The Importance Of The Study
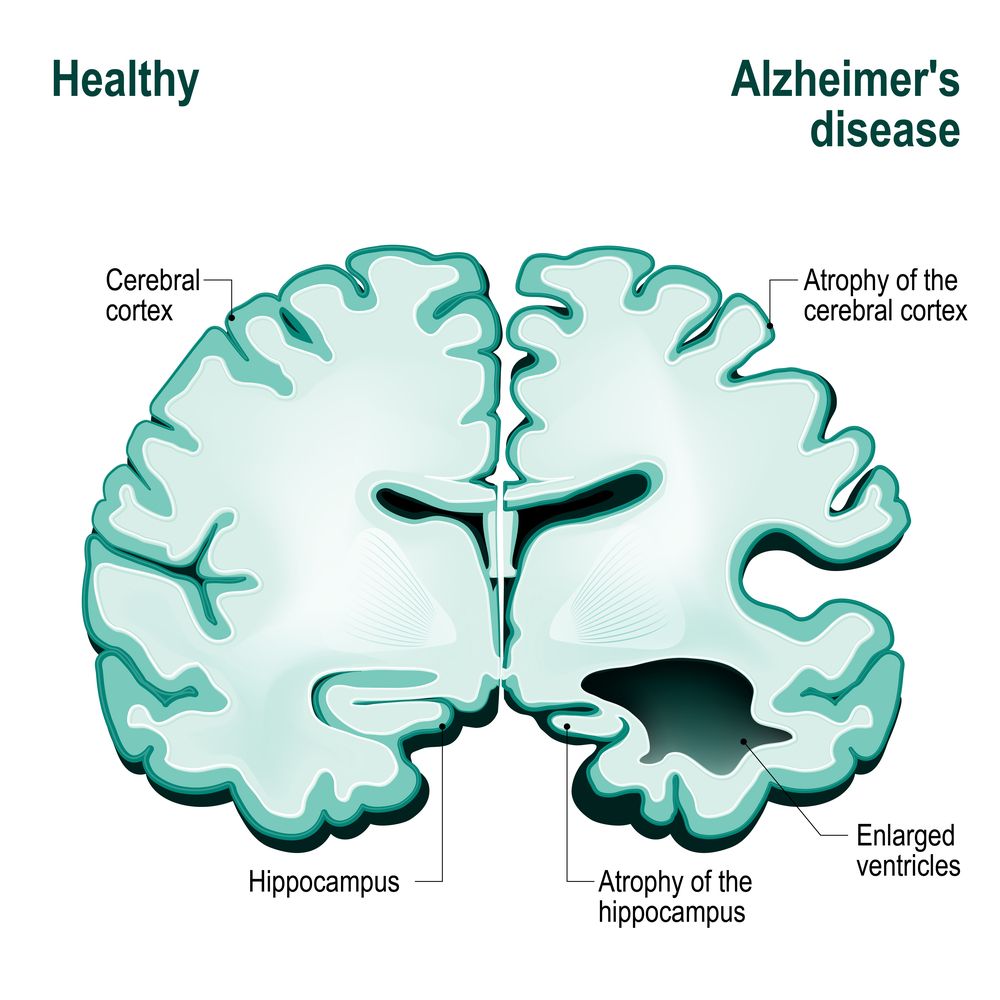
In Alzheimers disease, tau and another protein called amyloid-beta form tangles and plaques known as aggregates that cause the brain to shrink.
Amyloid deposits in the brain first, then tau aggregates begin to occur. Neuronal damage later develops, then clinical symptoms of memory loss, and finally, loss of functional independence known as dementia occurs, Dr. Sharon Sha, a clinical associate professor of neurology and neurological sciences at Stanford University in California, told Healthline.
The two proteins are thought to predate clinical symptoms by decades. Tau, specifically, because it deposits later in the disease process, may more closely align with clinical symptoms, she explained.
The researchers found that the progression of Alzheimers disease is based on the replication of these aggregates in single regions of the brain, not the spread of the aggregates from one area to another.
They say their study could help improve treatments for Alzheimers disease by targeting and stopping the replication of aggregates in the brain.
The key discovery is that stopping the replication of aggregates rather than their propagation is going to be more effective at the stages of the disease that we studied, Tuomas Knowles, PhD, co-senior author of the study and a researcher from the Department of Chemistry at Cambridge, said in a press release.
So What Is Alzheimers Disease
Alzheimers is a medical disease of the brain that gets progressively worse with time. It affects the brain and how it functions. Alzheimers is the most common form of dementia in people over the age of 65 years old. It is believed that Alzheimers develops long before the symptoms of dementia become apparent in the sufferer.
