A Clearance Through Intracellular And Extracellular Enzymatic Degradation
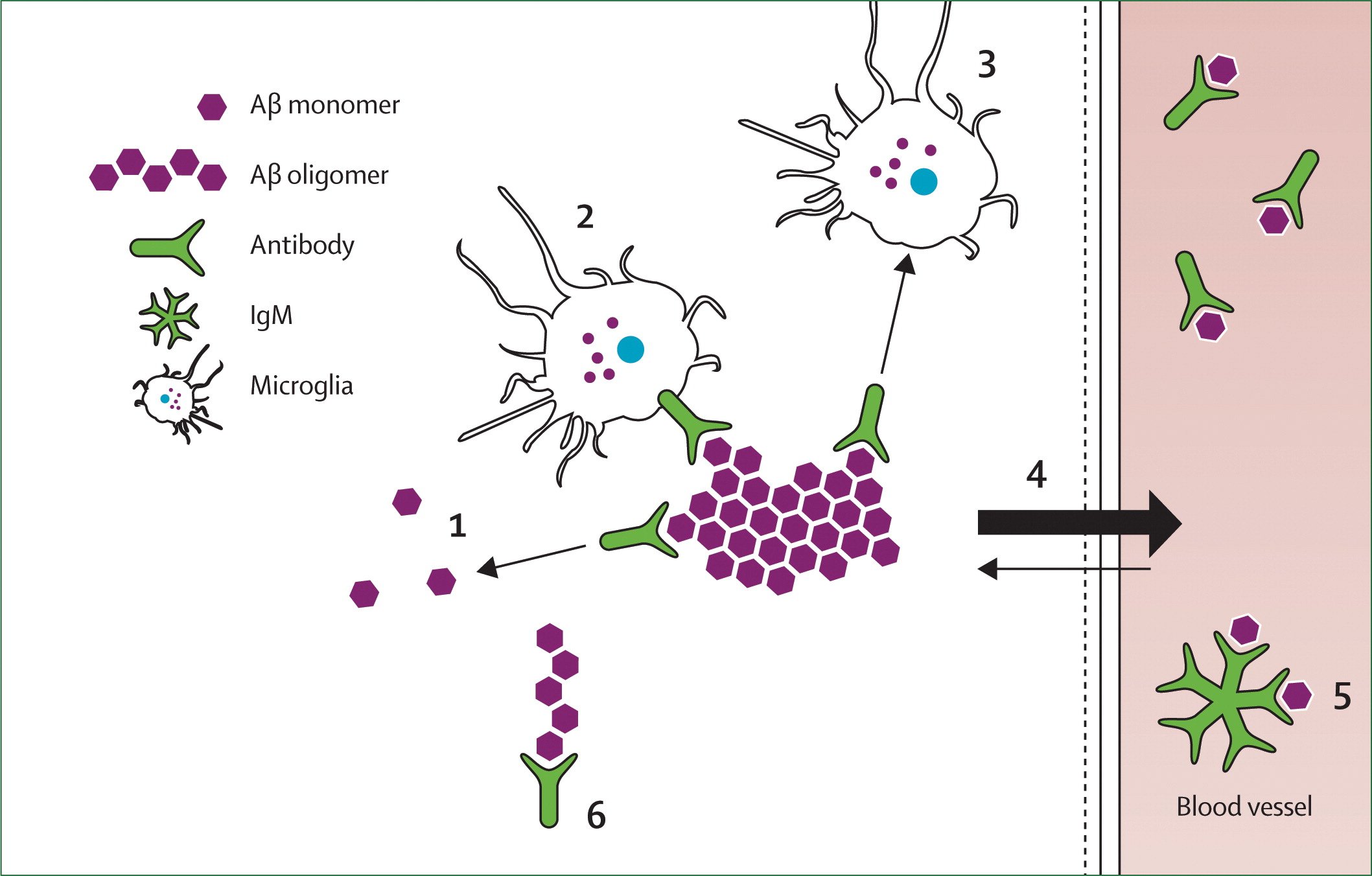
There is preliminary evidence showing that intracellular A can be degraded by proteasomes and A-degrading enzymes via the ubiquitin-proteasome pathway in neurons and the extracellular neprilysinmediated pathway, respectively . Mouse models of AD indicate that components of the ADE system can be impaired and that A can inhibit the proteasome, through cross-pathways influences, including a lysosomal cathepsin B-mediated mechanism . Therefore, experimental data suggest the existence of a self-reinforcing detrimental protein homeostasis cycle .
