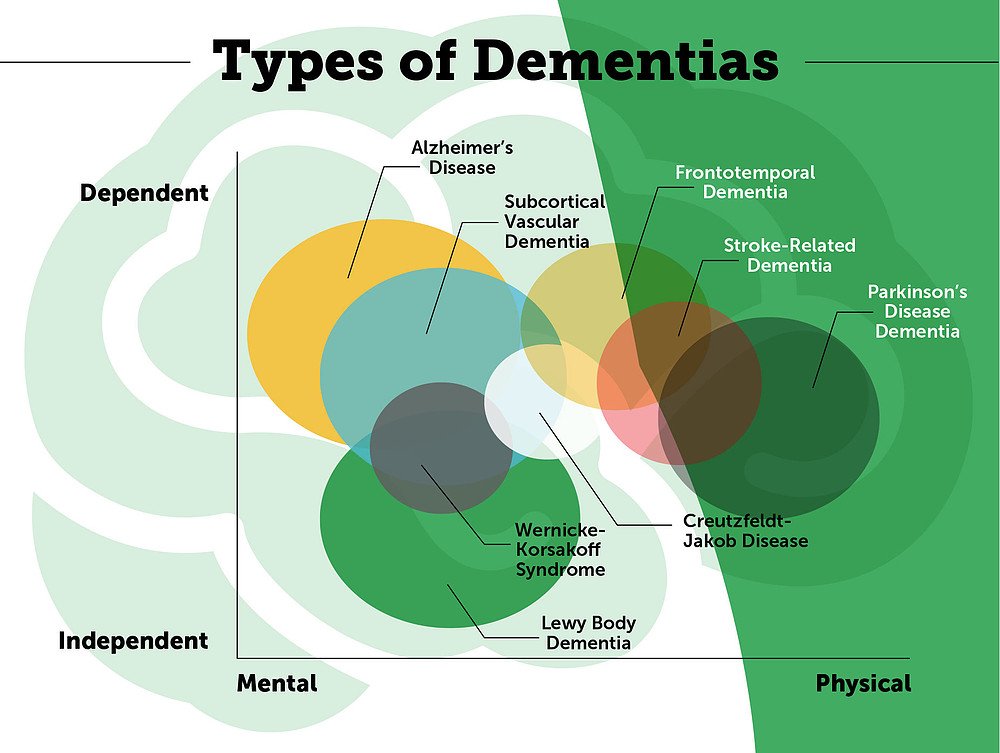
Types Of Alzheimer’s Disease
Nearly everyone with Alzheimerâs disease will eventually have the same symptoms — memory loss, confusion, trouble with once-familiar tasks, and making decisions. While the manner of the disease development remains unclear, all forms of Alzheimer’s appear to share overproduction and/or decreased clearance of a type of protein called amyloid beta peptides. Though the effects of the disease are similar, there are two main types.
- Early-onset Alzheimer’s. This type happens to people who are younger than age 65. Often, theyâre in their 40s or 50s when theyâre diagnosed with the disease. Itâs rare — up to 5% of all people with Alzheimer’s have early-onset. People with Down syndrome have a higher risk for it.Scientists have found a few ways in which early-onset Alzheimerâs is different from other types of the disease. People who have it tend to have more of the brain changes that are linked with Alzheimerâs. The early-onset form also appears to be linked with a defect in a specific part of a personâs DNA: chromosome 14. A form of muscle twitching and spasm, called myoclonus, is also more common in early-onset Alzheimer’s.
- Late-onset Alzheimer’s. This is the most common form of the disease, which happens to people age 65 and older. It may or may not run in families. So far, researchers havenât found a particular gene that causes it. No one knows for sure why some people get it and others donât.
Â
Genetic Testing For Alzheimers Disease
A blood test can tell which APOE gene you have, but the results canât predict whether youâll get Alzheimerâs. Doctors use these tests mostly for research purposes. The test can tell them who has certain risk factors so they can watch for brain changes in case the disease develops.
