Tau Pathology Templated By Ad Tau Fractions In Vivo
Similar amounts of tau in O-tau, SI1-tau, SI2-tau, and HS-tau determined by immuno-dot blots with mixture of two polyclonal pan-tau antibodies were injected into the hippocampus unilaterally in 18-month-old male and female FVB mice. Mice were deeply anesthetized and transcardially perfused with saline followed by buffered 4% paraformaldehyde 3 months after tau injection. Brain was post-fixed in the same fixation buffer overnight at 4 °C and dehydrated in buffered 30% sucrose solution. Brain was then cut into 40-m serial coronal sections by using a freezing microtome, and the sections were collected in a 12-well plate containing antifreeze solution in sequence, and the free-floating sections were preserved in antifreeze solution at20 °C before immunohistochemical staining.
Interest In Tau Growing As Other Therapies Stumble
Alzheimers researchers have long debated the relative importance of amyloid plaques and tau tangles two kinds of misfolded protein clusters seen in postmortem studies of patients brains, both first identified by Alois Alzheimer in the early 20th century. For decades, the amyloid camp has dominated, leading to multiple high-profile efforts to slow Alzheimers with amyloid-targeting drugs, all with disappointing or mixed results.
Tau PET brain scans in early clinical-stage Alzheimers patients accurately predict the location of brain atrophy measured by MRI 12 years later . Amyloid PET imaging does not predict the location of either tau or future brain atrophy.
Many researchers are now taking a second look at tau protein, once dismissed as simply a tombstone marking dying cells, and investigating whether tau may in fact be an important biological driver of the disease. In contrast to amyloid, which accumulates widely across the brain, sometimes even in people with no symptoms, autopsies of Alzheimers patients have revealed that tau is concentrated precisely where brain atrophy is most severe, and in locations that help explain differences in patients symptoms .
Effect Of Hyperphosphorylated Tau On Rough Endoplasmic Reticulum And Golgi
There is approximately as much tau in the somato-dendritic compartment as in the axon . In the somato-dendritic compartment tau is associated with rough endoplasmic reticulum and Golgi apparatus . The abnormal hyperphosphorylation of tau and its accumulation in the somato-dendritic compartment in AD might have been responsible for the morphological alterations of the RER and the Golgi apparatus and the abnormal N-glycosylation of tau in AD . In AD brain abnormally hyperphosphorylated tau, in addition to forming neurofibrillary tangles, is associated with granulovacuolar changes . Overexpression of tau, which results in its hyperphosphorylation, has been found to induce fragmentation of Golgi both in neuronal cultures and in neurons in JNPL3 P301L tau transgenic mice . In P301S tau transgenic mice, which show abnormal hyperphosphorylation of tau, a selective decrease in mitochondria and RER has been observed . The chronic accumulation of the hyperphosphorylated tau as a misfolded protein in the ER could cause neurodegeneration due to protracted ER stress . Hyperphosphorylation of tau might also be involved in neurodegeneration through alterations of RER and Golgi and a consequent reduction in RER and mitochondria.
Also Check: Senility Vs Dementia Vs Alzheimer’s
Tau Aggregation In Ad And Other Tauopathies
Besides destabilizing microtubules, hyperphosphorylated tau forms intraneuronal deposits upon detachment from microtubules. This is somewhat unexpected due to the very high solubility of tau . In a self-propagating aggregation process, tau at first forms dimers and then oligomers that accumulate to form protomers. Pairs of protomers twist around each other and generate paired helical filaments that associate and grow into bundles of neurofibrillary tangles . As fetal tau isoform does not form PHF even in a highly phosphorylated state, understanding its expression and post-translational modifications may be important for future research towards the development of AD treatment and prevention .
The contribution of glial tau pathology to the pathogenesis of tauopathies is not well understood, although at least theoretically, its targeting may bring some therapeutic hope for AD. Namely, it is considered that tau pathology in glial cells disrupts the normal role of glia in supporting neuronal functioning. These include the role of astrocytes in maintaining the blood-brain barrier and modulation of synaptic function, particularly through the secretion and uptake of glutamate, as well as the myelinating function of oligodendrocytes .