The ADE encompasses the zinc metalloendopeptidase -1 and -2, angiotensin-converting enzyme ), thiol-dependent metalloendopeptdiase ), serine proteases , cystein proteases , matrix metalloproteinase , Kallikrein-Related Peptidase 7 and others . Many genes identified through GWASes and established as risk factors for AD are linked to A degradation through the endosomal-lysosomal system or ubiquitin-proteasome pathway .
Fluorides Effect On The Brain
Unlike many foreign substances, fluoride can cross the blood-brain barrier giving it freedom to accumulate in the brain and cause widespread damage.5,6,7
Through fluorides extremely reactive and toxic nature it carries a well-established prooxidant effect in cells.8 This is the main mechanism that fluoride deploys to cause damage to the central nervous system.
However, fluorides effect on the brain does not stop there.
These are just observations of the effects that took place in the brain of subjects exposed to fluoride.
There are many ways these effects can contribute or cause Alzheimers but its hard to ignore the effect of damage to the hippocampus.
The hippocampus is well known for its role in learning and memory, a key feature Alzheimers patients lack.9
But lets find out what is fluoride and specifically its link to Alzheimers.
Study Solves Mystery Of How Amyloid Beta Forms In Brain Nerve Cells
- Date:
- Massachusetts General Hospital
- Summary:
- A key protein must be cut to create amyloid beta. Groundbreaking work identifies the ‘lipid raft’ in nerve cells where this process occurs, which could offer new targets for drug therapy.
In a major breakthrough, researchers at Massachusetts General Hospital have discovered how amyloid beta — the neurotoxin believed to be at the root of Alzheimer’s disease — forms in axons and related structures that connect neurons in the brain, where it causes the most damage. Their findings, published in Cell Reports, could serve as a guidepost for developing new therapies to prevent the onset of this devastating neurological disease.
The new investigation revealed that palAPP is stabilized and prepared for cleavage by beta secretase in special lipid rafts within the neuron known as mitochondria-associated endoplasmic reticulum membranes . “We showed for the first time not only that the MAM is where palAPP is processed by beta secretase to make Abeta, but that this happens exclusively in axons and neuronal processes where Abeta does most of its damage,” says Bhattacharyya, lead author of the Cell Reports paper. This role for MAMs was previously unknown, though earlier research indicated that they are increased in number and activity in the brains of people with Alzheimer’s disease.
Explore the latest scientific research on sleep and dreams in this free online course from New Scientistâ
You May Like: Does Diet Coke Cause Alzheimer’s
Understanding The Complexity Of App Biology Independently Of A Is Important To Understanding Ad Pathogenesis
APP synthesis, trafficking and cleavage are complex and highly regulated processes . It is important to recognise that familial AD APP and presenilin mutations may not only impact A production, but also the production of the other peptides produced from APP including sAPP, sAPP, p3 and AICD, as well as the relative levels of full-length APP. Interestingly, overexpression of AICD can cause an AD-like phenotype , whilst increased cleavage of sAPP is associated with familial Danish Dementia with similar aetiology to AD . Furthermore lowered levels of neurotrophic sAPP are seen in AD, and mutations which inhibit the -secretase enzyme ADAM10, which liberates sAPP from its precursor, are found in the promoter region and coding sequence of some individuals with AD . Depletion of sAPP by inhibition of ADAM10 trafficking can bring about sporadic AD phenotypes , corroborating an independent role for APP cleavage products other than A in bringing about disease phenotypes.
