Alzheimers Disease Prion Diseases Share Similarities
Dr. John Collinge
Fatal brain disorders called prion diseases might provide insight into the cause of Alzheimers disease, said Dr. John Collinge at a recent Wednesday Afternoon Lecture Series talk in Lipsett Amphitheater.
There are more and more similarities between what we see in prion disease and what were seeing in Alzheimers disease, said Collinge, professor of neurology at University College London , director of the United Kingdom Medical Council prion unit, director of the UCL Institute of Prion Diseases, director of the National Hospital for Neurology and Neurosurgerys United Kingdom National Prion Clinic and visiting professor of neurology at Harvard Medical School.
A prion is an abnormal form of a naturally occurring protein that has the ability to catalyze the conversion of the normal form into the same abnormal form. Prions have been implicated in several diseases known as transmissible spongiform encephalopathies, which affect animals and humans, Collinge said. Over time, prions accumulate and damage the brain.
Symptoms of prion disease include changes in behavior, rapid onset of dementia and movement problems. Prion diseases are usually rapidly progressive and always fatal, according to the CDC. Most cases of prion disease occur randomly, Collinge explained. Certain host mutations can increase the risk of getting the disease.
Weve seen patients with incubation periods up to 60 years, which is quite extraordinary, Collinge said.
The Spreading Confusion: Rethinking Alzheimers Disease
Proteins are molecular machines. They perform an incredible diversity of tasks that enable all living cells to function. Like any machine, a protein must be properly assembled in order to carry out its specific task, and if something goes awry, the cellular consequences can be dire. Take Alzheimers disease for example. Alzheimers disease is the most common neurodegenerative disorder, affecting over 5 million people in the United States alone . Alzheimers is a progressive disorder that manifests clinically as a loss of memory, personality traits, and the ability to carry out daily tasks . Scientists do not fully understand why or how this disease develops, but it is known that in the brains of Alzheimers patients, some of these protein machines malfunction, forming large, toxic clumps. A major goal of Alzheimers research has been to uncover the causes and understand the consequences of this protein clumping. A recent study published in the journal Nature draws an interesting parallel between Alzheimers and prion diseases , another group of neurodegenerative diseases caused by protein clumping. Prion diseases are unique for one important reason: they are infectious. This new study raises the important question: could Alzheimers too be infectious?
Scjd Induces H In Hu Brnaggs
Untreated Hu BrnAggs produced large bands of tau . Exposure to a normal brain homogenate produced smaller bands and 2 very weak bands . Exposure to sCJD produced bands between 40 and 50 kDa and strong bands of phosphorylated tau .
In another preliminary study, we made BrnAggs from transgenic mice expressing human mutated tau. These mouse tau BrnAggs spontaneously form small numbers of neurons containing H in nerve cell bodies and dendrites. When Mo BrnAggs were exposed to an A42-containing homogenate from a transgenic 2576 mouse, the number of neurons containing abnormal tau increased approximately 2-fold. In contrast, when tau BrnAggs were exposed to a brain homogenate from a wild-type mouse infected with Rocky Mountain Laboratories scrapie prions containing PrP Sc , the number of neurons and their processes containing abnormal tau increased 10-fold. These results were reproduced in triplicate. The findings argue that exposure to A42 and PrP Sc increases the levels of H inclusions in tau BrnAggs. We did not use normal age-matched human brain homogenate controls in these studies, which is why we classified them as preliminary .
Also Check: Alzheimer’s Paranoia Medication
Prion Disease Spreads To Wildlife
The biggest difference between Alzheimers disease, mad cow disease and chronic wasting disease is the species under attack. All are forms of prion disease, which is an unstoppable neurodegenerative disease. Thanks to misinformation and mismanagement, prion disease is spreading from infected humans to other mammals and from infected mammals to humans. It is now the fastest-growing cause of death in the world .
Since the prion pathogen migrates, mutates and multiplies, few cases are identical in terms of presenting symptoms or disease progression. CWD is essentially the same as mad cow disease, Alzheimers disease, Creutzfeldt-Jakob disease and other deadly prion diseases. As such, we prefer to cut out the smoke and mirrors and call it what it isa prion disease epidemic. Some cases strike and kill extremely fast, while others take longer. All cases, however, are fatal and transmissible.
