Risk To Family Memberslate
Genetic counseling for people with nonfamilial AD and their family members must be empiric and relatively nonspecific. It should be pointed out that AD is common and that the overall lifetime risk to any individual of developing dementia is approximately 1012%.
First-degree relatives of a person with AD have a cumulative lifetime risk of developing AD of about 1530%, which is typically reported as a 2025% risk., This risk is about 2.5 times that of the background risk .,
Disagreement exists as to whether the age of onset of the affected person changes the risk to first-degree relatives. One study found that early-onset AD increased the risk, whereas another study did not.
The number of additional affected family members probably increases the risk to close relatives, but the magnitude of that increase is unclear unless the pattern in the family is characteristic of autosomal-dominant inheritance. Having two, three, or more affected family members probably raises the risk to other first-degree relatives in excess of that noted above for nonfamilial cases, although the exact magnitude of the risk is not clear. Heston et al. found a 3545% risk of dementia in individuals who had a parent with AD and a sib with onset of AD before age 70 years. Jayadev et al. also report data suggesting that offspring of parents with conjugal AD had an increased risk of dementia.
Risk To Family Membersearly
Many individuals diagnosed as having early-onset AD have another affected family member, although family history is negative 40% of the time. Family history may be negative because of early death of a parent, failure to recognize the disorder in family members, or, rarely, a de novo mutation. The risk to sibs depends upon the genetic status of the affected proband’s parent. If one of the proband’s parents has a mutant allele, then the risk to the sibs of inheriting the mutant allele is 50%. Individuals with EOFAD have a 50% chance of transmitting the mutant allele to each child. The risk to other family members depends upon the status of the proband’s parents. If a parent is found to be affected, his or her family members are at risk.
How Do Genes Work
Genes are the basic building blocks that direct almost every aspect of how youâre built and how you work. Theyâre the blueprint that tells your body what color your eyes should be or if youâre likely to get some kinds of diseases.
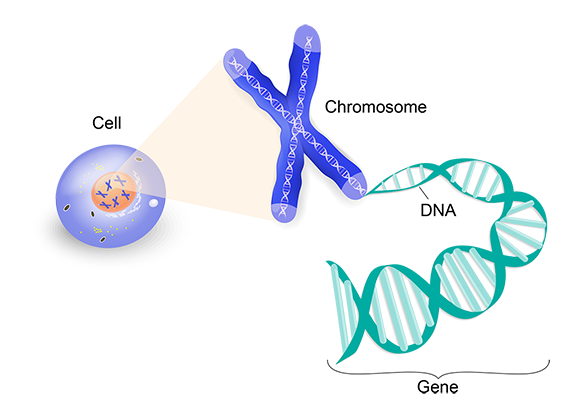
You get your genes from your parents. They come grouped in long strands of DNA called chromosomes. Every healthy person is born with 46 chromosomes in 23 pairs. Usually, you get one chromosome in each pair from each parent.
Also Check: Alzheimers Awareness Ribbons
What Is An Allele
An allele is a variant form of a gene. At conception, an embryo receives an allele of a gene from the mother and an allele of a gene from the father. This combination of alleles is what determines genetic characteristics such as the color of a persons eyes or hair. The APOE gene is located on chromosome number 19 and has three common alleles, including:
- APOE-e2: a rare allele that is thought to provide some protection against Alzheimers
- APOE-e3: an allele that is thought to be neutral as far as the risk of Alzheimers disease is concerned, this is the most common allele of the APOE gene
- APOE-e4: the third allele, which has been identified as increasing a persons risk of getting late-onset Alzheimers. A person can have zero, one, or two APOE4 alleles .
Its important to note that not everyone with the APOE4 allele will necessarily get Alzheimers. This may be because there are many factors involvedin addition to the genetic link to the diseasesuch as environmental and lifestyle factors.
Therefore, a person who inherits the APOE4 allele of the gene is not assured of getting Alzheimers disease. On the other hand, many people who get diagnosed with AD, do not have the APOE4 form of the gene.
Rsfmri Processing And Functional Connectivity Of Neural Circuits
The data of rsfMRI were preprocessed using SPM and BRANT software , which resulted in an additional exclusion of seven scans with excess motion . After preprocessing, a seed-based approach was performed to calculate the functional connectivity . Mean rsfMRI signals were extracted from each of the ROIs separately by averaging the time course signals of all voxels within the ROI. Pearsons correlation coefficients were computed between caudate, putamen, and rMFG and between the hippocampus and PCC. The correlation coefficients of each pair of regions were transformed to z values by Fishers Z transformation process to make it in accordance with Gaussian distribution . Then z values of each pair of regions of each individual, representing FC between two ROIs, were used for subsequent group comparisons.
