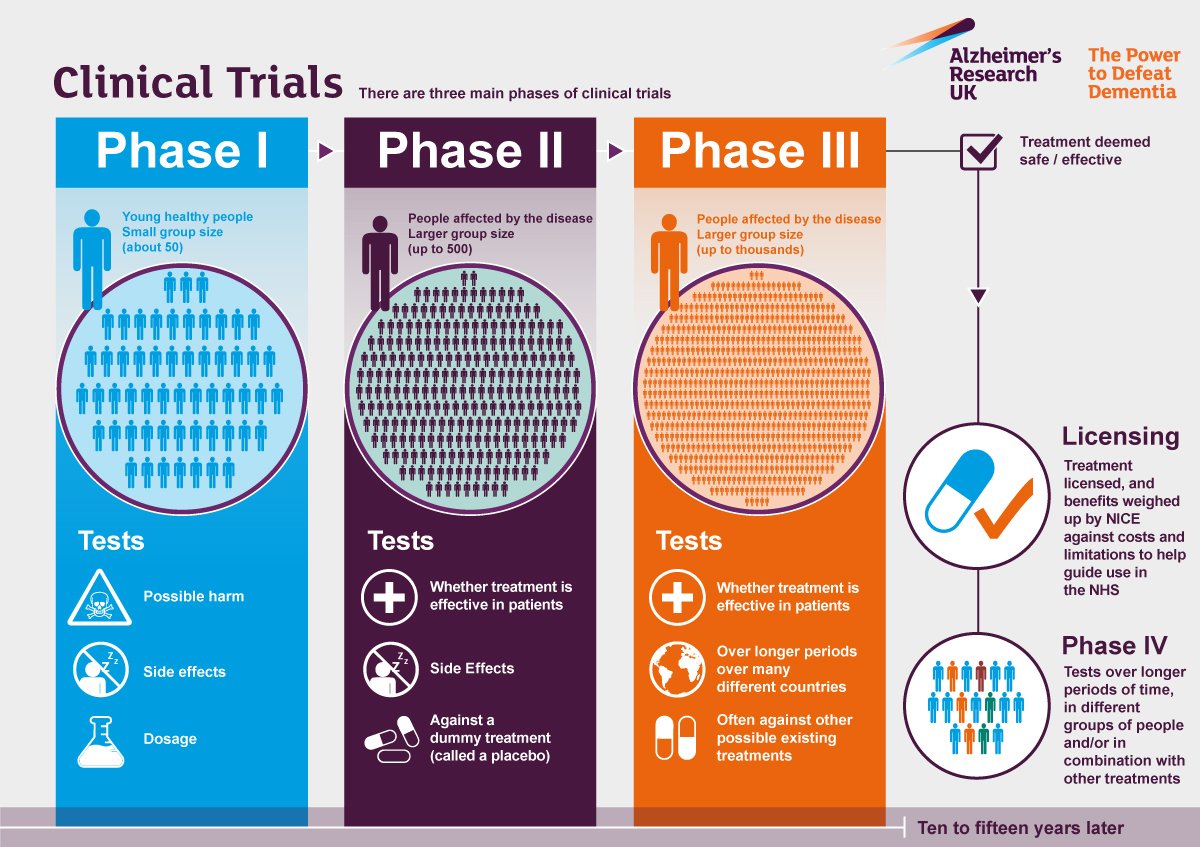
Ramping Up The Immune System
Bapineuzumab, the first widely-tested passive immunotherapy, was withdrawn from development due to limited effectiveness and concern about adverse effects including tiny brain bleeds. Testing of solanezumab as a treatment for mild dementia was deemed unsuccessful although it is still being tested as a preventive agent in high-risk individuals. Several more recent monoclonal antibodies have failed in their clinical trials, but hope was recently revived for the success of one of them. Aducanumabs data failed a futility analysis in March of 2019 but subsequent data analysis including additional data was encouraging enough to support an FDA application for treatment of Alzheimers disease. The FDAs decision is anticipated late in 2020. If aducanumab is approved, additional immunotherapeutic agents are all the more likely to be submitted.