How Does Alzheimers Disease Affect The Brain
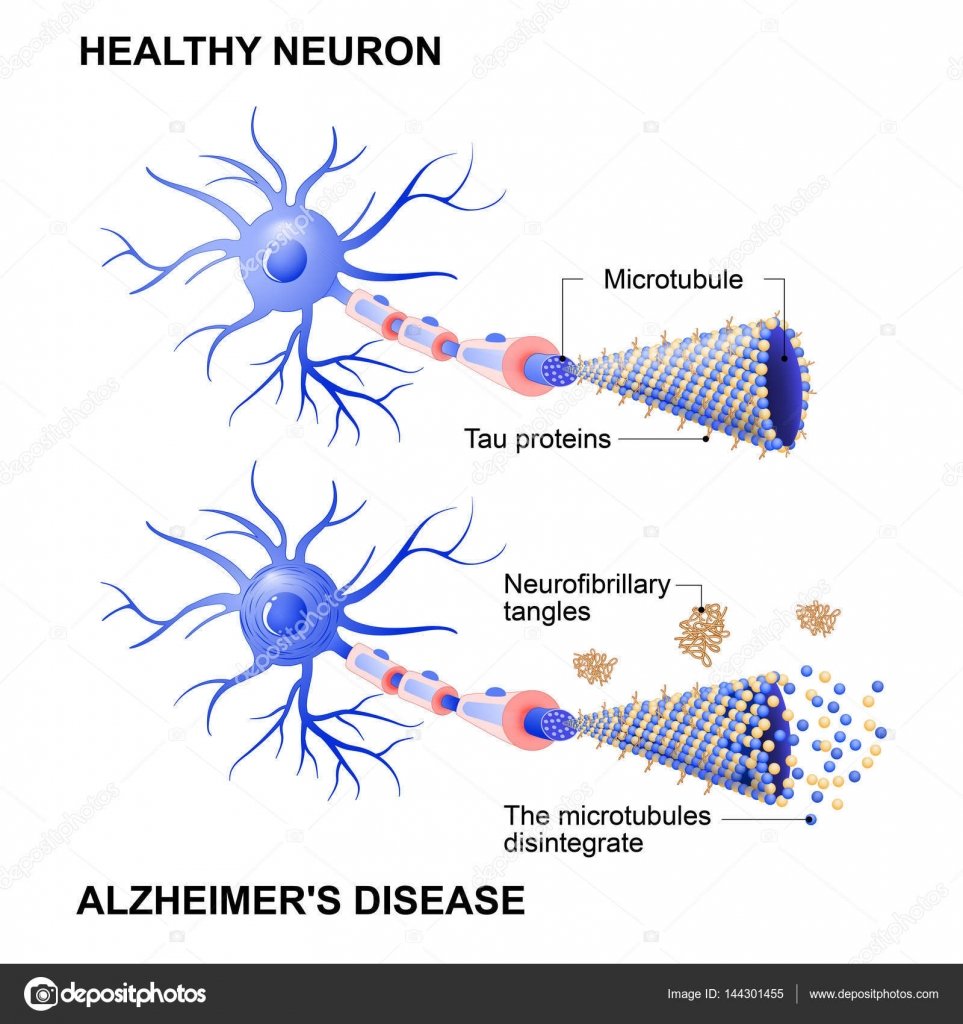
The brain typically shrinks to some degree in healthy aging but, surprisingly, does not lose neurons in large numbers. In Alzheimers disease, however, damage is widespread, as many neurons stop functioning, lose connections with other neurons, and die. Alzheimers disrupts processes vital to neurons and their networks, including communication, metabolism, and repair.
At first, Alzheimers disease typically destroys neurons and their connections in parts of the brain involved in memory, including the entorhinal cortex and hippocampus. It later affects areas in the cerebral cortex responsible for language, reasoning, and social behavior. Eventually, many other areas of the brain are damaged. Over time, a person with Alzheimers gradually loses his or her ability to live and function independently. Ultimately, the disease is fatal.
You May Like: Does Bob Knight Have Dementia
Key Biological Processes In The Brain
Most neurons have three basic parts: a cell body, multiple dendrites, and an axon.
- The cell body contains the nucleus, which houses the genetic blueprint that directs and regulates the cells activities.
- Dendrites are branch-like structures that extend from the cell body and collect information from other neurons.
- The axon is a cable-like structure at the end of the cell body opposite the dendrites and transmits messages to other neurons.