Don’t Miss: What Color Ribbon Is Alzheimer’s
The Maddening Saga Of How An Alzheimers Cabal Thwarted Progress Toward A Cure For Decades
Other scientists werent so sure. The main doubt: This patient, like everyone, has tens of thousands of other rare variants, any one of which might be why she did not develop early-onset Alzheimers as her PSEN1 mutation should have caused.
There are thousands of variants in our genome, said Nikolaos Robakis of the Icahn School of Medicine at Mount Sinai, who discovered one of the first mutations for early-onset Alzheimers. So, from the get-go, its unlikely that this is the one that let the woman escape what would have otherwise been her genetic fate.
One reason for doubt: Having one copy of the Christchurch variant rather than two has apparently no benefit, Stanfords Greicius pointed out. Four of the seven also have the early-onset Alzheimers mutation, and all developed the disease.
It would have been most convincing to show that while two copies of the Christchurch variant move the age of onset from early 40s to early 70s, one copy had a middling effect, moving the age to the early 50s, he said. But there was no dose effect. That, agreed Robakis, is evidence against the claim that this rare form of APOE3 acts as an anti-Alzheimers talisman.
But even skeptics agreed on one thing: The role of APOE in Alzheimers is vastly understudied. Remedying that could be the Colombian womans most valuable contribution to Alzheimers research.
How Genes Influence Alzheimers
Genes literally control every function in each of the human bodys cells. Some genes determine the characteristics of the body, such as a persons eye or hair color. Others make a person more likely to get a disease.
Several genes have been identified that are linked to Alzheimer’s disease. Some of these genes can increase the risk of getting Alzheimers . Other genes, most of which are rare, guarantee that a person will develop a disease. These are called deterministic genes.
You May Like: Alzheimer’s Color Ribbon
Other Genes Linked With Late
According to Mayo Clinic, several other genes in addition to the APOE4 gene have been linked with an increased risk of late-onset Alzheimers. These include:
- ABCA7: the exact way it is involved in an increased risk of AD is not well known, but this gene is thought to play a role in how the body utilizes cholesterol
- CLU: plays a role in helping to clear beta-amyloid from the brain. The bodys normal ability to clear amyloid is vital to the prevention of Alzheimers.
- CR1: this gene produces a deficiency of protein, which may contribute to inflammation of the brain
- PICALM: this gene is involved in the method that neurons communicate with each other, promoting healthy brain cell functioning and effective formation of memories
- PLD3: the role of this gene is not well understood, but it has been linked with a significant increase in the risk of AD
- TREM2: this gene plays a role in regulating the brains response to inflammation. Variants of TREM2 are thought to increase the risk of AD.
- SORL1: variations of this gene on chromosome 11 may be linked with Alzheimer’s
Cause Of Alzheimer’s Disease Traced To Mutation In Common Enzyme
Researchers from Tokyo Metropolitan University have discovered a new mechanism by which clumps of tau protein are created in the brain, killing brain cells and causing Alzheimer’s disease. A specific mutation to an enzyme called MARK4 changed the properties of tau, usually an important part of the skeletal structure of cells, making it more likely to aggregate and more insoluble. Getting to grips with mechanisms like this may lead to breakthrough treatments.
Alzheimer’s disease is a life-changing, debilitating condition, affecting tens of millions of people worldwide. According to the World Health Organization, it is the most common cause of senile dementia, with numbers worldwide expected to double every 20 years if left unchecked.
Alzheimer’s is said to be caused by the build-up of tangled clumps of a protein called “tau” in brain cells. These sticky aggregates cause neurons to die, leading to impairment in memory and motor functions. It is not yet clear how and why tau builds up in the brain cells of Alzheimer’s patients. Understanding the cause and mechanism behind this unwanted clumping would open up the way to new treatments and ways to prevent the disease.
MARK4 has also been found to cause a wide range of other diseases that involve the aggregation and buildup of other proteins. So the team’s insights into tau protein buildup may lead to new treatments and preventative measures for an even wider variety of neurodegenerative conditions.
Explore further
You May Like: Bob Knight Dementia
What Is A Gene Variant
The same gene can differ between individuals these are known as gene variants. These can help to explain why people are different to each other for instance, why one person has blue eyes and another person has brown eyes.