H In The Mtl Of Priond And Ad Cases
Unlike controls in which abnormal tau staining was focal, H staining of MTL in CJD-only , GSS-only , CJD-AD , and AD-only was continuous or multifocal and extended from the entorhinal cortex into the transentorhinal cortex . Cases of CJD-only and CJD-AD showed strong positive and continuous staining in layers 1 and 2 of the entorhinal cortex and transentorhinal cortex . Lower-intensity H staining was also present in neuropil threads and neurons in deeper layers of the entorhinal cortex in CJD-only cases. Cases of CJD-AD showed intense immunostaining in the superficial layers and less intense immunostaining in the deeper layers of the en-torhinal cortex . In AD-only cases, many H threads and nerve cell bodies were stained . The most intense staining occurred in the GSS-only case . In all of the groups, the presubiculum showed little H staining. The hippocampal CA1 region and subiculum were intensely stained in CJD-AD and AD-only cases, whereas the GSS-only case had staining in portions of the CA1 region and the subiculum.
You May Like: Prevagen For Dementia
Prp Sc And A42 Plaque Distribution
In most patients, PrP Sc was distributed either in all layers of the cortex or in deep layers 4 to 6. Rarely was the distribution of PrP Sc found in superficial layers 1 to 3. For cases in which PrP Sc was deposited in deeper cortical layers, such as an 81-year-old woman who was near the end of the age distribution for the Prion-AD group , A42 plaques tended to be distributed in superficial layers 1 to 3 . In another case of a 53-year-old woman who was at the beginning of the Prion-AD age distribution, PrP Sc staining was confined to layers 5 and 6 in the lateral frontal cortex and A42 plaques were located in layer 1 . In the parietal cortex of the same patient, PrP Sc accumulated in all 6 cortical layers , and A42-positive plaques were found in cortical layers 1 to 5 . This suggests that A42 plaques are first formed in layer 1 of the cerebral cortex and subsequently progress in order from layers 2 through 6.
Sample Collection And Processing
Patient material was obtained after the approval of local ethics committees at the University Medical Center, Goettingen. Frontal cortex samples from patients with spAD , rpAD , dementia with Lewy bodies , age-matched non-demented controls and other rapid dementias including small vessel disease , rapidly progressive dementia with Lewy bodies , and dementia with frontotemporal lobar degeneration cases were provided by the brain bank of the Institute of Neuropathology and the biobank of the Hospital Clinic-IDIBAPS, Spain, according to their biomedical study legislation . Frontal cortex samples from patients with sporadic Creutzfeldt-Jakob disease subtypes were obtained from the Department of Neurology at the University Medical Center, Göttingen, Germany. The rpAD patients met the current selection criteria for rpAD . These inclusion criteria are as follows:
Initial classification as prion diseases based on clinical features
Presence of typical AD pathological features, i.e., higher Braak stages
Post diagnostic survival time shorter than four years
Exclusion of other forms of rapid progressive dementias and copathologies e.g. prion diseases, extensive Lewy body pathology, vascular damage or tumors based on postmortem neuropathological examination
Absence of a family history suggestive of familial AD
Don’t Miss: Can Aricept Make Memory Worse
Overlap Of Priond And Az
For CERAD staging of AD, we examined multiple brain regions, including the hippocampus, entorhinal cortex, transentorhinal cortex, inferior temporal cortex, lateral frontal cortex areas 45 and 46, lateral parietal cortex areas 39 and 40, cingulate cortex area 24, and medial occipital cortex . Of the 266 PrionD cases reviewed for the study, 46 cases showed both PrP Sc and extracellular A42 plaques and were designated as the Prion-AD group . This group included 41 sCJD cases, 4 fCJD cases, and 1 GSS case. In 18 of 46 Prion-AD cases, a sufficient number of A42 plaques were detected in all cerebral cortical regions and in some subcortical regions to qualify for definite AD by CERAD criteria. In the remaining 28 Prion-AD cases, the number of A42 plaques was insufficient to make a diagnosis of definite AD therefore, we designated them as incipient AD. The 220 cases with PrP Sc but no detectable A42 plaques were designated as Prion-only cases.
Tau Prion Levels Strongly Correlate To Longevity
Importantly, higher levels of prion-like amyloid beta and tau were found in those with early onset of Alzheimers who died at an earlier age, with tau buildup showing the strongest correlation. Compared to a patient who died of Alzheimers at the age of 90, a patient who died at 40 had on average 32 times higher amounts of tau prions in their brain. As noted by UCSF:28
Alzheimers disease is currently defined based on the presence of toxic protein aggregations in the brain known as amyloid plaques and tau tangles, accompanied by cognitive decline and dementia.
But attempts to treat the disease by clearing out these inert proteins have been unsuccessful. The new evidence that active Aß and tau prions could be driving the disease could lead researchers to explore new therapies that focus on prions directly.