Superresolution Imaging Of Amyloid
Next, we sought to characterize a cellular function of cholesterol loading . To establish the movement of APP to each of its processing enzymes, -, -, and -secretases, directly in cellular membranes, we imaged N2a cells with dSTORM. APP was previously found in both cluster- and noncluster-like fractions of DRMs . DRMs are similar to GM1 lipid clusters. However, imaging APP is important, since the amount of cluster-like association in DRMs could easily be affected by the detergent concentration used for preparation. We labeled GM1 domains with fluorescent CTxB and the amyloid proteins with Cy3b-labeled, fluorescent antibodies and determined cluster localization by pair correlation using density-based spatial clustering of applications with noise of two-color dSTORM images .
shows that APP, -secretase, and -secretase are associated with GM1 clusters , consistent with experiments in DRMs.
In contrast, -secretase is not associated with GM1 domains . The pair correlation ranged from 10 to 15 for APP, -secretase, and -secretase, while the pair correlation for -secretase was < 3, a value typical for little or no pair correlation, a result also consistent with previous experiments in DRMs .
Don’t Miss: Smelling Farts Dementia
Drugs Approved By The Fda
To date, only a total of five drugs developed to improve the symptoms of Alzheimer’s disease have been approved by the FDA. It is important to note that a new drug, Namzaric was approved in 2014. The five drugs function by two different mechanisms. One is cholinesterase inhibition, which delays Alzheimer’s disease by blocking hydrolysis of the critical neurotransmitter acetylcholine. This category of drugs includes donepezil ,, approved in 1996 rivastigmine ,, approved in 2000 and galantamine , approved in 2001. The other one is memantine , approved in 2003, a non-competitive N-methyl-D-aspartate channel blocker that reduces the activity of the neurotransmitter glutamate, which plays an important role in learning and memory by binding to the NMDA receptor. Memantine can inhibit the prolonged influx of Ca2+ ions, particularly from extrasynaptic receptors, that forms the basis of neuronal excitotoxicity. It is an option for the management of patients with moderate to severe Alzheimer’s disease. Namzaric is a combination of the two drugs to reduce the levels of both acetylcholine and glutamate .
Table 2 Summary of approved drugs for the treatment of Alzheimer’s disease.
MicroRNAs
Synaptic Formation And Repair
The most-substantiated role for APP is in synaptic formation and repair its is during neuronal and after neural injury. Roles in , , and have been proposed and supported by as-yet limited research. In particular, similarities in post-translational processing have invited comparisons to the signaling role of the surface protein .
APP are viable and have relatively minor effects including impaired long-term potentiation and memory loss without general neuron loss. On the other hand, transgenic mice with upregulated APP expression have also been reported to show impaired long-term potentiation.
The logical inference is that because A accumulates excessively in Alzheimer’s disease its precursor, APP, would be elevated as well. However, neuronal cell bodies contain less APP as a function of their proximity to amyloid plaques. The data indicate that this deficit in APP results from a decline in production rather than an increase in catalysis. Loss of a neuron’s APP may affect physiological deficits that contribute to dementia.
Also Check: Dementia Ribbon Color
A Clearance Via Brain Interstitial Fluid Bulk
The perivascular drainage pathway has a significant role in ISF bulk-flow clearance of A . Failure of perivascular drainage of A and increased A deposition in arterial walls has two detrimental downstream effects: microbleeds due to rupture of A-laden arteries, namely cerebral amyloid angiopathy that has high comorbidity with AD, and AD itself where the failure of elimination of ISF, A, and other soluble metabolites from the brain alters homeostasis and the neuronal micro-environment, and is associated with synaptic decline and cognitive-functional impairment.