Doctors donât typically recommend genetic testing for late-onset Alzheimerâs because the results can be confusing and cause emotional distress. If youâre showing symptoms or have a family history, your doctor may recommend testing to help diagnose early-onset Alzheimerâs. Doctors can usually diagnose Alzheimerâs without a genetic test.
Genetic Testing And Counseling
In contrast, EOFAD, with an age at onset before 60 to 65 years old, has an autosomal dominant mode of inheritance in which 20% to 70% of cases are estimated to have a PSEN1 mutation, 10% to 15% of cases are estimated to have an APP mutation, and PSEN2 mutations are rare., Indeed, approximately 60% of patients with EOAD have another known affected family member. The remaining 40% of patients with EOAD may lack a family history because of an early death of a parent, failure to recognize the disorder in family members, or, very rarely, a de novo mutation. If the parent of a patient with EOAD has a mutant allele, then the risk to the patients sibling of inheriting the mutant allele is 50%. The child of a patient with EOAD who carries a mutation has a 50% chance of transmitting the mutant allele to each of their children.
In this article, we have reviewed the genetics of AD. Further molecular genetic investigations should clarify the roles of additional known genes in the pathogenesis of both common sporadic as well as rare familial forms of AD. Already, investigations of the normal and aberrant functions of A protein and ApoE has provided insight into the underlying mechanisms for AD. Such research will continue to provide new strategies for therapeutic interventions.
Dementia Associated With Corticobasal Degeneration
Corticobasal degeneration , sometimes referred to as corticobasal ganglionic degeneration, is considered to be a part of the spectrum of Frontotemporal dementia . It is characterized by nerve cell death and atrophy or shrinkage of multiple areas of the brain, including the cerebral cortex and basal ganglia. CBD typically occurs in individuals between the ages of 45-70. Rarely, there is a family history of dementia, psychiatric problems or a movement disorder.
Signs & Symptoms: Individuals with CBD usually present with either a movement disorder or cognitive deficits. As the disease progresses, they can go on to develop both types of symptoms.
The movement symptoms can be very similar to Parkinsons disease, with slowness, and rigidity, but tremor is less common. These symptoms do not respond to Parkinsons disease medications. Many individuals with CBD experience a subtle change in sensation or an inability to make the affected limb follow commands. They may have difficulties completing some specific tasks such as brushing teeth, opening a door or using tools such as a can opener. When it affects the legs, a person can have difficulty dancing or may show an increased tendency to trip and fall. Other symptoms include involuntary stiffening, twisting or contraction , or uncontrolled movement of the affected limb .
For more information, visit the National Institute of Neurological Disorders and Stroke:
How Do Genes Work
Genes are the basic building blocks that direct almost every aspect of how youâre built and how you work. Theyâre the blueprint that tells your body what color your eyes should be or if youâre likely to get some kinds of diseases.
You get your genes from your parents. They come grouped in long strands of DNA called chromosomes. Every healthy person is born with 46 chromosomes in 23 pairs. Usually, you get one chromosome in each pair from each parent.
If Familial Alzheimers Disease Is Suspected
Genetic testing can identify specific changes in a persons genes. This test can tell if a person has FAD and if a child has inherited the changed gene from a parent and will develop the disease in the future. It cannot determine when the symptoms will begin. It is essential to ensure that suspected cases in the family have, or have had, Alzheimers disease and not some other form of dementia. This can only be done through a medical examination, or a careful analysis of past medical records if the person is no longer alive.
Do Genes Cause Diseases
Genetic mutations can cause diseases. If a person inherits a genetic mutation that causes a certain disease, then he or she will usually get the disease. Sickle cell anemia, cystic fibrosis, and some cases of early-onset Alzheimer’s disease are examples of inherited genetic disorders.
Other changes or differences in genes, called genetic variants, may increase or decrease a person’s risk of developing a particular disease. When a genetic variant increases disease risk but does not directly cause a disease, it is called a genetic risk factor.
