Loss Of Neuronal Connections And Cell Death
In Alzheimers disease, as neurons are injured and die throughout the brain, connections between networks of neurons may break down, and many brain regions begin to shrink. By the final stages of Alzheimers, this processcalled brain atrophyis widespread, causing significant loss of brain volume.
Learn more about Alzheimer’s disease from MedlinePlus.
Missense Mutations In The App Gene And Its Neuropathological Profile
Like the duplications in APP, most missense variants in APP are associated with autosomal-dominant inheritance of AD, usually with complete penetrance by age 60 . An autosomal-recessive mutation at codon 673 of APP was recently discovered and reported to be associated with AD . The APP gene is encoded by 18 exons that are alternatively spliced to produce proteins ranging in size from 695 to 770 amino acids. The A peptide is encoded by parts of exons 16 and 17 . To date, 26 pathogenic missense mutations have been reported within the APP gene . These mutations are located within or immediately flanking the A sequence. A is generated from APP by the sequential cleavage of two enzymes, -secretase and -secretase .2). We will discuss further the mechanisms by which these mutations act and display their neuropathological hallmarks.
The Stages Of Alzheimers Disease
The clinical phases of Alzheimers disease can be classified into pre-clinical or the pre-symptomatic stage, which can last for several years or more. This stage is characterized by mild memory loss and early pathological changes in cortex and hippocampus, with no functional impairment in the daily activities and absence of clinical signs and symptoms of AD . The mild or early stage of AD, where several symptoms start to appear in patients, such as a trouble in the daily life of the patient with a loss of concentration and memory, disorientation of place and time, a change in the mood, and a development of depression . Moderate AD stage, in which the disease spreads to cerebral cortex areas that results in an increased memory loss with trouble recognizing family and friends, a loss of impulse control, and difficulty in reading, writing, and speaking . Severe AD or late-stage, which involves the spread of the disease to the entire cortex area with a severe accumulation of neuritic plaques and neurofibrillary tangles, resulting in a progressive functional and cognitive impairment where the patients cannot recognize their family at all and may become bedridden with difficulties in swallowing and urination, and eventually leading to the patients death due to these complications .
Read Also: Shampoos That Cause Alzheimer’s
Key Biological Processes In The Brain
Most neurons have three basic parts: a cell body, multiple dendrites, and an axon.
- The cell body contains the nucleus, which houses the genetic blueprint that directs and regulates the cells activities.
- Dendrites are branch-like structures that extend from the cell body and collect information from other neurons.
- The axon is a cable-like structure at the end of the cell body opposite the dendrites and transmits messages to other neurons.
The function and survival of neurons depend on several key biological processes:
Neurons are a major player in the central nervous system, but other cell types are also key to healthy brain function. In fact, glial cells are by far the most numerous cells in the brain, outnumbering neurons by about 10 to 1. These cells, which come in various formssuch as microglia, astrocytes, and oligodendrocytessurround and support the function and healthy of neurons. For example, microglia protect neurons from physical and chemical damage and are responsible for clearing foreign substances and cellular debris from the brain. To carry out these functions, glial cells often collaborate with blood vessels in the brain. Together, glial and blood vessel cells regulate the delicate balance within the brain to ensure that it functions at its best.
Patterns Of Protein Expression
To identify the unique fingerprint of Alzheimers, the scientists analyzed patterns of protein expression in more than 2,000 brains and almost 400 samples of cerebrospinal fluid. They used samples from people with the condition and from healthy, age-matched controls.
They also compared cerebrospinal fluid samples from people with Alzheimers with those from people with six other neurodegenerative conditions.
Among the proteins expressed in greater amounts in Alzheimers disease were several involved in breaking down glucose in the brain to provide energy.
In a previous study , the same research team found irregularities in glucose metabolism.
Specifically, they found that the activity of enzymes controlling glycolysis that is, the main process by which cells break down glucose was lower in the brains of people with Alzheimers than in those with normal brain tissue samples.
These irregularities in people with Alzheimers correlated with the number of amyloid plaques and tangles in their brains. Lower enzyme activity also correlated with the severity and development of symptoms such as memory loss.
Weve been studying the possible links between in the way the brain metabolizes glucose and Alzheimers-related changes for a while now, says study co-author Dr. Madhav Thambisetty, of the NIAs Laboratory of Behavioral Neuroscience.
You May Like: Low Blood Pressure Dementia
Everything You Need To Know About The Causes Of Alzheimers Disease
Dementia isnt just a part of ageing. Its caused by diseases.
There are several diseases that cause dementia including vascular dementia and dementia with Lewy bodies, but the most common cause of dementia is Alzheimers disease.