The glymphatic system was proposed as a CSF-ISF exchange system in absence of direct lymphatic access to the brain and with astrocytes as cellular links between brain parenchyma and the perivascular pathway, with eventual solute transport to the cerebrovenous network and meningeal lymphatic vessels . While there is limited knowledge of the anatomy and function of the glymphatic system in humans, mouse models of aging and AD show that the glymphatic pathways represent a vital clearance system for driving the removal of soluble A from the interstitium . Several other glymphatic-related factors with implications for AD include expression and localization of aquaporin 4 channels on astrocytic endfeet, arterial pulsation, and diurnal glymphatic cycles corresponding to sleep-awake rhythms .
The A Deposition Paradox
How wonderful that we have met with a paradox. Now we have some hope of making progress.
Niels Bohr
A deposition occurs in cognitively normal individuals
Up to 40% of non-demented elderly can reach some level of neuropathological criteria for AD . A positive correlation also exists between A deposits and increases in phosphorylated tau, the other major cerebral histological inclusion in AD protein, in cognitively normal patients . In one study only 17% of cognitively normal elderly patients had few or no degenerative brain changes , and in neuroimaging amyloid-PET studies 10-30% of cognitively normal individuals have amyloid-positive scans . Around 50% of people over the age of 85 have AD , rising to 77.5% of centenarians who meet criteria for mild confusion or severe dementia based on cognitive testing . This could be interpreted to suggest that amyloid deposition is predominantly associated with normal aging and is not a disease per se.
In sum, the distribution of amyloid deposits in the brain does not correlate well with neuropathology, loss of neural function from specific brain areas, or cognitive impairment. A conclusion that plaque is not the cause of LOAD provides one possible explanation for A vaccination trials not improving patient outcome, even when plaque was removed.
Why are plaques present in cognitively normal individuals?
Several valid interpretations of AD data could equally explain the A deposition paradox:
The rise of the soluble A hypothesis
Read Also: Life Expectancy Dementia Age 80
What Happens To The Brain In Alzheimer’s Disease
The healthy human brain contains tens of billions of neuronsspecialized cells that process and transmit information via electrical and chemical signals. They send messages between different parts of the brain, and from the brain to the muscles and organs of the body. Alzheimers disease disrupts this communication among neurons, resulting in loss of function and cell death.
Tau Posttranslational Modification In Ipscneurons
Tau phosphorylation is also developmentally regulated, with high phosphorylation in early development thought to be related to the requirement for dynamic remodelling of the microtubule network . Hyperphosphorylation of tau is observed across the tauopathies, and accurate determination of the sites of phosphorylation and the relative stoichiometry in control versus disease is important in inferring the presence of pathological tau in preclinical models. The majority of studies using iPSCs to model tauopathy have examined phosphorylation at specific sites using phosphospecific antibodies, although a few studies have directly identified sites of phosphorylation using mass spectrometry.
You May Like: Neil Diamond Alzheimer’s
The Tau Hypothesis Of Ad
The concept of hyperphosphorylated tau being a primary mediator of AD, like amyloid, has a long history, which continues to grow . Much interest still exists in where tau sits in the pathogenesis of AD . In our view, AD is sufficiently diverse that it is conceivable that the role of tau, and where it sits in AD pathology, could vary among individuals. If tau is a primary activator of disease in some cases, it is imperative that the reported harmlessness of phosphorylated tau to neurons during mammalian hibernation be discussed in AD research circles. Furthermore, hyperphosphorylated tau can be considered another histological sign of cytokine activity .
A Monomer Homeostasis Is Essential For Synaptic Function
A biogenesis and turnover is an essential physiological process conserved in neurons of vertebrates. The central point of difference between the old A Cascade hypothesis and the new Dysfunction hypothesis is the claim for a physiological role of the A monomer in non-diseased individuals. There is multiple evidence that A monomer plays an important role in synaptic activity regulation . The group of Copani has suggested that the loss-of-function hypothesis for the role of A in neurons should be taken into consideration . The homeostasis of synaptic A monomer levels is critical and extremely complex. The complexity of A monomer physiology is due to the interplay of a metastable hydrophobic peptide with membranes, lipids, and vesicles. Quantifying metastable A monomer concentrations in living organisms is challenging since IDPs can adopt their structure upon extraction. While there has been some success with a combination of synchrotron-based Fourier transform infrared micro-spectroscopy and non-denaturing gel electrophoresis to characterize intermediate A species in APP TG mice , it remains extremely difficult to determine nascent A monomer concentrations in vivo.