Identifying genetic variants may help researchers find the most effective ways to treat or prevent diseases such as Alzheimer’s in an individual. This approach, called precision medicine, takes into account individual variability in genes, environment, and lifestyle for each person.
The expression of geneswhen they are switched on or offcan be affected, positively and negatively, by environmental and lifestyle factors, such as exercise, diet, chemicals, or smoking. The field of epigenetics is studying how such factors can alter a cell’s DNA in ways that affect gene activity.
Alzheimer’s Genetics: Recent Progress
Linkage studies on late-onset AD have suffered similar limitations to those observed in association studies. Multiple genome-wide studies have been performed, often including overlapping samples, yet they have not been consistent in the genomic regions they identify and they rarely replicate each other’s results. Only a few genomic regions including chromosomes 9, 10 and 12 have been more consistently identified by linkage ; however, the presence of risk alleles within these regions remains unproven. The success of GWAS has led investigators today to pay less attention to linkage in complex disorder, leading to a current lack of new linkage results. It must be noted that under allelic heterogeneity, linkage would be successful where association tests would fail to give a positive result. In view of FAD being caused by more than 150 different mutations in the PSEN1 gene, it should be no surprise if the risk for late-onset AD is increased by more than ten different variants in a gene. Today it is sensible to follow the GWAS approach to find the first risk genes after APOE, because laboratory and analytical methods for identifying multiple rare disease-causing alleles across many genes are still in their infancy. New and emerging analytical methods and second-generation sequencing technologies will soon make it possible to explore this hypothesis in projects that will probably be guided by prior linkage and GWAS findings.
Managing Alzheimer’s Disease Behavior
Common behavioral symptoms of Alzheimers include sleeplessness, wandering, agitation, anxiety, and aggression. Scientists are learning why these symptoms occur and are studying new treatments drug and nondrug to manage them. Research has shown that treating behavioral symptoms can make people with Alzheimers more comfortable and makes things easier for caregivers.
Alzheimers Disease: From Genetic Variants To The Distinct Pathological Mechanisms
- 1Department of Geriatric Neurology, Xiangya Hospital, Central South University, Changsha, China
- 2Center for Advanced Therapeutic Strategies for Brain Disorders and Center for Hormone Advanced Science and Education, Roskamp Institute, Sarasota, FL, United States
- 3National Clinical Research Center for Mental Disorders, Beijing Key Laboratory of Mental Disorders, Beijing Anding Hospital, Capital Medical University, Beijing, China
- 4Beijing Institute for Brain Disorders, Capital Medical University, Beijing, China
- 5Neurodegenerative Disorder Research Center, University of Science and Technology of China School of Life Sciences, Hefei, China
- 6Hefei Material Science at Microscale National Laboratory, Hefei, China
Genetic Testing In Family Planning
Being able to test for the genetic mutations that cause familial dementia has now made genetic testing as part of family planning possible. This is an option when one prospective parent is known to carry a single-gene mutation, or is at risk because their parent had a mutation.
In these circumstances, genetic testing of the embryo allows the parents to have a child who does not carry the mutation. The technique used is called ‘pre-implantation genetic diagnosis’ . It is a modified form of in vitro fertilisation in which DNA from very early embryos is tested for a mutation in the lab. Only embryos which do not have the mutation are put back into the mother’s womb.
If successful, PGD means that the baby is almost certain not to have the mutation or to grow up to develop the particular kind of dementia.
It is possible in some centres to use this technology without the at-risk parent themselves knowing if they have inherited the mutation, if they choose not to.
PGD has been used for several years to help couples with Huntington’s disease who are starting a family. It has also recently become available on the NHS for families affected by familial Alzheimer’s disease or genetic FTD. As with predictive genetic testing, families considering PGD are offered extensive genetic counselling.
Genetic And Hereditary Risk Factors
As new discoveries develop in Alzheimers research, scientists are beginning to ascertain pieces to the puzzle concerning the cause. Many scientific discoveries are pointing to a strong link between Alzheimers disease, genetics, and hereditary risk factors.
