid deposits and isolated their 40-residue constituent peptide , which subsequently led to the cloning of the APP type I integral membrane glycoprotein from which A is proteolytically derived. The APP gene was then mapped to chromosome 21q, which accounted for the observation that patients with Down syndrome develop amyloid deposits and the neuropathological features of AD when in their 40s.,
Since then, over 32 different APP missense mutations have been identified in 85 families. Interestingly, most of these mutations are located at the secretase cleavage sites or the APP transmembrane domain on exons 16 and 17 . Information regarding APP mutations are available in the NCBI database and the Alzheimer Disease Mutation Database . Mutations within APP account for 10% to15% of early-onset familial AD ,, appear to be family specific, and do not occur within the majority of sporadic cases with AD. The majority of these EOFAD mutations are in or adjacent to the A peptide sequence , the major component of amyloid plaques , Most cases containing APP mutations have an age of onset in the mid-40s and -50s.
Gene location and structure
Gene function and expression
Genetic variation
AD3: Presenilin 1
Inheritance and clinical features
Recommended Reading: Familial Dementia
Risk Factors For Alzheimer’s Disease
Early-onset AD is infrequent, making up approximately 10% of AD cases seen in clinical practice. Most cases of early-onset AD are familial they have been linked to specific identifiable mutations and have an autosomal-dominant pattern of inheritance with nearly full penetrance. Mutations in the Alzheimer precursor protein gene on chromosome 21, the Presenilin-1 gene on chromosome 14, and the Presenilin-2 gene on chromosome 1 have been identified, and they account for approximately 50% of all cases of early-onset familial AD. Also, most individuals who have trisomy-21 , and therefore three APP genes instead of two, develop AD at relatively early ages, supporting the theory that the overexpression of the APP protein may be involved in causing AD.
Jose A. Soria Lopez, … Gabriel C. Léger, in, 2019