If a gene variant increases a person’s risk of developing a disease, it is known as a ‘risk variant’ for that disease.
Genetic And Hereditary Risk Factors For Alzheimer’s Disease
Alzheimers disease is an incurable condition, involving the loss of memory and cognitive skills. As the incidence of Alzheimers continues to rise, so too does the push for medical science to discover the cause of the disease. Is it genetic? If so, what are the hereditary risk factors of Alzheimers disease?
Also Check: What Color Ribbon Is Alzheimer’s
The Genetics Of Alzheimer’s Disease And Frontotemporal Disorders
The Genetics of Alzheimer’s Disease
Most cases of Alzheimers disease-type dementia result from a mix of genetic, environmental and lifestyle factors. Overall, 65 year olds have a 10 percent risk of developing symptoms. In other words, 1 in 10 people over age 65 will develop it, and those who reach 85 or older, the rate rises to 1 in 3. That level of risk varies in subtle ways across individuals. About 20 gene variants are known to confer low levels of increased risk for late-onset Alzheimers disease, while others endow resilience.
Some cases of Alzheimer’s disease, and other neurodegenerative diseases such as frontotemporal dementia, are caused by a genetic mutation, or permanent change in one or more specific genes. About 3% of Alzheimer cases are familial, or caused by single mutations inherited from a parent. Familial Alzheimer’s disease usually onsets before age 65. Read on to learn about the genetic differences between early- and late-onset Alzheimer’s disease, and frontotemporal degeneration .
Identifying genetic variants may help researchers find the most effective ways to treat or prevent diseases such as Alzheimer’s or frontotemporal degeneration in an individual. This approach, called precision medicine, takes into account individual variability in genes, environment, and lifestyle for each person.
Late-Onset Alzheimers Disease Genetics
* Adapted from the National Institute on Aging: Alzheimer’s Education and Referral Center
More information sources:
Dementia Caused By A Complex Disease
Nearly all cases of dementia are the result of a complex disease. In these cases, genes may increase the risk of developing dementia, but they dont cause it directly.
What causes complex disease dementias?
When dementia is the result of a complex disease, it is likely to be caused by a combination of risk factors. These include:
- non-genetic factors for example, members of the same family may all smoke or have an unhealthy diet, which are both risk factors for dementia.
- genetic factors a person may inherit the same dementia risk variants as other members of their family. This could include variants in genes such as APOE.
These factors are often shared by members of the same family. This is why many people have some family history of dementia for example, they may have a parent who developed dementia in their 90s and a brother who developed dementia in his 70s. This is not the same as familial dementia.
Is it possible to directly inherit dementia when it’s caused by a complex disease?
No. People who are related to each other by birth are more likely to have the same risk variants as each other. However, risk variants for dementia do not directly cause dementia . This means that its not possible to directly inherit dementia through risk variants.
However, a person who has dementia risk variants is at higher risk of developing dementia than someone who does not have risk variants. Despite this higher risk, they still might not develop dementia.
You May Like: Can Aricept Make Dementia Worse
How Genetic Changes Lead To Familial Alzheimer’s Disease
- Date:
- NIH/National Institute of Neurological Disorders and Stroke
- Summary:
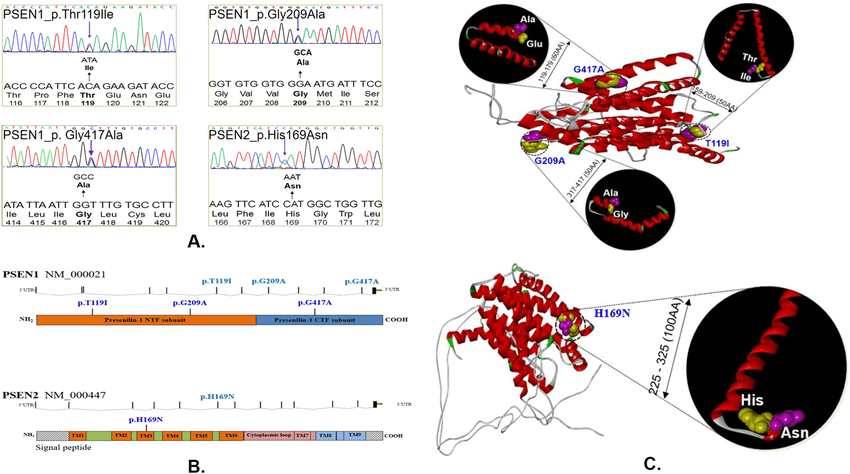
- Mutations in the presenilin-1 gene are the most common cause of inherited, early-onset forms of Alzheimer’s disease. In a new study, scientists replaced the normal mouse presenilin-1 gene with Alzheimer’s-causing forms of the human gene to discover how these genetic changes may lead to the disorder. Their surprising results may transform the way scientists design drugs that target these mutations to treat inherited or familial Alzheimer’s, a rare form of the disease.