Alzheimers disease is the culprit in around 2/3 of dementias.
Overviews Of Current Therapeutics Targeting A
According to the conventional approaches targeting A, therapeutic strategies focus on reducing A production via inhibition of – and -secretases to prevent A aggregation and facilitate A clearance. However, the results are not so inspiring, as all the strategies have failed in clinical trials. Recently, immunotherapies by two monoclonal antibodies against A have been tried. One is Bapineuzumab that could recognize both soluble and insoluble forms of A the other one is Solanezumab that targets A central domain and recognizes only soluble A. Yet both of them failed to improve the clinical outcomes in patients in phage III trials , which suggests that targeting A alone might not be enough to impede AD progression and multiple steps of A modulations should be taken into consideration according to the different clinical phenotypes in AD patients. e.g., the activity of Foxp3+ regulatory T cells has been reported to be related with A plaque clearance, suggesting novel immunosuppression curing way . Moreover, other approaches besides immunotherapy also need to be explored in order to understand multiple regulations of A for the development of therapies for treating AD.
Read Also: What Is The Difference Between Senility And Dementia
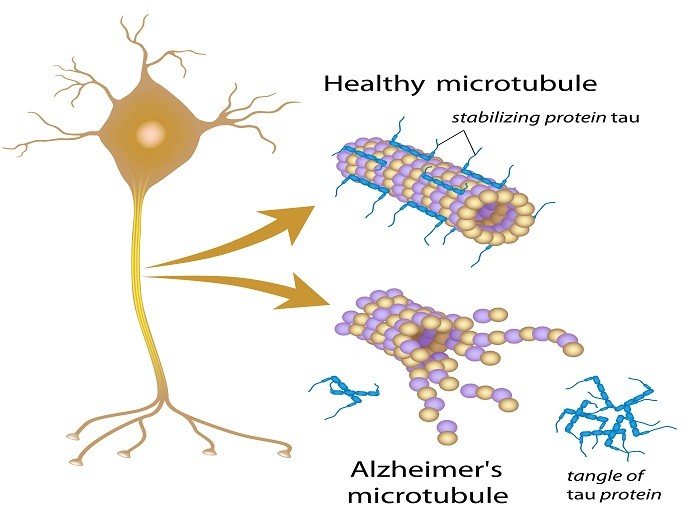
What Causes Tau Buildup
Scientists have long pointed to the importance of tau in AD because of evidence linking the spread of tau with disease progression. The accumulation of beta amyloid in the brain of a person with AD is largely completed at an earlier clinical stage known as mild neurocognitive disorder. However, tau accumulation continues throughout the course of the disease. Beginning in the parts of the brain called the entorhinal cortex and hippocampus, brain tau continues to accumulate as AD progresses. Recent evidence suggests that tau spreads through the brain by means of oligomer seeds that travel across a structure, called a synapse, which allows a nerve cell to pass an electrical or chemical signal to another nerve cell. The total amount of abnormal tau in the AD brain is linked to disease stage and severity.
What Protein Causes Alzheimer’s Disease
Alzheimer’s diseaseproteindiseasecausedproteinprotein
. In this regard, what are the 2 key proteins associated with Alzheimer’s disease?
Alzheimer’s disease is characterized by clumps of two proteins amyloid beta and tau in the brain, but the link between the two has never been entirely clear.
Secondly, what are other names for Alzheimer’s disease? Other Names for This Condition
- AD.
- familial Alzheimer disease
Thereof, what is the main cause of Alzheimer’s?
Causes and risk factors. Like all types of dementia, Alzheimer’s is caused by brain cell death. It is a neurodegenerative disease, which means there is progressive brain cell death that happens over time. In a person with Alzheimer’s, the tissue has fewer and fewer nerve cells and connections.
What type of memory is affected by Alzheimer’s?
In its early stages, Alzheimer’s disease typically affects short-term memory. 1?? For example, this might involve forgetting what you ate for breakfast or repeating yourself in conversation. However, as the disease progresses, people gradually experience more long-term memory loss, also called amnesia.
Recommended Reading: Does Diet Coke Cause Dementia
App Locus Duplication Families
Another feature of the mammalian genome that induces variation in gene expression is the presence of copy number variations , which include both gene duplications and deletions. CNVs are regions of DNA, which can be variable in size, the copy number of which varies between individuals. Both common and rare de novo CNVs have been reported in the human genome . Much effort has been expended to identify and map CNVs in normal individuals and in disease . Duplication of a region of HSA21 containing the APP gene has been reported to cause AD in several families with an autosomal-dominant form of the disease . Genomic duplications of small regions of HSA21, including the APP locus, have been reported in nine families of different ethnic originsFrench, Dutch, Japanese, and Swedish . These families have different overlapping duplications that each includes the APP locus. None of the families exhibited any clinical features suggestive of DS, other than progressive dementia of the AD type. Neuropathological examination of these brains showed abundant A deposits and neurofibrillary tangles in the parenchyma and the induction of A-related cerebral amyloid angiopathy in the cerebral vasculature .