Though these injections are all passive immunotherapies, they vary significantly in their mechanism of attack on beta-amyloid. Variously, they attack amyloid-beta or plaques, in the peripheral circulation or in the brain.
The newest immunotherapy players represent a re-emergence of the active vaccine approach which, if successful, could be less costly and more lasting in its effectiveness. One experimental vaccine, called CAD106, induces immunity to amyloid-beta without exciting an autoimmune response.
First Drug That Can Slow Alzheimer’s Dementia
Health editor, BBC News online
A US drug company says it has created the first therapy that could slow Alzheimer’s disease, and it is now ready to bring it to market.
Currently, there are no drugs that can do this – existing ones only help with symptoms.
Biogen says it will soon seek regulatory approval in the US for the “groundbreaking” drug, called aducanumab.
It plans to file the paperwork in early 2020 and has its sights on Europe too.
Approval processes could take a year or two. If successful, the company aims to initially offer the drug to patients previously enrolled in clinical studies of the drug.
The announcement is somewhat surprising because the company had discontinued work on the drug in March 2019, after disappointing trial results.
But the company says a new analysis of a larger dataset of the same studies shows that higher doses of aducanumab can provide a significant benefit to patients with early Alzheimer’s, slowing their clinical decline so they preserve more of their memory and every day living skills – things that the disease usually robs.
Early Stage Smaller Trials
After initial trials showed that Aducanumab was safe to use in humans, a further trial called PRIME assessed if the drug could slow the progression of Alzheimers disease in people who were in the early stages of the disease. This included people who had mild cognitive impairment or early-stage dementia.
The PRIME trial started in 2012. Biogen, the pharmaceutical company who developed the drug, reported that treatment led to a reduction in amyloid levels in the brain. The company said the drug appeared to slow the rate of cognitive decline for people with mild Alzheimers disease receiving the drug.
Also Check: What Does A Brain Look Like With Dementia
Inhaling Insulin And Deactivating Gum Disease Bacteria Could Benefit Memory
by Cheryl Platzman Weinstock, AARP, July 18, 2019
While there was no major breakthrough announced among the drug news saved for the final day of the Alzheimer’s Association International Conference 2019, there were a couple of novel approaches inhaling insulin to banish memory loss, for one with at least some positive results behind them.
These small if promising clinical trial results helped part, at least a bit, the dark research cloud that had hung over the conference after it was announced on the eve of the major yearly get-together of Alzheimer’s disease experts that two clinical trials of Alzheimer’s drugs were met with failure.
That news had stirred debate on the viability of finding a cure by targeting amyloid beta in particular, as the failed drugs had been designed to do. While most researchers accept that understanding how and why the neuron-destroying protein builds up in the brain is an important part of the Alzheimer’s riddle, some now say that as a drug target, amyloid beta likely appears too far along in the disease progression to make halting it an effective treatment goal.
Today’s clinical trial results, still in the early stages of review, are related to new approaches that have little to do with amyloid beta.
His company’s drug deactivates the action of the bacterium. It has been shown to be safe and well-tolerated in early trials, Detke says.
Boost Your Brain Health With Staying Sharp
How The Discovery Of An Alzheimers Tipping Point Could Improve Drug Trials
A Q& A with UCSF neurologist William Seeley, MD
Researchers have known for many years that Alzheimers disease involves two proteins called amyloid beta and tau, and that interaction between them drives the spread of neurodegeneration. What hasnt been clear is where and how those interactions occur over time as disease progresses. In a study published April 19 in the journal Neuron, neurologist William Seeley, MD, and his colleagues at UC San Francisco and in South Korea identified two key moments in the natural history of the disease.
The new findings may point to a window of opportunity for treatment with amyloid-lowering drugs like the controversial Adulhelm and similar medications currently in the pipeline. Seeley took some time to tell us about the importance of the recent discovery.