Neurochemical And Histopathological Changes In Cholinergic And Non
At postmortem, Alzheimers disease is characterised by neuronal loss and neurofibrillary tangle formation in circumscribed regions of the neocortex and hippocampus, primarily affecting pyramidal neurons and their synapses. Neurotransmitter specific subcortical nuclei that project to the cortex are also affected by neurodegenerative processes, including the cholinergic nucleus basalis of Meynert and medial septum, the serotonergic raphe nuclei, and the noradrenergic locus coeruleus.
In addition to cholinergic dysfunction, other strong correlates of dementia are the chemical and histopathological markers of excitatory amino acid releasing cortical pyramidal neurons. These neurons, considered to contribute to normal cognitive function in their own right, also seem to have a pivotal role in cholinergic function as they are cholinoceptive. Although neurochemical studies of EAA neurotransmission have failed to show profound or extensive alterations in EAA neuronal indices, this may be related to the difficulty in distinguishing the transmitter pool of aspartate and glutamate from the metabolic pool. Nevertheless, glutamate concentration was reduced by 14% in temporal lobe biopsy samples of patients with Alzheimers disease. Greater reductions were evident at postmortem in regions enriched with EAA nerve terminals.Uptake of d-aspartate, a putative marker of EAA nerve endings, is also reduced in many cortical areas in Alzheimers disease brains.
Don’t Miss: What Color Ribbon Is Alzheimer’s
Aducanumab: Right Treatment Wrong Time
As with treatments for many other human diseases, it might be that treatments such as aducanumab might only be effective if they are given early enough, before the disease has caused irreversible changes. A better understanding of the environmental and genetic factors behind Alzheimers disease combined with ever more sensitive brain imaging techniques will help doctors identify warning signs even earlier, before even minor memory loss occurs.
While screening and diagnosing people before symptoms have set in for an as-yet incurable disease, raises many ethical dilemmas, it might present a fresh window of opportunity to retest beta-amyloid drugs, such as aducanumab. Ultimately, we need to focus our research on understanding the early stages of the disease so that we can prevent Alzheimers disease before dementia takes hold.
The Quest For Prevention
As time and my own understanding progress, I think that we should be looking for a way to prevent Alzheimers disease rather than a way to cure it once it has been diagnosed. In the 100+ years since Dr. Alzheimer first described the disease, very little progress has been made toward this end, whether cure or prevention. On the other hand, our understanding of the brain has advanced markedly in the last few years. This gives me some hope that we will soon discover a way to prevent neurological diseases like Alzheimers.
Suggested articles
Read Also: Does Neil Diamond Have Alzheimer’s
Behavioural Consequences Of Cholinomimetic Drugs And Cholinergic Lesions
Many pharmacological studies have examined the effect of cholinomimetic drugs and cholinergic receptor antagonists on learning and memory tasks. The most commonly used model is based on the finding that scopolamine, a muscarinic receptor antagonist, induces amnesia in young healthy subjects comparable with that in old, untreated subjects. These deficits may be reversed by ChE inhibitors. Compounds that reverse these scopolamine induced deficits in experimental animals may be considered as potential drugs to treat cognitive impairment.
It is, however, difficult to separate reliably the effects on learning and memory processes from effects on other behavioural domains. For example, methylscopolamine is as active as scopolamine in several models of cognitive function, indicating that peripheral changes induced by these compounds indirectly influence performance in cognitive tasks. It is, therefore, very important to distinguish central versus peripheral effects of cholinminetic agents. Scopolamine induced impairment of performance may also be mediated by direct effects on sensorimotor function or motivation deficits.Further, it is likely that the scopolamine induced impairment in the performance of both experimental animals and humans in the delayed matching to position task is secondary to attentional deficits that are induced by the drug.
Who First Discovered Alzheimers Disease
Alzheimers disease was first discovered in 1907 by German Psychiatrist Dr. Alois Alzheimer.
He noticed that a female patient of his slowly lost her memory and the ability to think and talk and that she developed judgment problems.
After she died, Doctor Alois Alzheimer looked at her brain through a microscope and noticed that her brain had abnormal growths, which he named plaques and tangles.
These changes occur in the brain of everyone who develops Alzheimers disease.
You can find more information on the discovery of Alzheimers disease by Doctor Alois Alzheimer and more of the discoveries of Dr. Alois Alzheimer on Wikipedia here
Don’t Miss: Smelling Farts Dementia
Scientists Unlock Key To Progression Of Alzheimers Disease
Dave Yasvinski,
The research, , relied on a chemical kinetics approach developed at Cambridge University combined with advances in precision PET scanning to gain new insights into a disease that has previously only been understood by studying animal models or the post-mortem brain samples of patients.
The thinking had been that Alzheimers develops in a way thats similar to many cancers: The aggregates form in one region and then spread through the brain,said Georg Meisl , first author of the study from Cambridges Yusuf Hamied Department of Chemistry. But instead, we found that when Alzheimers starts there are already aggregates in multiple regions of the brain and so trying to stop the spread between regions will do little to slow the disease.