Alzheimers Disease Diagnostic Criteria
A patient suspected to have AD should undergo several tests, including neurological examination, magnetic resonance imaging for neurons, laboratory examinations such as vitamin B12, and other tests besides the medical and family history of the patients . Vitamin B12 deficiency has been long known for its association with neurologic problems and increasing risks of AD, according to some studies. A special marker of vit. B12 deficiency is elevated homocysteine levels, which can cause brain damage by oxidative stress, increasing calcium influx and apoptosis. Diagnoses of vit. B12 deficiency can be done by measuring serum vit. B12 level alongside complete blood count and serum homocysteine levels tests .
In 2011, The National Institute on AgingAlzheimers Association made several changes and updated the 1984 NINCDS-ADRDA criteria for higher specificity and sensitivity in the diagnosis of Alzheimers disease. The newly proposed criteria include probable and possible AD dementia for the use in clinical settings and probable or possible AD dementia with pathophysiological evidence for research purposes, in addition to clinical biomarkers. There are two categories of Alzheimers disease biomarkers: markers of brain amyloid such as positron emission tomography and cerebrospinal fluid , and markers of neuronal injury like cerebrospinal fluid tau, fluorodeoxyglucose for metabolic activity, and magnetic resonance imaging for atrophy measurement .
Read Also: Difference Between Dementia And Senility
Proteins From The Liver May Cause Alzheimer’s In The Brain
Thursday, 16 September, 2021
Australian researchers led by Curtin University have claimed that a probable cause of Alzheimers disease is the leakage from blood into the brain of fat-carrying particles transporting toxic proteins, with lead investigator Professor John Mamo saying his team had identified the probable blood-to-brain pathway that can lead to the disease.
While we previously knew that the hallmark feature of people living with Alzheimers disease was the progressive accumulation of toxic protein deposits within the brain called beta-amyloid, researchers did not know where the amyloid originated from, or why it deposited in the brain, said Prof Mamo, Director of the Curtin Health Innovation Research Institute .
Our research shows that these toxic protein deposits that form in the brains of people living with Alzheimers disease most likely leak into the brain from fat-carrying particles in blood, called lipoproteins.
This blood-to-brain pathway is significant because if we can manage the levels in blood of lipoprotein-amyloid and prevent their leakage into the brain, this opens up potential new treatments to prevent Alzheimers disease and slow memory loss.
As we predicted, the study found that mouse models producing lipoprotein-amyloid in the liver suffered inflammation in the brain, accelerated brain cell death and memory loss, Prof Mamo said.
and . You can also to our weekly newsletters and bimonthly magazine.
We May Finally Know What Causes Alzheimers And How To Stop It
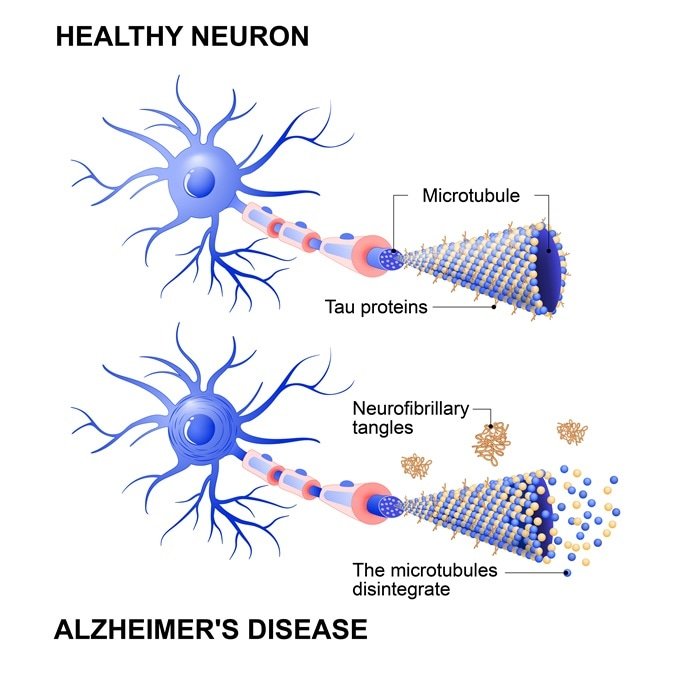
Alzheimers disease has destroyed neurons in the right-hand brain above
Jessica Wilson/Science Photo Library
AFTER decades of disappointment, we may have a new lead on fighting Alzheimers disease. Compelling evidence that the condition is caused by a bacterium involved in gum disease could prove a game-changer in tackling one of medicines biggest mysteries, and lead to effective treatments or even a vaccine.