Alzheimers disease is becoming known as a complex multifactorial disorder. This means that even though scientists are unaware of exactly how Alzheimers begins, they believe it is caused by environmental circumstances combined with genetic factors .
Risk Genes For Alzheimer’s Disease
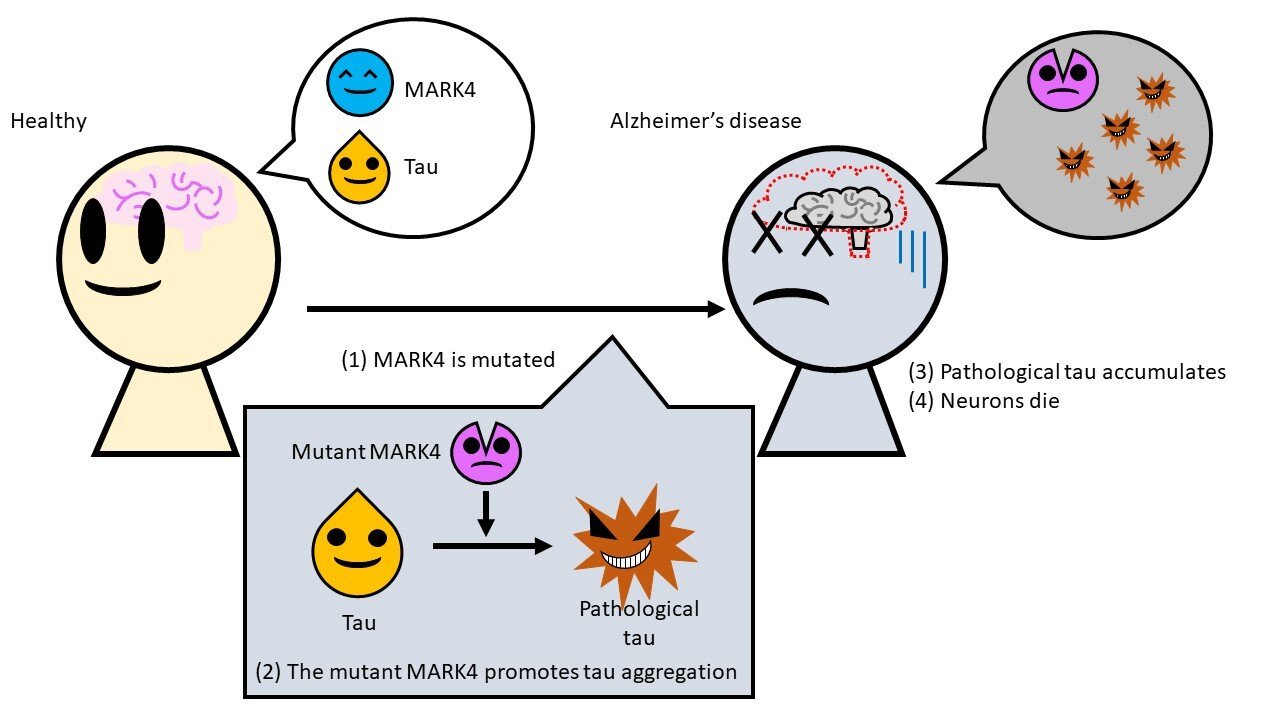
The vast majority of people with Alzheimer’s disease do not inherit it from a parent as a single-gene mutation with a simple inheritance pattern. Instead, the inheritance follows a more complex pattern. The disease might skip a generation, affect people on both sides of the family, appear seemingly from nowhere or not be passed on at all.
More than 20 gene variants have now been identified which affect – to different degrees – the chances of a person developing Alzheimer’s disease. The effects of these genes are subtle. Different variants act to slightly increase or decrease the risk of a person developing Alzheimer’s disease, but do not directly cause it. These ‘risk genes’ interact with each other and with other factors, such as age and lifestyle, to influence someone’s overall risk of getting the disease.