It can only be postulated that A pathophysiology is assumed to start, when the formation and metabolism process is disturbed. Table 1 summarizes key papers supporting the physiological A role.
Table 1. Key findings to the essential role of A monomer in physiological synaptic processes.
Recommended Reading: Smelling Farts Cure Cancer
Alcohol Cytokines And The Peripheral Immune System
Alcohol affects both cytokines and the peripheral immune system. Alcohol-induced neuroinflammation includes increased brain levels of pro-inflammatory cytokines interleukin 1 and tumour necrosis factor alpha . Reduced anti-inflammatory cytokine IL-10 have been observed in response to chronic high alcohol administration in short-term pre-clinical models. Notably, these persisted for several days while peripheral circulating plasma cytokines were undetectable, favouring a pro-inflammatory milieu in the brain .
In pre-clinical models, the impact of ethanol may have dichotomous effects on the peripheral immune system depending on the concentrationhaving a suppressive effect at high and stimulatory effect at low concentration .
Clinical studies show that serum pro-inflammatory cytokines have been increased in patients with chronic alcoholism . Increased pro-inflammatory and anti-inflammatory cytokines early in abstinence have been described , as has increased pro-inflammatory cytokine production by dendritic cells from alcohol-dependent participants. In addition to the pathways above, peripheral inflammation is highly likely due to alcohol-related increase in gut bacteria translocation. These peripheral cytokines are known to affect microglial phenotypes .
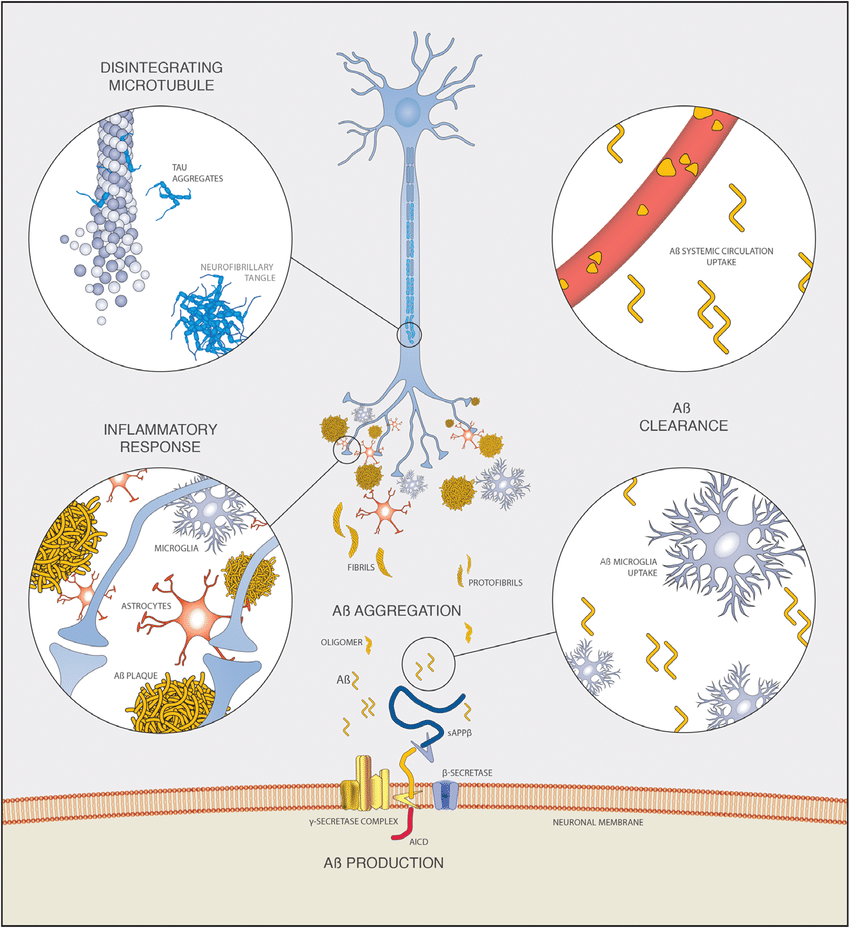
A Generation From App
Canonical A is produced when APP is sequentially cleaved by secretase and secretase. However, there exists a spectrum of A species that are produced when APP is cleaved by alternate enzymes, leading to truncations at both N and C termini . As such, Takami et al used a targeted LCMS/MS approach to show that secretase has Cterminal truncating activity as the longest forms of A are successively processed through parallel tripeptide cleavage pathways to produce the most abundant fragments of A .