The function and survival of neurons depend on several key biological processes:
Neurons are a major player in the central nervous system, but other cell types are also key to healthy brain function. In fact, glial cells are by far the most numerous cells in the brain, outnumbering neurons by about 10 to 1. These cells, which come in various formssuch as microglia, astrocytes, and oligodendrocytessurround and support the function and healthy of neurons. For example, microglia protect neurons from physical and chemical damage and are responsible for clearing foreign substances and cellular debris from the brain. To carry out these functions, glial cells often collaborate with blood vessels in the brain. Together, glial and blood vessel cells regulate the delicate balance within the brain to ensure that it functions at its best.
Vascular Contributions To Alzheimers Disease
People with dementia seldom have only Alzheimers-related changes in their brains. Any number of vascular issuesproblems that affect blood vessels, such as beta-amyloid deposits in brain arteries, atherosclerosis , and mini-strokesmay also be at play.
Vascular problems may lead to reduced blood flow and oxygen to the brain, as well as a breakdown of the blood-brain barrier, which usually protects the brain from harmful agents while allowing in glucose and other necessary factors. In a person with Alzheimers, a faulty blood-brain barrier prevents glucose from reaching the brain and prevents the clearing away of toxic beta-amyloid and tau proteins. This results in inflammation, which adds to vascular problems in the brain. Because it appears that Alzheimers is both a cause and consequence of vascular problems in the brain, researchers are seeking interventions to disrupt this complicated and destructive cycle.
Read Also: Does Meredith Grey Have Alzheimer’s
Ad Tau Fractions Are Truncated Differentially
Tau in AD brain appears in various pools and in monomeric, oligomeric, and filamentous forms . In addition to aggregated tau, AD brain also expresses similar levels of normal tau , which is heat-stable . Aggregated tau is sarkosyl-insoluble . Normal tau and pathological tau can be separated by sedimentation. In the present study, by combining Kopkes and Guos protocols , we isolated various tau fractionsO-tau, SI1-tau, SI2-tau, and HS taufrom AD brains . Negative staining microscopy showed paired helical filaments in fraction SI1-tau, mostly straight filaments in SI2-tau, short filaments in O-tau, and non-filament in HS-tau .
It is widely believed that truncation of tau plays a critical role in tau pathogenesis . To reveal the truncation of various AD tau fractions, we isolated aggregated O-tau, SI1-tau, and SI2-tau from four AD cerebral cortices and analyzed tau protein patterns by using polyclonal and monoclonal pan-tau antibodies to various regions of the protein . HS-tau from one AD brain was used as a reference. In general, we found SDS- and -MEresistant high-molecular weight-tau in O-tau, SI1-tau, and SI2-tau detected by R134d, 92e, and 111e . Heat treatment is known to remove aggregated tau . We found no detectable HMW-tau in HS fraction . Compared with blots developed with antibodies R134d and 92e, a lesser amount of tau in SI2 fraction was detected with antibody 111e .
Fig. 2
Tau In An Oxidative Environment
Tau is susceptible to OS and OS is considered as an important factor contributing to NFT formation . NFTs are identified as sites of catalytic redox reactivity in the hippocampal tissue of AD patients . Accumulation of hyperphosphorylated tau species stimulates the production of ROS and induction of OS conditions, but OS in turn directly promotes tau hyperphosphorylation. In this vicious cycle levels of both ROS and abnormal tau increase progressively, ultimately leading to neuronal death . Moreover, OS-mediated abnormal tau phosphorylation is likely a critical deregulator of physiological tau function at the synaptic terminals and an important underlying mechanism of a synaptic failure, in addition to its role in Fyn-mediated and A-driven synaptotoxicity via excitotoxic pathways .
You May Like: Does Smelling Farts Help Prevent Dementia
Conformational Changes Required For Tau Polymerization
It is generally accepted that tau hyperphosphorylation promotes detachment from microtubules and its polymerization and aggregation. PHFs can be formed from the fragments of tau protein that lack a majority of the phosphorylation sites . Also, PHFs isolated without protease digestion are labeled by antibodies directed against phosphorylated epitopes in the N-terminal part of tau, but after removal of a fuzzy coat, the immunoreactivity is lost. This indicates that a fuzzy coat contains most of the amino acid residues that are phosphorylated . Moreover, tau phosphorylation at specific phosphorylation sites may antagonize PHF formation. Thus, phosphorylation at Ser214 and Ser262 prevents tau binding to microtubules and PHFs assembly, suggesting that phosphorylation has a protective role by preventing tau aggregation . In another study, the formation of NFTs was associated with reduced oxidative damage, leading to the conclusion that tau phosphorylation could have an antioxidative function, serving as a response intended to maximally protect neuronal structure and function against oxidative injury . As the self-assembling of the constructs corresponding to the repeat region occurs after the formation of cross-linked dimers, it is suggested that an increase in neuronal redox potential and excessive protein oxidation are mainly responsible for PHFs formation .