The Genetics Of Alzheimers Disease
Robert C. Barber
1Department of Pharmacology and Neuroscience, University of North Texas Health Science Center, 3500 Camp Bowie Boulevard, Fort Worth, TX 76107, USA
Abstract
Alzheimers disease is a progressive, neurodegenerative disease that represents a growing global health crisis. Two major forms of the disease exist: early onset and late onset . Early onset Alzheimers is rare, accounting for less than 5% of disease burden. It is inherited in Mendelian dominant fashion and is caused by mutations in three genes . Late onset Alzheimers is common among individuals over 65 years of age. Heritability of this form of the disease is high , but the etiology is driven by a combination of genetic and environmental factors. A large number of genes have been implicated in the development of late onset Alzheimers. Examples that have been confirmed by multiple studies include ABCA7, APOE, BIN1, CD2AP, CD33, CLU, CR1, EPHA1, MS4A4A/MS4A4E/MS4A6E, PICALM, and SORL1. Despite tremendous progress over the past three decades, roughly half of the heritability for the late onset of the disease remains unidentified. Finding the remaining genetic factors that contribute to the development of late onset Alzheimers disease holds the potential to provide novel targets for treatment and prevention, leading to the development of effective strategies to combat this devastating disease.
1. Introduction
2. Familial Alzheimers Disease
2.1. Amyloid Precursor Protein
2.2. Presenilins 1 and 2
Genes Associated With Risk In Sporadic Ad
AD2: APOE
Inheritance and clinical features
The APOE gene has been associated with both familial late-onset and sporadic late-onset AD in numerous studies of multiple ethnic groups. The APOE4 genotype is associated with higher risk of AD, earlier age of onset of both AD and Down syndrome , and a worse outcome after head trauma and stroke, both in humans and in transgenic mice expressing human APOE4. The frequency of the APOE4 allele varies between ethnic groups, but regardless of ethnic group, APOE4 carriers are more frequently found in controls and APOE4+ carriers are more frequently found in patients with AD.,
Gene location and structure
Apolipoprotein E structure and single nucleotide polymorphisms . The general protein structure of APOE is shown. The 2 SNPs in exon 4 and corresponding protein locations are shown . The 3 APOE 4 alleles are defined by 2 SNPs, rs429358 and rs7412, with 2 defined by nucleotides T-T; 3 defined by T-C, and 4 defined by C-C, respectively.
Gene function and expression
The mechanisms that govern apoE toxicity in brain tissue are not fully understood. Some proposed mechanisms include isoform-specific toxicity, APOE4-mediated amyloid aggregation, and APOE4-mediated tau hyperphosphorylation.
In addition, individuals carrying APOE4 have higher amyloid and tangle pathology and an increase in mitochondrial damage compared to those carrying other APOE polymorphisms.
Genetic variation
Genes: A Blueprint For Health And Disease
The genes encoded in our DNA are profoundly involved in many aspects of our health. They act as a sort of blueprint for the construction, operation, and repair of our bodies throughout life. Genes provide instructions for the creation and regulation of our bodys building blocks. We inherit one copy of each gene from each of our parents, which is one reason why every person has a unique appearance and metabolism.
The effects of a gene can be dramatically changed by mutation of even one pair of its molecules. Specific versions of genes, called alleles, are passed down through a familys lineage, potentially creating an entire population of people who share a healthy characteristic such as resistance to cancer; a more neutral characteristic such as eye color; or a heightened risk for a specific disease such as Alzheimers disease .
Many of us have learned about Mendel, who was an Austrian monk, and the pea plants he bred as he discovered basic principles of genetic inheritance. Just as with Mendels plants, some physical characteristics can be transmitted to our offspring through inheritance of even one version of a gene. Many diseases are inherited this way, too.
Health Environmental And Lifestyle Factors
Research suggests that a host of factors beyond genetics may play a role in the development and course of Alzheimers. There is a great deal of interest, for example, in the relationship between cognitive decline and vascular conditions such as heart disease, stroke, and high blood pressure, as well as conditions such as diabetes and obesity. Ongoing research will help us understand whether and how reducing risk factors for these conditions may also reduce the risk of Alzheimers.