Don’t Miss: How Long Does Each Stage Of Lewy Body Dementia Last
Who Is A Candidate For Aducanumab
Based upon the clinical trials in which it was studied, the drug can be potentially considered for use in individuals with early-stage Alzheimers disease. This means individuals who have Alzheimers in its mild cognitive impairment or mild dementia stage. Individuals in this early stage of Alzheimers may be able to function normally, or they may require a little bit of help with complicated activities like paying bills, grocery shopping, preparing meals, or balancing their checkbook. Individuals who need help with dressing, bathing, or other basic activities would be beyond the early stage and the drug would not be indicated for them.
Note that not everyone with mild cognitive impairment or mild dementia has Alzheimers disease. There are other causes of these problems as well. To determine if Alzheimers is present, you need to get either an amyloid PET scan or a lumbar puncture to be sure you have the amyloid plaques of Alzheimers. Currently, a lumbar puncture is generally covered by insurance, but an amyloid PET scan is not.
Selecting More Drugs For Trial
MND-SMART is designed to run continuously for years to come to test treatments that may slow, stop or reverse the progression of MND. To help select future drugs we will trial, we will repeat the process of reviewing published information from MND and neurodegenerative disease studies at regular intervals to include the most recent published information.
We want to keep improving this drug selection process and so are developing new computer programmes to make compiling and reviewing published studies more efficient. We are also working on gathering further information on how we can test the potential of shortlisted drugs. One way we are doing this is by testing them in our labs on stem cells derived from people with MND.
Recommended Reading: How To Recover From Dementia
What Did You Learn About This Interaction That Helps Explain How Alzheimers Unfolds
We were able to identify two key interactions between amyloid beta and tau during the course of Alzheimers progression based on large, publicly available datasets, which include PET scans for detecting pathological amyloid beta and tau. Our goal was to determine how the normal architecture of brain connections might facilitate these interactions between tau and amyloid and influence tau spreading.
First, we looked at interactions between amyloid beta and tau across a distance. Based on what we know about connections between parts of the brain, we could model the pathways through which these interactions might occur. We found that the earliest of these remote interactions takes place in the entorhinal cortex, the area where tau first appears in the cerebral cortex.
Second, we looked at what happened when the two proteins got close enough to actually co-mingle within a brain region. This type of interaction occurs first in the inferior temporal gyrus, or ITG, which is an important area for conveying visual information to other parts of the brain.
The ITG appears to act like a big city train station, a transportation hub with dozens of tracks leaving it for other brain regions. Once the two proteins interact locally within this hub, the amyloid seems to catalyze an acceleration phase in which the tau leaves the station bound for other areas in the neocortex where amyloid is already accumulating.
Are There Any Known Side Effects From Aducanumab
From the clinical trials, the main side effect of Aducanumab treatment reported was amyloid-related imaging abnormalities . These are thought to be micro-bleeds or swelling in the brain and have been reported in other anti-amyloid trials, particularly following high-dose treatment.
Biogen reported individuals with ARIA showed mild to moderate symptoms, with some displaying dizziness, headaches and nausea. These effects mean it will be important to continually monitor patients on undergoing Aducanumab treatment.
Read Also: What To Say To Someone Whose Mum Has Dementia
A Hypothesis And A Huge Gamble
The amyloid hypothesis says its the accumulation of beta-amyloid in the brain that is the primary cause of the memory-destroying disease. Studies on mice showed that removing the buildup worked wonders, and the hypothesis has been the driving force behind Alzheimers research for over 20 years.
Yet all attempts to develop amyloid-targeting drugs in people with the disease have ended in failure.
It was a huge gamble from the start, said Harvard neurologist Dr. Rudy Tanzi, who directs the Genetics and Aging Research Unit at Massachusetts General Hospital.
We know that amyloid deposition starts a decade or so before symptoms and its too late to treat it then, Tanzi said. Its like trying to put out a forest fire by putting out the match.
Instead, he said, drug makers should be focusing on early detection and early intervention, similar to how we treat cancer, diabetes and heart disease.
It has to be early detection and early treatment, years before symptoms begin, for it to work, Tanzi said. When is big pharma going to learn?
Study Tests Two Drug Treatments For Early
Treatment with either gantenerumab or solanezumab, two monoclonal antibodies, did not slow down cognitive decline in people who have a type of early-onset dementia called dominantly inherited Alzheimers disease , according to a recent study. However, gantenerumab did reduce some biomarkers of the disease. The study, which was funded in part by NIA, was published in Nature Medicine on June 21.