Phosphorylated Tau Interactome In The Human Alzheimers Disease Brain
Eleanor Drummond and Geoffrey Pires contributed equally to this work.
Correspondence may also be addressed to: Thomas Wisniewski Centre for Cognitive Neurology, Department of Neurology, New York University School of Medicine, New York, NY, USA E-mail:
Brain
Recommended Reading: Dementia Awareness Color
What Causes Tau Buildup
Scientists have long pointed to the importance of tau in AD because of evidence linking the spread of tau with disease progression. The accumulation of beta amyloid in the brain of a person with AD is largely completed at an earlier clinical stage known as mild neurocognitive disorder. However, tau accumulation continues throughout the course of the disease. Beginning in the parts of the brain called the entorhinal cortex and hippocampus, brain tau continues to accumulate as AD progresses. Recent evidence suggests that tau spreads through the brain by means of oligomer seeds that travel across a structure, called a synapse, which allows a nerve cell to pass an electrical or chemical signal to another nerve cell. The total amount of abnormal tau in the AD brain is linked to disease stage and severity.
Effect Of Tau Phosphorylation And Tau Kinases On Tau Function
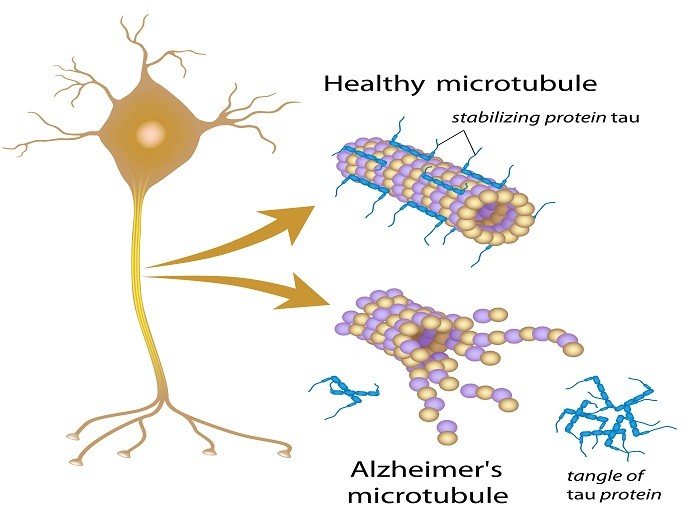
Tau is susceptible to many posttranslational modifications that modulate its function and contribute to the heterogeneity of tau isoforms present in the brain. The diversity of biological tau functions is mainly regulated by phosphorylation that attracted much attention as a modification particularly relevant for the propagation of AD pathology . In AD tau is hyperphosphorylated and is not attached to microtubules. Hyperphosphorylation, represented as an overall increase of phosphorylation at multiple residues, reduces the binding affinity of tau for microtubules and is responsible for the loss of the physiological tau functions. Hyperphosphorylation disturbs interactions between tau and microtubules and results in microtubule instability, decreased microtubule bundling, impairment of axonal transport and neuronal architecture, and ultimately cell death .
Recommended Reading: What Color Ribbon Is Alzheimer’s
Comparison Of The Phosphorylated Tau Interactome With Previously Published Studies
To place our results in the context of what is already known about proteins that are associated with tau, we compared our pTau interactome with previously published studies using a combination of systematic literature searches and data mining of previous proteomics studies. We found that 98% of the 125 proteins in our pTau interactome had been previously associated with Alzheimers disease . These previous findings confirm that the pTau interactome proteins that we identified are already known to be proteins of interest for Alzheimers disease. We were then interested to determine which pTau interactome proteins had been previously associated with tau. Tau association was designated if there was published evidence of co-localization in NFTs, interaction with tau, or if a protein was mechanistically linked to tau in a targeted study. Ninety-six of the pTau interactome proteins were associated with tau in previous studies 33 that were directly associated with pTau and 63 that were associated with tau generally, therefore, validating our results . The high degree of overlap between our results and previous studies provides additional confidence in the 29 novel pTau interacting proteins that we identified. Importantly, 26 of these novel proteins have been previously associated with Alzheimers disease, therefore, here we provide new evidence that the involvement of these proteins in Alzheimers disease likely involves interaction with tau.
