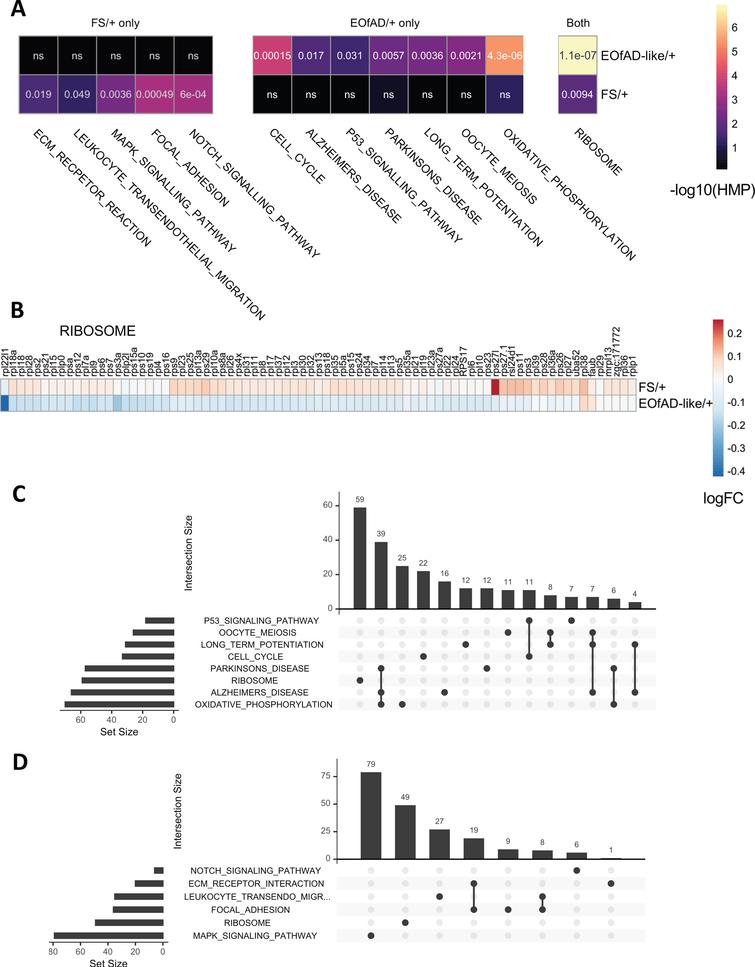
Iron Inflammation And Neurodegeneration
Iron is the most abundant element in our planet and the most abundant transition metal in the human body . It has been a central component of energy metabolism since the dawn of life, over one billion years before oxygenation of the Earths atmosphere drove the evolution of energy production by oxidative phosphorylation . Iron has been used medicinally for thousands of years before elucidation of its role in biology began with its discovery as a component of our blood .
Inflammation is recognized as a key, unifying characteristic of all forms of neurodegeneration so it is unsurprising that iron appears to play key roles in many neurodegenerative diseases. Some examples of this are to be provided but the main aim of this paper is to examine the role iron may play in the most common form of dementia, Alzheimers disease.
What Are The Different Types Of Alzheimers Disease And How Do I Refer To Them Correctly
Both early-onset familial Alzheimers disease and late-onset Alzheimers disease are progressive neurodegenerative diseases characterized by the damaging presence of amyloid plaques and tau tangles in the brain. The main differences between the two types of Alzheimers disease are the age of onset of the disease and the cause of the disease.
A simplified explanation of the two is below, as well as a table that summarizes that information.
Early-onset familial Alzheimers disease : EOFAD affects much younger individuals compared to LOAD. It develops in individuals before the age of 65, usually between the ages of 30 60 years with an average age of about 45. Unlike LOAD, EOFAD is a rare genetic disorder that is inherited in an autosomal dominant manner. For this reason, many families with EOFAD often see multiple family members over multiple generations affected by the disease. As a rare genetic disease, EOFAD represents less than 1% of all Alzheimers disease cases , affecting 5.3 out of 100,000 individuals . It has been estimated that ~45,000 individuals worldwide are affected by EOFAD.