A nutritious diet, physical activity, social engagement, and mentally stimulating pursuits have all been associated with helping people stay healthy as they age. These factors might also help reduce the risk of cognitive decline and Alzheimers. Researchers are testing some of these possibilities in clinical trials.
What Is Alzheimers Disease
Alzheimers disease damages your brain, gradually destroying memory and thinking skills. Researchers believe that the damage begins up to a decade before symptoms appear. Abnormal deposits of proteins form hard plaques and tangles throughout the brain. These deposits interfere with normal brain function.
As they grow, plaques can interrupt communication between neurons, the messengers in your brain. Eventually these neurons die, damaging your brain so much that parts of it begin to shrink.
What Is An Allele
An allele is a variant form of a gene. At conception, an embryo receives an allele of a gene from the mother and an allele of a gene from the father. This combination of alleles is what determines genetic characteristics such as the color of a persons eyes or hair. The APOE gene is located on chromosome number 19 and has three common alleles, including:
- APOE-e2: a rare allele that is thought to provide some protection against Alzheimers
- APOE-e3: an allele that is thought to be neutral as far as the risk of Alzheimers disease is concerned, this is the most common allele of the APOE gene
- APOE-e4: the third allele, which has been identified as increasing a persons risk of getting late-onset Alzheimers. A person can have zero, one, or two APOE4 alleles .
Its important to note that not everyone with the APOE4 allele will necessarily get Alzheimers. This may be because there are many factors involvedin addition to the genetic link to the diseasesuch as environmental and lifestyle factors.
Therefore, a person who inherits the APOE4 allele of the gene is not assured of getting Alzheimers disease. On the other hand, many people who get diagnosed with AD, do not have the APOE4 form of the gene.
Genetic Risk Prediction In Complex Ad
The role of genetics in diagnosis and risk prediction in late-onset complex AD is much less straightforward. Despite the established evidence of APOE 4 as a risk factor for AD, its value in disease prediction in a clinical setting is limited, not only due to the restriction of current therapeutic consequences but also because APOE 4 is neither necessary nor sufficient to cause the disease. Up to 75% of individuals heterogeneous for APOE 4 do not develop AD during life, and up to 50% of people with AD do not carry the high-risk 4 allele.
Effect Of Genes In Other Types Of Dementia
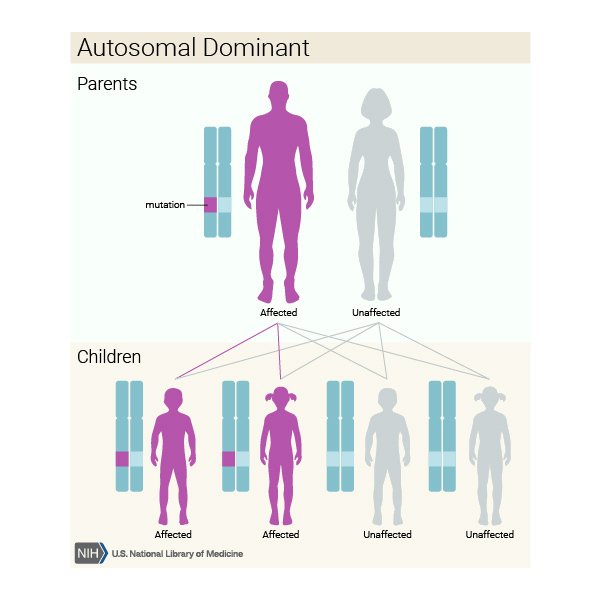
Some types of dementia have links to other genetic malformations.
Huntingtons disease, for example, alters chromosome 4, which can lead to progressive dementia. Huntingtons disease is a dominant genetic condition. This means that if either parent has the condition, they can pass on the gene to their offspring and they will develop the disease.