Localized Proteomics Of Neurofibrillary Tangles
Sample preparation
LC-MS/MS analysis
An aliquot of each sample was loaded onto a trap column connected to an analytical column using the autosampler of an EASY-nLC 1000 HPLC . The peptides were gradient eluted directly into a Q Exactive mass spectrometer using the following gradient: in 120 min from 2 to 30% solvent B, in 10 min to 40% solvent B and to 100% solvent B in another 10 min. The Q Exactive mass spectrometer acquired high-resolution full MS spectra with a resolution of 70000, automatic gain control target of 106, with a maximum ion time of 120 ms, and scan range of 4001500 m/z. Following each full MS, 20 data-dependent high-resolution higher-energy collisional dissociation MS/MS spectra were acquired using a resolution of 17 500, AGC target of 5×104, maximum ion time of 120 ms, one microscan, 2 m/z isolation window, fixed first mass of 150 m/z, normalized collision energy of 27 and dynamic exclusion of 30 s.
LC-MS/MS data analysis
Recommended Reading: What Color Ribbon Is Alzheimer’s
Ethics Approval And Consent To Participate
All procedures involving mice were reviewed and approved by our Institutional Animal Care and Use Committee and were carried out according to guidelines of the National Institutes of Health. The use of autopsied frozen human brain tissue was in accordance with the National Institutes of Health guidelines and was exempted by the Institutional Review Board of the New York State Institute for Basic Research in Developmental Disabilities because the research does not involve intervention or interaction with the individuals nor is the information individually identifiable.
Oxidative Stress And Tau Aggregation
OS includes conditions in which an increase in the level of reactive oxygen and nitrogen species surpasses the endogenous mechanisms of antioxidant defense provided by various enzymatic and non-enzymatic antioxidants. Through the increased formation of ROS and RNS, OS induces oxidative damage of essential macromolecules that threatens neuronal structure and ultimately function .
Figure 2. Role of tau hyperphosphorylation and oxidation in tau aggregation. Oxidative stress activates Alzheimers disease -related kinases and inhibits tau dephosphorylation by PP2A, leading to tau hyperphosphorylation. OS-mediated lipid peroxidation results in toxic aldehydes that further activate tau kinases and trigger conformational changes of hyperphosphorylated tau promoting aggregation.
Also Check: Shampoos That Cause Alzheimer’s Disease
How Tau Fibrils Elongate
Huseby and colleagues started with a basic two-step model of tau aggregation. Step one consists of two tau proteins slowly binding together, and step two involves additional tau molecules attaching themselves to the two proteins.
The researchers expanded this basic model to include additional ways in which tau fibrils behave. Scientists have previously described fibrils as the tangles untangled.
The amended model predicted that the tau protein would break down into several short fibrils. However, the researchers knew that under the microscope, tau tangles reveal long fibrils, not short ones.
So, in an attempt to explain the discrepancy between what the model predicted and the microscopic reality, the researchers wondered whether shorter fibrils joined together to form long fibrils, in a similar way to hair extensions.
Further experiments in which the scientists labeled tau fibrils with fluorescent colors revealed that indeed long fibrils were made up of shorter, differently-colored fibrils that had joined at the ends.
To the authors knowledge, these findings show for the first time that tau fibrils can grow in size by adding more than just a single protein at a time. Rather, shorter fibrils can attach to each other, elongating a fibril more quickly.
Furthermore, say the researchers, the findings help elucidate how tau fibrils can grow to be hundreds of nanometers long. Also, such knowledge can lead to a new class of drugs, which could stop tau from aggregating.
The Role Of Copper In Tau
- 1Laboratory for Developmental Neuropathology, Department for Neuroscience, Croatian Institute for Brain Research, University of Zagreb Medical School, Zagreb, Croatia
- 2Nash Family Department of Neuroscience, Icahn School of Medicine at Mount Sinai, New York, NY, United States
- 3Ronald M. Loeb Center for Alzheimers Disease, Icahn School of Medicine at Mount Sinai, New York, NY, United States
- 4Friedman Brain Institute, Icahn School of Medicine at Mount Sinai, New York, NY, United States
- 5Laboratory for Protein Dynamics, Division of Molecular Medicine, Ruer Bokovi Institute, Zagreb, Croatia
- 6Department of Psychology, Catholic University of Croatia, Zagreb, Croatia
Read Also: Is Dementia Related To Alzheimer’s