Late-onset Alzheimers disease : As the name suggests, LOAD tends to affect older individuals, usually after the age of 65. It is a much more common disease ) and tends to have a less aggressive disease course than EOFAD. Although some risk factors have been identified, we do not know exactly what causes LOAD.
Information About Genetic Testing
Having a test to look for a faulty gene that causes dementia is only appropriate for a very small number of people. This is because inherited dementia is rare.
If you are worried that you have a strong history family of early-onset Alzheimers disease or frontotemporal dementia, you can speak to your doctor about this.
Not all gene mutations that cause dementia have been identified, meaning that some families may have many affected members, but no mutation can be found. Therefore, a negative test result cannot always rule out a genetic cause of a disease.
If a test is appropriate, your doctor should be able to refer you to a genetic counsellor or specialist. This could be a cognitive neurologist or memory clinic psychiatrist. They will discuss with you the pros and cons of taking a test and what will be involved. They will also tell you where the results will be kept, who they will be shared with, and what the next steps would be. For people found to have a genetic mutation that causes dementia, these discussions will also cover the options available if you are considering starting a family.
To find out more about genetic testing and what support is available you can visit www.raredementiasupport.org or call 020 3325 0828. Leave a message and you will be referred to the most appropriate team member.
Read Also: Are Hallucinations Common With Dementia
Dementia With Lewy Bodies And Its Symptoms
This affects around 100,000 people in the UK or 15 per cent of those with dementia. DLB, also known as Lewy body disease, can cause common dementia symptoms such as memory loss, spatial awareness problems and a worsening of problem-solving skills. In mixed dementia cases, it may also be combined with Alzheimers disease.
affects around 100,000 people in the UK
In addition, there are some specific symptoms associated with DLB, some of which may also be seen in Parkinsons-related dementia. They include changes in alertness, confusion, slowed movements, muscle stiffness and tremors, visual hallucinations, sleep disturbances which can cause people to move about or talk in their sleep, fainting, unsteadiness and falls.
DLB can progress slowly over several years but the speed of progression and exact symptoms vary from person to person. Aside from age, specific risk factors for DLB are not known at this stage.
There is some evidence that the cholinesterase inhibitors used to treat Alzheimers may help to improve some of the symptoms of DLB, including disturbing visual hallucinations.
Subjects And Family Ascertainment
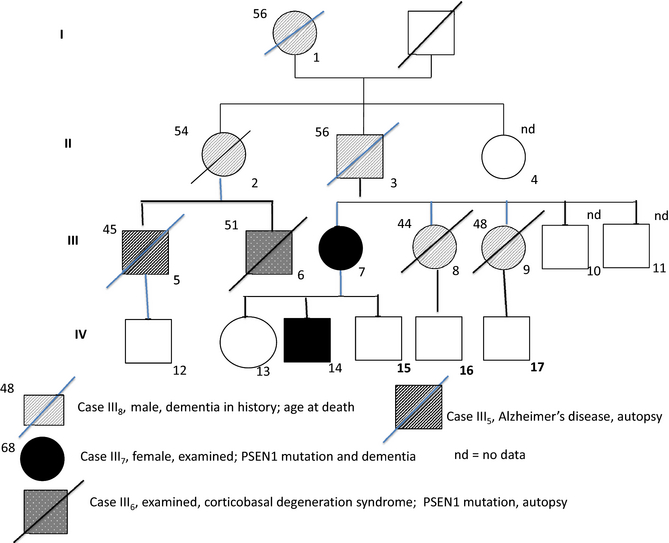
Informed consent was obtained from the subject or next of kin for all DNA samples collected in both pedigrees, and from the next of kin for neuropathological studies. The research programme was approved by the Ethics Committee of the Central Sydney Area Health Service.
EOFAD2
Family members are descendants of a man of AngloCeltic ethnicity who died at age 50;years of dementia. Little is known of his parents or siblings, but five of his six children were affected, including the proband, III:18, who died at age 60;years with Alzheimers disease confirmed at autopsy. Fourteen subjects in three generations have been affected, with DNA available from seven affected and four unaffected individuals. The clinical phenotype in the family was that of presenile dementia, with no confirmed instances of spastic paraparesis at the time of ascertainment. However, one subject who was asymptomatic at the time of blood collection has since developed spasticity and cognitive deficits. Four subjects have had autopsy confirmation.