DIAD is a rare form of Alzheimers disease. It is an inherited condition caused by mutations in certain genes. People who have DIAD often start having symptoms of dementia, such as confusion and problems with memory, reasoning, and judgment, between the ages of 30 and 50. Currently, there is no treatment to prevent or slow down the disease. In the study, researchers led by a team at Washington University School of Medicine in St. Louis tested whether gantenerumab or solanezumab can effectively treat this condition. This study was conducted as part of the Dominantly Inherited Alzheimer Network Trials Unit .
The researchers found that in participants who had symptoms of dementia at the beginning of the trial, treatment with gantenerumab or solanezumab did not slow down or stop the worsening of these symptoms. Some participants taking solanezumab had a faster cognitive decline. Participants who had no symptoms at the start of the study did not develop symptoms during the study.
Read Also: How To Prevent Alzheimer’s Patients From Wandering
Risks And Ripple Effects
While the benefits of Aduhelm remain murky, some risks are clear. The same Phase III trials showed that 41% of patients who received the high dose of Aduhelm the dose that may have led to cognitive benefits experienced cerebral swelling or hemorrhage. Although the majority of these were minor and asymptomatic, a 75-year-old woman in the trial died after experiencing brain swelling and seizures. The risk was considered high enough for the FDA to require the company to place a warning on Aduhelms label, advising physicians to monitor patients and obtain two MRI brain scans during the first year of treatment.
Patients selected for the Phase III studies were excluded if they had any of the many medical complications common for older people. This includes cardiac problems, the use of blood thinners or impaired liver or kidney function. These selected patients, who may be healthier than those in the general public who would receive the medication, underwent not two but seven MRIs for monitoring. MRIs are expensive procedures they raise the real cost of Aduhelm by about 20%.
Why Is Diversity Important In Clinical Trials
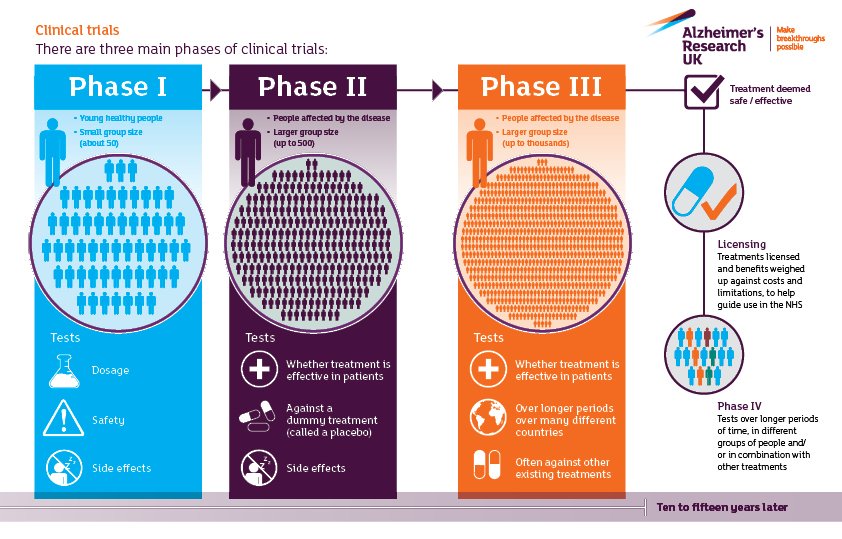
Researchers need participants who represent all types of races and ethnicities, genders, geographic locations, and sexual orientations.
When research involves a group of people who are similar, the findings may not apply to or benefit everyone. When clinical trials include diverse participants, the study results may have a much wider applicability.
Having diverse people in studies can help researchers understand how dementia affects certain groups, why some communities are disproportionately affected by certain dementias, and which treatments or prevention strategies may be most effective in particular groups.