Symptoms of Huntingtons disease do not usually become evident until a person reaches 3050 years of age. This can make it difficult for doctors to predict or diagnose before a person has children and passes on the gene.
Researchers think that dementia with Lewy bodies or Parkinsons dementia may also have a genetic component. However, they also believe that other factors outside of genetics might play a role in the development of these conditions.
Researchers have identified several risk factors for Alzheimers disease.
These include:
- difficulty recognizing people
- frequently misplacing objects and belongings
The aging process might naturally impair a persons memory, but Alzheimers disease leads to more consistent periods of forgetfulness.
Over time, a person with Alzheimers may need an increasing amount of assistance with daily living, such as brushing teeth, getting dressed, and cutting food.
They may also experience agitation, restlessness, personality withdrawals, and speech problems.
How Is It Diagnosed
The gene that causes Huntingtons disease was identified in 1993. Genetic testing is now available to confirm the presence of the gene in symptomatic people with suspected symptoms and those without symptoms, but who are determined to be at risk of developing it based on a family history of the disease.
Genes And Dementia With Lewy Bodies
Dementia with Lewy bodies is caused by a build-up of abnormal proteins in the brain and may have symptoms similar to those seen in Parkinsons disease. Age is currently the biggest known risk factor for dementia with Lewy bodies, although research is underway to find out whether genes may also play a role.
Causes Of Alzheimers: Is It Hereditary
Increasing cases of Alzheimers disease
The Alzheimers Association states that Alzheimers disease is the sixth leading cause of death in the United States, and that more than 5 million Americans are affected by the condition. Additionally, one out of three seniors dies of Alzheimers or some other type of dementia. That number will likely increase as the aging population increases.
Scientists have been researching Alzheimers for decades, but still there is no cure. Learn more about how genes are related to the development of Alzheimers, as well as other potential causes of the condition.
Contribution Of Ad Genetics To Treatment And Diagnosis
It is now more than 15 years since the discovery of the first genes involved in AD, and while we are still on a quest for additional genes, we have learned a lot about the disease. The processing of APP through cleavage by -secretase, an enzymatic complex whose catalytic subunit is formed by the presenilins , is considered by many as a key in the disease process. It leads to generation of the amyloidogenic peptide A and its aggregation into fibrils and toxic oligomeric forms, the earliest effectors of synaptic compromise , followed by neurodegeneration. The direct involvement of the products of at least three out of the four known genes in this hypothesis is no coincidence and significantly strengthens the confidence that this is a promising target for treatment. These genes have greatly enhanced our knowledge of the pathway leading to the production of amyloid , which has in turn provided targets for intervention.
Figure 1
The amyloidogenic and non-amyloidogenic cleavage pathway of APP. Mutations in APP, PSEN1 or PSEN2cause early-onset AD. PSEN1 and 2 are components of -secretase. The amyloidogenic peptide A is shown in orange.
Genes And Frontotemporal Dementia
Frontotemporal dementia , originally called Picks disease, is a rarer type of dementia mostly affecting people under the age of 65 years. The symptoms of FTD can be quite varied but include changes that mostly affect behaviour or language. There are different types of FTD, and these are likely to have different causes.
Some people with FTD have a family history of dementia and the condition may be inherited in some of these families. For behavioural variant FTD, a third to half of people could have a family history. This figure is thought to be much lower for other types of FTD.
Overall, around one in ten cases of FTD are thought to be caused by a faulty gene passed down in families. Several genes have been found that can cause these inherited types of FTD, including:
- tau
- progranulin
Mutations in the MAPT gene can cause the tau protein to behave abnormally, forming toxic clumps that can damage brain cells. We still need to understand more about how mutations in progranulin and C9ORF72 cause the disease.
The C9ORF72 gene can cause people to develop motor neurone disease, FTD or both conditions, and may affect members of the same family differently.
In cases of FTD that are not caused by faulty genes, the risk factors are not yet fully understood, and research is ongoing.
Is genetic testing available for frontotemporal dementia?