EOFAD3
Recommended Reading: How To Calm Down A Dementia Person
A Stress Threshold Change Of State Into Alzheimers Disease
FIGURE 5. A model of Alzheimers disease development encompassing both EOfAD and LOsAD genetics. An early onset age of Alzheimers disease may occur due to metabolic stress on the brain, possibly due to iron dysregulation. This, in its effects, resembles hypoxic stress and supplements the gradually rising hypoxic stress normally driven by vascular aging so that a stress tolerance threshold is exceeded sooner, after which the brain shifts into a hypometabolic state, possibly in order to delay further damage due to rising oxidative stress. The timing and extent of this process will vary across different brain regions depending on basal metabolic load, glycolytic capacity, oxygen supply, etc.
App An Expression Conundrum
An encouraging aspect of refocusing EOfAD analysis around changes in iron homeostasis is that it requires us to give greater attention to APPs role in stress responses. Hypoxia causes oxidative stress that upregulates expression of APP, BACE1, and the PSENs and can, thereby, greatly increase production of A that can function as an antioxidant . Accumulating evidence also supports a role for A as an antimicrobial peptide . A was reported to suppress the growth of Candida albicans and a variety of bacterial species at concentrations equal to, or lower than, another characterized human antimicrobial peptide, LL37, and depletion of A from Alzheimers disease brain homogenates reduces the latters ability to inhibit growth of C. albicans . An antimicrobial function for A would explain its high structural conservation in most species of the tetrapod evolutionary lineage .
Read Also: How To Get Tested For Alzheimer’s Gene
What Is Alzheimers Disease
Dementia is an umbrella term used to describe a group of symptoms that occur when nerve cells die and brain tissue is lost. There are a number of different types of dementia; the most common, most studied and best understood form is Alzheimers disease, which accounts for about two-thirds of cases.
While the biggest risk factor for Alzheimers is age, it is not a natural or inevitable part of ageing. On average, people live for between eight and 10 years after an Alzheimers diagnosis, but researchers believe that the disease process in the brain may begin up to 20 years before the first symptoms appear.
Alzheimers is a progressive disease with three stages, beginning with the early or mild stage, progressing to the middle or moderate stage and ultimately, the late or severe stage. While early signs, including mild forgetfulness, can be hard to distinguish from those of normal ageing, other typical symptoms may be disorientation or confusion about the time of day.
People with dementia may find it increasingly hard to communicate and often need more help with everyday functions
Although the speed of deterioration varies between individuals, people with dementia may find it increasingly hard to communicate and often need more help with everyday functions, such as eating or dressing, as the disease progresses.
Clinical Genetics Of Alzheimers Disease
Zhangyu ZouHuapin Huang
1Department of Neurology, Fujian Medical University Union Hospital, Fuzhou 350001, China
Abstract
Alzheimers disease is the most common progressive neurodegenerative disease and the most common form of dementia in the elderly. It is a complex disorder with environmental and genetic components. There are two major types of AD, early onset and the more common late onset. The genetics of early-onset AD are largely understood with mutations in three different genes leading to the disease. In contrast, while susceptibility loci and alleles associated with late-onset AD have been identified using genetic association studies, the genetics of late-onset Alzheimers disease are not fully understood. Here we review the known genetics of early- and late-onset AD, the clinical features of EOAD according to genotypes, and the clinical implications of the genetics of AD.
1. Introduction
Alzheimers disease , the most common progressive neurodegenerative disease and the most common form of dementia in the elderly, results from irreversible loss of neurons, particularly in the cortex and hippocampus. Clinically, AD is characterized by progressive impairments of memory, judgment, decision making, orientation to physical surroundings, and language. Pathologically, AD is characterized by the presence of extracellular neuritic plaques containing the -amyloid peptide and neurofibrillary tangles composed of hyperphosphorylated tau protein in the brain.