Senior author Dr. Stanley Prusiner, director of the UCSF Institute for Neurodegenerative Diseases, commented on the results:29
I believe this shows beyond a shadow of a doubt that amyloid beta and tau are both prions, and that Alzheimers disease is a double-prion disorder in which these two rogue proteins together ruin the brain.
The fact that prion levels also appear linked to patient longevity should change how we reckon about the way forward for developing treatments for the disease.
One of the studys lead authors, Carlo Condello, Ph.D., assistant professor of neurology in the Institute for Neurodegenerative Diseases, added:30
Also Check: Dementia Ribbon Tattoo
What Are The Symptoms
Symptoms of Creutzfeldt-Jakob disease and the order in which they appear can vary significantly from person to person. Common symptoms include:
- Rapidly worsening confusion, disorientation, and problems with memory, thinking, planning and judgment
- Depression
- Agitation, apathy and mood swings
- Difficulty walking
- Muscle stiffness, twitches and involuntary jerky movements.
Expression Of G2l2 And Associated Proteins
Among the proteins identified as HDP interactors in rpAD, G2L2 was selected for further functional verifications, given its essential role in cytoskeletal integrity, i.e. in actin-mediated microtubule growth, and the possible relevance of the latter for AD pathology. G2L2 is reported for its role in co-alignment of actin filaments and microtubules, where it acts by cross-linking the two structures. This actin/microtubule cross-linking is assisted by the end-binding protein-1 at the plus end of the microtubules . In order to study G2L2 in the sample cohorts in more detail, proteins functionally associated with the physiology of G2L2, including EB-1, tubulin and actin, were also studied. Expression of these proteins in frontal cortex homogenates was assessed by western blot analysis. We found no significant differences in the levels of G2L2 between the AD subtypes and controls. Likewise, no significant differences were detected between the groups for EB-1, -tubulin, and -actin .
Fig. 3
You May Like: Alz Ribbon
What Makes Amyloid Infectious
A study31 published in the journal Prion in 2014 sought to determine why certain proteins prone to form amyloids have the capacity to infect their neighbors. Here, too, the author referred to Alzheimers as a prion disease, specifically with reference to the amyloid plaques formed:
The conformational diseases, linked to protein aggregation into amyloid conformations, range from non-infectious neurodegenerative disorders, such as Alzheimers disease , to highly infectious ones, such as human transmissible spongiform encephalopathies . They are commonly known as prion diseases.
But, since all amyloids could be considered prions it is necessary to find an underlying cause of the different capacity to infect that each of the proteins prone to form amyloids has.
As proposed here, both the intrinsic cytotoxicity and the number of nuclei of aggregation per cell could be key factors in this transmission capacity of each amyloid.32
The author goes on to state that while amyloids are universal and share certain internal structural characteristics, prions represent only a tiny drop in the amyloid ocean. In order for an amyloid to become a prion, something has to occur causing the aggregation process to become self-perpetuating and infectious.
Recent findings in the field have shown that the number of nuclei of aggregation could be a factor that affects the infection capacity of amyloid-prone proteins, just as their intrinsic cytotoxicity does.
A And Tau Prion Activity Linked To Alzheimers Patients Longevity
The most remarkable finding of the new study may be the discovery that the self-propagating prion forms of tau and Aß are most infectious in the brains of Alzheimers patients who died at a young age from inherited, genetically driven forms of the disease, but much less prevalent in patients who died at a more advanced age.
In particular, when compared to measurements of overall tau buildup which is known to increase with age in Alzheimers brains the researchers found a remarkable exponential in the relative abundance of the prion forms of tau with age. When the researchers plotted their data, they saw an extremely strong correlation between tau prions and patients age at death: relative to overall tau levels, the quantity of tau prions in the brain of a patient who died at age 40 were on average 32 times higher than in a patient who died at 90.
I still remember where I was sitting and what time of day it was when I first saw this data over a year ago, said co-author and leading neurodegeneration researcher William Seeley, MD, a professor of neurology who directs the UCSF Neurodegenerative Disease Brain Bank, which provided tissue used in the study. Ive very rarely, if ever, seen this kind of correlation in human biological data. Now the job is to find out what the correlation means.
Co-lead author Carlo Condello, PhD, an assistant professor of neurology. Image courtesy Carlo Condello.