Mutations in the presenilin-1 gene are the most common cause of inherited, early-onset forms of Alzheimer’s disease. In a new study, published in Neuron, scientists replaced the normal mouse presenilin-1 gene with Alzheimer’s-causing forms of the human gene to discover how these genetic changes may lead to the disorder. Their surprising results may transform the way scientists design drugs that target these mutations to treat inherited or familial Alzheimer’s, a rare form of the disease that affects approximately 1 percent of people with the disorder. The study was partially funded by the National Institute of Neurological Disorders and Stroke , part of the National Institutes of Health.
One of the presenilin mutations also caused impairment of memory circuits in the mouse brain and age-dependent death of neurons.
Story Source:
Neuropsychological And Clinical Correlations Of Neural Circuits
Additional file : Figure S2 shows the results of neuropsychological and clinical correlations of the imaging measures of neural circuits that showed overall group differences. In SMC, AxD of the right caudate-rMFG tract correlated significantly negatively with MMSE and MoCA total score , and significantly positively with CDR global score FC of the left caudate-rMFG tract correlated significantly positively with MMSE total score . In addition, there were significant correlations of relative volume in all the ROIs with MMSE, MoCA, and CDR global score , but not with EYO. Combining SMC and PMC, AxD of the left hippocampus-PCC tract correlated significantly negatively with MMSE and MoCA total score . In PMC, RD of right caudate-rMFG tract correlated significantly positively with MMSE total score in PS1 carriers but not in APP carriers relative volume of right putamen correlated significantly positively with MMSE total score in PS1 carriers but not in APP carriers . In the control group, none of the imaging measures showed significant correlation with neuropsychological or clinical measures .
Read Also: Does Andrea Mitchell Have Dementia
Clinical Manifestations Of Alzheimer Disease
The clinical manifestation of Alzheimer disease is dementia that typically begins with subtle and poorly recognized failure of memory and slowly becomes more severe and, eventually, incapacitating. Other common findings include confusion, poor judgment, language disturbance, agitation, withdrawal, and hallucinations. Occasionally, seizures, Parkinsonian features, increased muscle tone, myoclonus, incontinence, and mutism occur.
Death usually results from general inanition, malnutrition, and pneumonia. The typical clinical duration of the disease is 810 years, with a range from 1 to 25 years.
Gene Role Of Alzheimers Disease Can Find Presence In Brain Cells
The human brain is the center of all of the activities done by the body, whether physical or psychological. The disease Alzheimers affects the brain functions, and hence one cannot carry out routine activities also as he does.
The genes are the main responsible factors that drive this disease and make the life of one pathetic over a period which is found by a team of experts recently.
According to the study findings, Jiangsu Kim is the lead author and researcher of this study he says that being a fellow of molecular genetics as well as medical genetics research team had found the gene which is deleted by ABI3 by this occurrence, there is a significant decrease which is followed by the amyloid-beta plaque through accumulation inside the brain where it is reduced by the microglia with certain amounts formed around the plaques.
P. Michael Conneally is the lead author and professor of this study he says that this study has facilities which can provide the insights for understanding about the key functions in microglia likewise contributes towards the disease and also helps to find the new treatment and therapeutic targets
Karahan says that based on this study research, human genetics of this study are depended upon the participants, and 85000 people were reported where half of them are associated with Alzheimers disease.
The study concludes that every project has a goal which ends by identifying the druggable targets for different disease and treatments.
- TAGS
Read Also: Senility Vs Dementia Vs Alzheimer’s
T1 Image Processing And Volume Of Gray Matter Roi
T1 images were preprocessed using fslmaths and FreeSurfer software , which resulted in an additional exclusion of nine T1 scans. After preprocessing, six subcortical gray matter regions of interest including bilateral caudate, putamen, and hippocampus were obtained for each subject four cortical gray matter ROIs were obtained, including bilateral PCC and rostral middle frontal gyrus , which likely represents DLPFC based on a previous study . The volume of each ROI for each subject was calculated from the FreeSurfer software automatically. Then the relative volume of each ROI for each subject was calculated , so as to correct the effect of difference in brain size among subjects.