3.1. APP Gene
Recommended Reading: Is Weight Loss A Symptom Of Dementia
Dysregulation Of Neuronal Iron Homeostasis As An Alternative Unifying Effect Of Mutations Causing Familial Alzheimers Disease
- 1College of Medicine and Public Health, Flinders University, Adelaide, SA, Australia
- 2South Australian Health and Medical Research Institute, Adelaide, SA, Australia
- 3Neurochemistry Laboratory, Department of Psychiatry-Neuroscience, Massachusetts General Hospital , Harvard Medical School, Harvard University, Charlestown, MA, United States
- 4Neuronal Injury and Repair Laboratory, Centre for Neuroscience, College of Medicine and Public Health, Flinders University, Adelaide, SA, Australia
- 5Centre for Molecular Pathology, School of Biological Sciences, University of Adelaide, Adelaide, SA, Australia
- 6Discipline of Pathology, Sydney Medical School, University of Sydney, Sydney, NSW, Australia
- 7School of Pharmacy and Biomedical Sciences, Faculty of Health Sciences, Curtin Health Innovation Research Institute, Curtin University, Bentley, WA, Australia
The Intimate Relationship Between Iron Homeostasis Energy Metabolism And Responses To Hypoxia
Many proteins require Fe2+ as a cofactor, especially mitochondrial proteins involved in the TCA and in the protein complexes of the electron transport chain . Therefore, a deficiency of cytosolic Fe2+ is expected to interfere with normal mitochondrial function and increase oxidative stress while excessive cytosolic iron accumulation is thought to cause oxidative stress via Fenton chemistry . This is consistent with the fact that aberrant mitochondrial activity is a common phenomenon observed in Alzheimers disease studies .
Electron transport chains causing reduction of oxygen exist both in mitochondria and in the MAM of the ER . Both processes produce considerable quantities of H2O2 that can interact, in Fenton chemistry, with either Fe2+ or Fe3+ to produce highly reactive hydroxyl or hydroxyperoxyl radicals, respectively . Oxidative stress that exceeds mitochondrias antioxidant capacity will damage iron-containing proteins leading to additional iron release in a potential positive feedback loop. This further decreases mitochondrial function and cells capacity for production of energy by oxidative phosphorylation .
Don’t Miss: What Is A Memory Test For Dementia
What Happens With Early
Someone in the early stages of dementia may often become confused. When memory, thinking, or judgment lapses, confusion may arise as they can no longer remember faces, find the right words, or interact with people normally. Confusion can occur for a number of reasons and apply to different situations.
What Is Vascular Dementia And How Does It Start
Vascular dementia can occur when blood flow to the brain becomes reduced. Mixed dementia is where people have both Alzheimers and another disease such as vascular dementia.
The second most common form of dementia, vascular dementia can have symptoms similar to Alzheimers, including memory loss, disorientation and problems with communication. There can also be more specific symptoms, such as difficulty in walking or personality changes, including depression and apathy. These can differ depending on the area of the brain that is affected.
The most common types of vascular dementia are stroke-related dementia, which can occur following either a single stroke or a series of mini-strokes, and subcortical vascular dementia caused by changes to very small blood vessels in the brain.
While there are currently no specific treatments for vascular dementia, a doctor may prescribe medication to treat underlying, related medical conditions such as diabetes, high blood pressure, high cholesterol or heart problems.
Some of the symptoms may be managed by physiotherapy, occupational therapy or speech therapy, and as with Alzheimers, cognitive therapy may stimulate thinking skills and help keep patients engaged with others.
You May Like: How To Differentiate Delirium From Dementia
What Is Early Onset Familial Alzheimer Disease
Definition: What Is eFAD?
;Early onset familial Alzheimer disease is hereditary and marked by Alzheimer disease symptoms that appear at an unusually early age. Symptoms can start in a person’s thirties, forties, and fifties . Generally, if you are diagnosed with eFAD, then one of your parents will also have had it if he or she lived long enough, and your siblings and your children may have a 50-50 chance of having inherited it. Very rarely, eFAD can make a first-time appearance in a family through a new genetic mutation.