Read Also: Alzheimer Ribbon
Alzheimers Disease Linked To Prions
For a number of years now, researchers have theorized and found evidence suggesting Alzheimers disease may in fact be a type of prion-based disease,7,8,9 capable of being contracted via meat10 and transmitted via certain invasive medical procedures.11
Researchers have noted that Alzheimers behaves like a slow moving version of CJD,12,13,14 and according to one paper,15Prions are considered a subclass of amyloids in which protein aggregation becomes self-perpetuating and infectious. As reported by Scientific American:16
Between 1958 and 1985, a number of individuals with small stature received shots of human growth hormone extracted from the pituitary glands of cadavers Some of these samples were contaminated with prions that caused certain patients to develop Creutzfeldt-Jakob disease , a rare and fatal brain disorder.
Treatments stopped once these reports came to light, but by that time an estimated 30,000 people had already received the injections. As of 2012, researchers have identified 450 cases of CJD worldwide that are the result of these growth hormone injections and other medical procedures, including neurosurgery and transplants.
Research Now Shows That Amyloid Plaques And Tau Tangles From Diseased Brains Can Infect Healthy Brain Tissue The Infectivity Of Brain Tissue Is Just The Tip Of The Iceberg
Prions invade the entire body, but they do their damage in the brain. Prions are in the bodily fluids and cell tissue of victims, including blood, saliva, mucus, urine and feces. Common household items become biohazards, including utensils and dishes. A cough or sneeze sends prions into the air. Prions are a real-world version of Pandoras Box. Prions are unstoppable. Prion disease is always fatal. This is an extremely important issue that demands the truth from government and industryespecially the medical world.
Prions migrate, mutate, multiply and kill with unparalleled efficiency. Containing prions and preventing their spread is the key to avoiding a public health disaster. Unfortunately, the floodgates have been wide open for years.
Prion disease also is known as transmissible spongiform encephalopathy . The operative word is transmissible. Prusiner claims that all forms of TSE are caused by infectious prions. TSE is a spectrum disease that varies in severity and symptoms. The diagnosis depends on which region of the brain is impacted first and by what prion mutation. Few cases are identical in terms of symptoms and diagnoses.
Prions are such a formidable threat that the U.S. governmentenacted the Bioterrorism Preparedness and Response Act of 2002, which includeda provision to halt research on prions in all but two laboratories.
Read Also: Senile Dementia Treatments
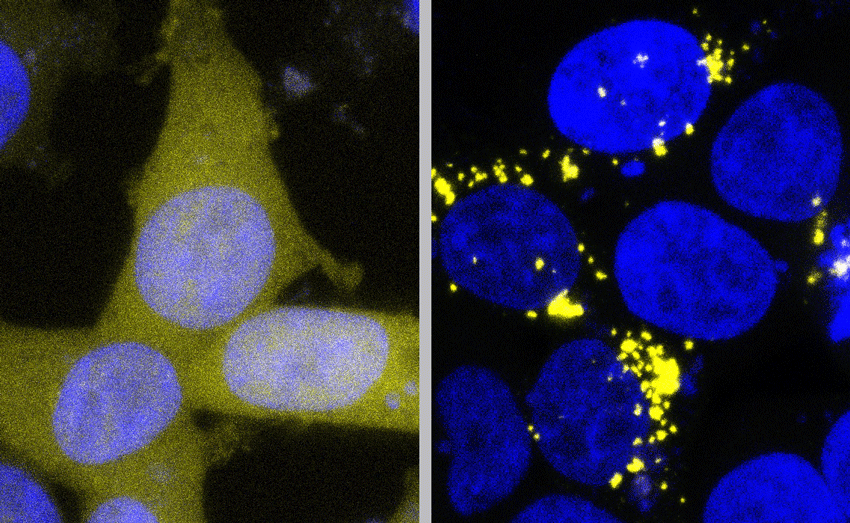
Laboratory Bioassays Reveal A And Tau Prions In Human Postmortem Brain Samples
In the new study, the researchers combined two recently developed laboratory tests to rapidly measure prions in human tissue samples: a new Aß detection system developed in the Prusiner lab and a tau prion assay previously developed by Marc Diamond, PhD, a former UCSF faculty member who is now director of the Center for Alzheimers and Neurodegenerative Diseases at UT Southwestern Medical Center.
Unlike earlier animal models that could take months to reveal the slow spread of Aß and/or tau prions, these cell-based assays measure infectious prion levels in just three days, enabling the researchers to effectively quantify for the first time the levels of both tau and Aß prions in processed extracts from post-mortem brain samples. In the new study, they applied the technique to autopsied brain tissue from over 100 individuals who had died of Alzheimers disease and other forms of neurodegeneration, which was collected from repositories in the United States, Europe, and Australia.