As populations have aged, dementia has skyrocketed to become the fifth biggest cause of death worldwide. Alzheimers constitutes some 70 per cent of these cases , yet we dont know what causes it. The condition, which results in progressive loss of memory and cognitive function, usually over a decade or so, is devastating both to those who have it and to their loved ones.
The condition often involves the accumulation of two types of proteins called amyloid and tau in the brain. As these are among the earliest physical signs of the disease, the leading hypothesis since 1984 has been that the condition is caused by the defective control of these proteins, especially amyloid, which accumulates to form large, sticky plaques in the brain.
The bulk of research into understanding and treating Alzheimers has centred on this amyloid hypothesis. Huge sums of money have been invested in experiments involving mice genetically modified to produce amyloid, and in developing drugs that block or destroy amyloid proteins, or sometimes degraded tangles of tau.
Recommended Reading: Alzheimer’s Awareness Color
The Amyloid Hypothesis On Trial
In early 2018, two high-profile clinical trials of drugs for Alzheimers disease ended in disappointment. The drugs joined a long list of potential treatments that have failed to deliver significant benefits in people. Their development was guided by an idea that has dominated research on Alzheimers disease for more than 25 years: the amyloid hypothesis, which is the assumption that accumulation of the peptide amyloid- is the main cause of the condition. Researchers proposed that when amyloid- clumps together to form deposits in the brain, it triggers neurodegenerative processes that lead to the loss of memory and cognitive ability that is observed in Alzheimers disease. Amyloid- is therefore an obvious therapeutic target if you can deal with the peptide, then you can treat the condition.
As Selkoe hints, the field could just be experiencing the frustrating journey of discovery that so often precedes the development of effective treatments. But if the idea on which this work is based is wrong, researchers could instead be nearing the end of a blind alley. Much hope is being pinned on ongoing trials in people who are deemed at risk of Alzheimers disease but who have yet to experience symptoms, including those with an inherited form of the condition. Yet amyloid- is not the only potential cause, and some researchers think that it is time to explore alternative avenues.
Different Forms Of Tau
The abnormal tau proteins found in these neurodegenerative diseases are not identical, although they are related. A genetically mutant form of tau can wreak havoc and does so in FTDP-17. The other diseases mentioned feature variants of tau protein created by splicing together taus smaller components in different patterns to create the six different human forms.
Then, after tau has been created from DNA, chemical activities in the brain further modify it in several ways. These chemical alterations of tau change its properties. No longer fit to carry out its usual job, it takes on characteristics that are potentially very damaging. This form of tau no longer sticks together in the same way. Instead, the fabric of connected tau proteins comes apart and reassembles in a disorganized, messy tangle that accumulates in brain cells and is not effectively disposed of through the cells usual ways of removing trash. Besides the microtubular form, which is composed of many tau molecules, tau also exists in smaller versions, called oligomers, which are made up of a few tau proteins. The smaller forms of tau circulate among the neurons, interfering with cellular function. They are found in brains that are developing AD decades before the disease blossoms clinically.
Read Also: Difference Between Senility And Dementia
How Will This Project Help People With Dementia
There is a well-established link between amyloid beta accumulation and the progression of Alzheimers disease. One strategy for designing new treatments for Alzheimers disease is developing drugs that target amyloid beta and cause it to be broken down.
Think this page could be useful to someone? Share it:
Categories
What Causes Death In People With Alzheimers Disease
The progressive destruction of nerve connections in the brain can eventually disrupt vital functions like swallowing. The majority of Alzheimers death are the result of aspiration pneumonia related to the onset of dysphagia . When this occurs, food or liquid can enter the trachea and cause injury or infection in the lungs that leads to pneumonia.
Recommended Reading: Does Diet Coke Cause Alzheimer’s
Causes And Risk Factors Of Alzheimers Disease
AD has been considered a multifactorial disease associated with several risk factors such as increasing age, genetic factors, head injuries, vascular diseases, infections, and environmental factors . The underlying cause of pathological changes in Alzheimers disease is still unknown. Several hypotheses were proposed as a cause for AD but two of them are believed to be the main cause: some believe that an impairment in the cholinergic function is a critical risk factor for AD, while others suggest that alteration in amyloid -protein production and processing is the main initiating factor. However, at present, there is no accepted theory for explaining the AD pathogenesis .
The risk factors for Alzheimers disease.