Genetics researchers studied eFAD families to discover the three known genes that cause familial AD: amyloid precursor protein , presenilin-1 , and presenilin-2 . Of these, PS1 mutations account for most eFAD, while APP and PS2 are more rare. Having a pathogenic mutation in one of these three genes virtually guarantees that one will develop early onset Alzheimer disease. Tests can determine which gene is at fauly . There are also cases of eFAD that cannot be linked to one of these three genes. There may be additional genes waiting to be discovered, if only researchers could connect with more eFAD families.
Prognosis: Is eFAD Different from LOAD?
How Common Is Early Onset Familial Alzheimer Disease?
For practical and research purposes, doctors and scientists need defined populations for study and the numbers change based on the definitions. The definition would seem to rest on two criteria:
| ; | |
| Early onset sporadic | Late-onset sporadic |
Exactly What Is Alzheimers Disease Is It The Same
- School
- 67%4 out of 6 people found this document helpful
This preview shows page 146 – 148 out of 173 pages.
Students who viewed this also studied
City Colleges of Chicago, Malcolm X
BIO 121
City Colleges of Chicago, Malcolm X BIO 121
BIO 121 Manual Final.pdf
University of Central Florida CHM 2040
Chapter 1_slides.pdf
West Virginia University BIOL 453
Case Study 5.docx
Bowie State University BIOLOGY 203
Tackling Genetic Possibilities.docx
Chamberlain College of Nursing ADVANCED P 508
ch14
South Texas College BIOL 11
Mader_BIO11e_LRG .doc
We have textbook solutions for you!
The document you are viewing contains questions related to this textbook.
Don’t Miss: When A Loved One Has Dementia
Uncertainty Regarding The Mechanistic Role Of A In Alzheimers Disease
As of June 2018, a search in PubMed for the term Alzheimers disease finds over 105,000 papers . Nevertheless, there is still no consensus on the pathological mechanism underlying this disease. Indeed, the very definition of the disease itself is disputed .
The past two decades have seen great enthusiasm for the Amyloid Hypothesis of Alzheimers disease that posits a pathological role for the A peptide cleaved from the AMYLOID BETA A4 PRECURSOR PROTEIN . The Amyloid Hypothesis is an irrefutable tenet for many in the Alzheimers disease research community since changes in A production appear to be the only common mechanism linking the few loci where mutations causing early onset familial Alzheimers disease are found. However, the mode of As purported toxicity remains to be clearly defined and recent prominent review papers have admitted to serious incongruities between observations of A deposition in the brain and the progress of the disease . It is thus uncertain whether A, accumulation of which is currently a required element in the definition of Alzheimers disease is a causative agent, a protective mechanism, or an innocent bystander in the pathological process.
Genes And Dementia With Lewy Bodies
Dementia with Lewy bodies is caused by a build-up of abnormal proteins in the brain and may have symptoms similar to those seen in Parkinsons disease. Age is currently the biggest known risk factor for dementia with Lewy bodies, although research is underway to find out whether genes may also play a role.
Recommended Reading: How Many People Does Alzheimer’s Affect
Types Of Alzheimer’s Disease
Nearly everyone with Alzheimerâs disease will eventually have the same symptoms — memory loss, confusion, trouble with once-familiar tasks, and making decisions.Â; While the manner of the disease development remains unclear, all forms of Alzheimer’s appear to share overproduction and/or decreased clearance of a type of protein called amyloid beta peptides. Though the effects of the disease are similar, there are two main types.
- Early-onset Alzheimer’s. This type happens to people who are younger than age 65. Often, theyâre in their 40s or 50s when theyâre diagnosed with the disease. Itâs rare — up to 5% of all people with Alzheimer’s have early-onset. People with Down syndrome have a higher risk for it.Scientists have found a few ways in which early-onset Alzheimerâs is different from other types of the disease. People who have it tend to have more of the brain changes that are linked with Alzheimerâs. The early-onset form also appears to be linked with a defect in a specific part of a personâs DNA: chromosome 14. A form of muscle twitching and spasm, called myoclonus, is also more common in early-onset Alzheimer’s.
- Late-onset Alzheimer’s. This is the most common form of the disease, which happens to people age 65 and older. It may or may not run in families. So far, researchers havenât found a particular gene that causes it. No one knows for sure why some people get it and others donât.