Read Also: Can Vascular Dementia Cause Hallucinations
Discuss With Your Doctor
Should you or a loved one take aducanumab? You should discuss this decision with your doctor. Because we dont know if aducanumab works or not, deciding to take it is similar to deciding to participate in a clinical trial. As mentioned above, the known side effects are that 30% of people experienced reversible brain swelling and more than 10% had tiny brain bleeds. Although it does clear amyloid plaques from the brain, we dont know if it will slow down the dementia or not.
How Much Does It Cost And How Soon Can I Get It
Biogen, the company that makes aducanumab, estimates its cost at $56,000 per year. It is not yet clear whether Medicare, Medicaid, or private insurance companies will pay for this medication. In its press release, Biogen noted that they are working on an agreement with the Veterans Health Administration to cover this medication for veterans enrolled in VA healthcare. Aducanumab is not currently available, but the company is working to make the drug available quickly, perhaps in a few months.
You May Like: How Quickly Can Alzheimer’s Disease Progress
Treatments To Manage Agitation Insomnia And Apathy
While efforts to prevent or treat the cognitive symptoms of AD remain a primary focus, researchers have also recognized that we need treatments for the non-cognitive behavioral symptoms of AD and other dementias. Preliminary results suggest that we may be on the verge of more effective approaches to managing these behavioral symptoms. In addition to the familiar medications being tested , several new medications are also in clinical trials for AD behavioral symptoms.
Tetrahydrocannabinol is being tested for agitation treatment. The dopamine blocking drug, brexiprazole, may have promise in treating agitation. Pimavanserin, which reduces psychotic symptoms in Parkinsons disease, is being tested for management of psychotic symptoms in a broader group of major neurocognitive disorders.
Why Does There Seem To Be Controversy About Aducanumab
The FDAs approval of aducanumab is controversial for several reasons.
- Clinical trials studying the effectiveness of aducanumab did not prove that the drug improved cognition. In fact, two trials were stopped early because no clinical benefit was shown. Biogen reviewed the data again and noted a small improvement of symptoms in one subgroup of patients.
- Aducanumab is shown to reduce amyloid in the brain, but it is still unclear whether amyloid causes memory loss or Alzheimers disease. Therefore, the direct link between amyloid reduction and improvement of memory loss or other Alzheimers symptoms has not been established, and the clinical benefit for memory loss needs to be further explored.
- All but one member of an independent expert panel — enlisted to review the drugs effectiveness and make a recommendation to the FDA — voted against approving the drug.
- The FDA approved aducanumab through an accelerated pathway that is designed to bring the drug to market sooner, compared with the traditional pathway that most drugs take. This accelerated pathway can be used for drugs that treat diseases with no known cure, even if the clinical benefit is unclear and needs more investigation. As part of the accelerated FDA approval, Biogen is required to perform more research studies to answer some important questions related to clinical benefit. Duke plans to participate in these studies.
- The wholesale cost of the drug is high, meaning it could be too expensive for many patients.
Recommended Reading: Can An Mri Diagnose Alzheimer’s Disease
Trial Of New Alzheimers Drug Reports Disappointing Results
The drug, crenezumab, failed to prevent early symptoms or slow cognitive decline, the latest setback in the long quest to find effective therapies for the disease.
-
Send any friend a story
As a subscriber, you have 10 gift articles to give each month. Anyone can read what you share.
Give this article
By Pam Belluck
A closely watched clinical trial of a potential Alzheimers drug failed to prevent or slow cognitive decline, another disappointment in the long and challenging effort to find solutions for the disease.
The was the first time people who were genetically destined to develop the disease but who did not yet have any symptoms were given a drug intended to stop or delay decline. The participants were members of an extended family of 6,000 people in Colombia, about 1,200 of whom have a genetic mutation that virtually guarantees they will develop Alzheimers in their mid-40s to mid-50s.
For many members of the family, who live in Medellín and remote mountain villages, the disease has quickly stolen their ability to work, communicate and carry out basic functions. Many die in their 60s.
In the trial, 169 people with the mutation received either a placebo or the drug, crenezumab, produced by Genentech, part of the Roche Group. Another 83 people without the mutation received the placebo as a way to protect the identities of people likely to develop the disease, which is highly stigmatized in their communities.
I had put all my hopes in this study, his wife said.