Amyloidbeta sequence, cleavage sites and insights from mass spectrometry. The amino acid sequence of A, with residues labile to posttranslational modifications highlighted: Greenphosphorylation and dityrosine/nitrotyrosine/nitration of serine and tyrosine residues. Goldpyroglutamate modification of glutamic acid residues. Redracemization/isomerization of aspartic acid residues. Arrows show the sites of secretase cleavage of A. Information learned from iPSC models subject to mass spectrometry analysis. Predominant forms of A in different stages of neuronal differentiation . The finding that N terminal cleavage can direct C terminal endoproteolysis . The antiamyloidogenic role of BACE2 in reducing relative amounts of amyloidogenic species in Down’s syndrome neurons . The specificity of avermectins, increasing the processivity of secretase while leaving short species of A unchanged
Also Check: What Color Is Alzheimer’s Ribbon
More Tips For Avoiding Damage To Your Brain
To keep iron levels down, make sure theres none added to your multivitamin . Men and postmenopausal women dont usually need to take iron supplements. In fact, taking iron can increase your risk for some cancers. Dont cook acidic foods, such as tomatoes or lemon juice, in iron pots. Acids leach iron out of these pots.
One trick to lower iron absorption is to drink some black tea with a meal high in iron . Black tea contains a chemical that deactivates iron. So a lunch of a salad with a glass of iced tea is a perfect way to eat a meal high in antioxidants and low in iron.
Linking Neuroinflammation In Alzheimer’s Disease And Alcohol Misuse
Proposed TLR4 signalling in microglia. Both alcohol and fA bind to TLR4, resulting in neuroinflammation via the MyD88-dependent and MyD88-independent responses.
There is an emerging consensus on the role of the innate immune system in both AD and in the effects of alcohol. As described, both AD and alcohol cause neuroinflammation via the action of TLRs, in particular TLR4 and TLR2. The action of microglial cells are suggested to be key to this process. Downstream inflammatory cascades are activated, and release of certain cytokines and chemokines are thought to lead to neuronal death. Modulation of the effects of A via TLR appears central to this. Alcohol in stimulating TLR4 may modulate this process and a positive feedback loop may be established via TNF .
You May Like: Sporadic Dementia
Tau Turnover In Ipscneurons
Stable isotope labelling kinetics allows for the measurement of protein production and turnover rates using labelling with heavy essential amino acids , followed by mass spectrometry to distinguish labelled from unlabelled peptides. Heavy leucine will be incorporated into newly synthesized proteins during the labelling phase, and the relative amount of labelled protein will reduce over time because of degradation. A thorough review of SILK in neurodegeneration is provided here . Sato and colleagues used this approach to understand tau turnover in iPSCneurons .
Interestingly, diseaseassociated tau species appear to have an increased turnover. For example 4R tau had a shorter halflife than 3R tau, and several tau peptides containing phosphorylation residues were shown to have a faster turnover than their nonphosphorylated counterparts , suggesting differential proteostasis of specific tau species. It is also intriguing to note that the halflife of tau in iPSCneurons is much shorter than in human participants . This indicates developmental changes in tau proteostasis and is concordant with the idea that protective mechanisms against protein aggregation may become less efficient during ageing.