There are over 500,000 Canadiansliving with dementia today, with another 25,000 diagnosed with the progressive disease every year, according to the Alzheimer Society . Two-thirds of those diagnosed over the age of 65 are women. At the rate at which the disease is growing, it costs over $12-billion a year to care for patients an expense that underscores the importance of improving the ability to diagnose and treat the disease before it becomes too advanced for intervention.
Dave Yasvinski is a writer withHealthing.ca
The First Use Of Alzheimers Disease
Alzheimer later published his descriptions of several similar patients in 1909 and Kraepelin included Ms. Deters case in the 1910 edition of his widely respected psychiatry textbook. It was Kraepelin who named this dementia after his junior colleague.Auguste Deter was not an elderly woman at the onset of her illness, and Alzheimers disease was therefore regarded as a presenile dementia to distinguish it from the familiar senile dementia thought to result from aging-related vascular disease. Further investigation, however, showed that plaques and tangles were present in the brains of the majority of older adults with symptoms of dementia.
In the late 1960s, the British psychiatrists Tomlinson and Roth described the importance of these plaques in older adults, and in 1970 Dr. Roth questioned the meaningfulness of the age criterion that distinguished AD from senile dementia of the Alzheimers type.
Recommended Reading: What Color Ribbon Is Alzheimer’s
Health Environmental And Lifestyle Factors
Research suggests that a host of factors beyond genetics may play a role in the development and course of Alzheimers. There is a great deal of interest, for example, in the relationship between cognitive decline and vascular conditions such as heart disease, stroke, and high blood pressure, as well as conditions such as diabetes and obesity. Ongoing research will help us understand whether and how reducing risk factors for these conditions may also reduce the risk of Alzheimers.
A nutritious diet, physical activity, social engagement, and mentally stimulating pursuits have all been associated with helping people stay healthy as they age. These factors might also help reduce the risk of cognitive decline and Alzheimers. Researchers are testing some of these possibilities in clinical trials.
Young Onset And Familial Alzheimers Disease Return To The Spotlight
The past century has seen a number of transformations in the conceptualization of Alzheimers disease. The 2013 G8 dementia summit recognized Alzheimers disease as a growing global health and economic problem, requiring serious action in terms of investment in research and development of disease-modifying therapies but also that alongside the search for prevention and treatment strategies we must invest in enabling people to live well with dementiaensuring that patients and their families have access to early diagnosis and support that is all too often lacking. A hundred years on Alzheimers legacy is more relevant than everin fact the question of how tractable is this disease and how we care for those with it is perhaps one of the key challenges for the coming century.
Also Check: Alzheimer’s Disease Ribbon Color
Cause Of Alzheimers Disease Progression In Brain Discovered
Toxic protein clumps in the brain are believed to be partly responsible for Alzheimers disease, researchers believe
Clusters of toxic proteins, thought to be responsible for the cognitive decline associated with Alzheimers disease, reach different areas of the brain and accumulate there over decades, according to a new study released Friday.
The article, published in Science Advances, is the first to use human data to quantify the rate of molecular processes leading to this neurodegenerative disease, and could influence the way treatments are designed.
It also overturns the theory that clumps form in one place and trigger a chain reaction in other areas, a pattern seen in mice. Such a spread can occur, but is not the main driver, according to the researchers.
Two things made this work possible, says Georg Meisl, a chemist at Cambridge University and one of the lead authors of the article.
First the study of very detailed data from PET scans and various sets of data put together, and the mathematical models that have been developed over the past ten years .
The researchers used 400 brain samples taken after the death of people with Alzheimers and 100 PET scans from people living with the disease to track tau protein aggregation.
This and another protein called beta-amyloid build up, causing brain cell death and brain shrinkage.
Likely Cause Of Alzheimers Disease Discovered In Fat

PERTH, Australia Alzheimers disease is becoming more and more common, with an estimated six million Americans currently living with the condition. Making matters worse is the fact that there is no effective cure yet. New research out of Australia, however, is shedding light on at least one cause of Alzheimers that may open the door for a breakthrough treatment. Researchers from Curtin University report that a probable cause of Alzheimers disease is the leaking of fat-carrying particles transporting toxic proteins into the brain from blood.
Modern medicine has deciphered a great deal about Alzheimers and its origins. For example, the abnormal build up of tau proteins in the brain appears to be a significant trigger for the disease. Theres still a whole lot we dont know, though, which is why these new findings are so noteworthy. Study authors discovered this possible cause of Alzheimers, dubbed the blood-to-brain pathway, in experiments conducted on mouse models.
While we previously knew that the hallmark feature of people living with Alzheimers disease was the progressive accumulation of toxic protein deposits within the brain called beta-amyloid, researchers did not know where the amyloid originated from, or why it deposited in the brain, lead investigator and Curtin Health Innovation Research Institute Director Professor John Mamo says in a university release.
You May Like: Senile Vs Dementia
Does Alzheimers Start In The Liver
In earlier research, the Curtin team found that beta-amyloid forms outside the brain from lipoproteins. In this study, they tested the blood-to-brain pathway hypothesis by genetically engineering mice capable of producing human amyloid-only livers that create lipoproteins.
As we predicted, the study found that mouse models producing lipoprotein-amyloid in the liver suffered inflammation in the brain, accelerated brain cell death and memory loss, Prof. Mamo explains. While further studies are now needed, this finding shows the abundance of these toxic protein deposits in the blood could potentially be addressed through a persons diet and some drugs that could specifically target lipoprotein amyloid, therefore reducing their risk or slowing the progression of Alzheimers disease.
Alzheimers WA Chairman Adjunct Professor Warren Harding believes this work may one day change the lives of millions.
Having universities like Curtin working with the pharmaceutical industry is important if we are to tackle this devastating disease, Harding concludes. In Australia, approximately 250 people are diagnosed with dementia daily, adding to the staggering half a million Australians who are already living with dementia. Without significant medical advances like the breakthrough Professor Mamos team has made, it is estimated that the number of Australians living with dementia will exceed one million by 2058. This has a significant impact on families, carers and communities.
Theres A Way To Stop This Process
Along with finding what causes tau tangles in the brain to get worse, researchers also discovered a way to reverse it too. Study authors say, even in patients who deal with growing amounts of cell damage and oxidative stress, the expression of more antioxidants counters these effects.
Antioxidants appear to clear away tau proteins, resulting in lower tau levels and less risk of neurodegenerative diseases forming. The team hopes their findings are the first step in forming a treatment for tau toxicity and removing harmful plaques. Currently, there is no definitive cure for dementia.
Read Also: Dementia Awareness Ribbon Color
In English Spanish French
On November 3, 1906, a clinical psychiatrist and neuroanatomist, Alois Alzheimer, reported “A peculiar severe disease process of the cerebral cortex” to the 37th Meeting of South-West German Psychiatrists in Tubingen, He described a 50-year-old woman whom he had followed from her admission for paranoia, progressive sleep and memory disturbance, aggression, and confusion, until her death 5 years later. His report noted distinctive plaques and neurofibrillary tangles in the brain histology. It excited little interest despite an enthusiastic response from Kraepelin, who promptly included “Alzheimer’s disease” in the 8th edition of his text Psychiatrie in 1910. Alzheimer published three further cases in 1909 and a “plaque-only” variant in 1911, which reexamination of the original specimens in 1993 showed to be a different stage of the same process, Alzheimer died in 1915, aged 51, soon after gaining the chair of psychiatry in Breslau, and long before his name became a household word.
Keywords: Alois Alzheimer Alzheimer’s disease German psychiatry case of Auguste D. case of Josef R history.
Leading Art Historian 75 With Alzheimer’s Disease Is Found Dead By Police In Woods Of Upstate New York State Park Five Days After Being Reported Missing
- Richard Kendall, 75, was a UK-born renowned art historian and impressionist movement expert specializing in the works of French painter Edgar Degas
- He was last seen walking near his home in Nyack, New York, on November 3 and was found dead in ‘a heavily wooded area’ on November 8, according to police
- Kendall curated exhibits at major museums around the world, including the Metropolitan Museum of Art in NYC and the Tate Modern in London
- He authored many books, including Picasso Looks At Degas and Degas And The Dance with his wife Jill DeVonyar, a fellow art historian and Degas expert
Don’t Miss: Senility Vs Dementia Vs Alzheimer’s
Delay The Onset Of The Disease
Maybe if we can improve them a bit, we can significantly delay the onset of critical illness.
Alzheimers disease is classified according to Braaks stages, and scientists have found that it takes about 35 years to go from stage 3, where mild symptoms appear, to stage 6, the most advanced.
The aggregates are growing exponentially, which explains why the disease takes so long to develop, and why the condition of people tends to worsen rapidly, according to Georg Meisl.
Hopefully this study and others will help guide the development of future treatments targeting the tau protein, so that they have a better chance of slowing the disease and helping people with dementia, said in a statement. press release Sara Imarisio, from Alzheimers Research UK.
Alzheimers: Travel therapy to soothe the sick
LONG FORMAT. The Bel-Air retirement home, in Tournon-dAgenais in Lot-et-Garonne, has a train travel simulator that helps improve the quality of life of patients with Alzheimers disease. An innovative therapeutic tool available to only a few establishments.
Fighting Alzheimers with a cup of coffee
Drinking coffee has an impact on our cognitive abilities. Caffeine is responsible for these effects. Chemical derivatives could even be used as drugs against Alzeihmers disease.