Indirect Evidence For Glutamates Involvement
Post mortem and genetic studies
Post mortem studies provide evidence for alterations to glutamatergic functioning in schizophrenia. Studies of NMDA receptor expression in post mortem samples have produced some positive findings such as reduced NMDAR1 subunit density in the superior frontal cortex and the superior temporal cortex . However, overall findings regarding NMDA receptor density have been inconsistent . It seems that the abnormality in schizophrenia may primarily be aberrant glutamate receptor localisation as opposed to a generalised deficit . This abnormality could arise as a result of changes in glutamate receptor trafficking molecules . Furthermore there is evidence for a variety of functional changes affecting the intracellular effects of the NMDA receptor that would have a major impact on glutamatergic signalling.
The potential role of glutamate in the pathophysiology of schizophrenia is also supported by recent genetics findings . GRIN2A which codes for an NMDA receptor subunit was found to be associated with schizophrenia, as was SRR which plays a key role in pathways leading to the activation of the NMDA receptor.
NMDA receptor antagonists
How Do Neurotransmitters Work
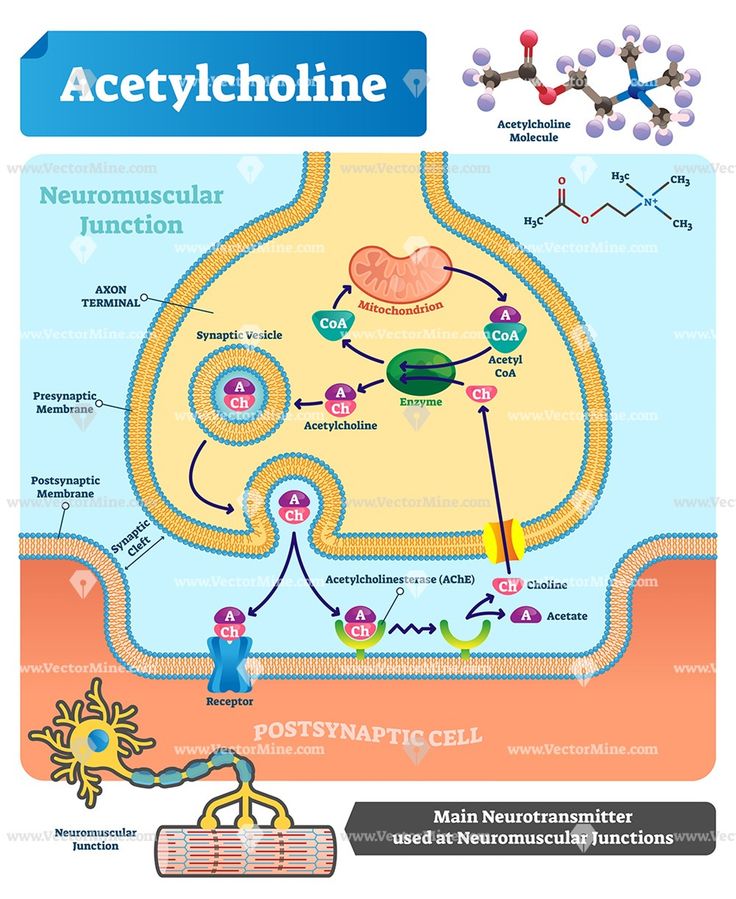
For neurons to work appropriately and effectively send messages throughout the body, they need something to traverse the gap between them.
This gap, called a synapse, requires neurotransmission or electrical impulse for proper chemical transmission between the presynaptic neuron, chemical messenger, and the postsynaptic neuron that receives the messages.
Usually, the neurotransmitter molecules will be released from the axon terminal after an action potential reaches the synapse.
When the electrical signal reaches the end of a neuron, it will trigger the release of neurotransmitters via tiny sacs called synaptic vesicles. Then, the neurotransmitters can cross the small gap to other cells that contain receptors.
The neurotransmitter can then bind to the specific receptors that can cause changes in the target cells. This is where the neurotransmitter will have exciting, inhibiting, or modulatory action.
The neurotransmitter acts as a critical role, and the receptor acts as a lock. Each of the different neurotransmitters may only act on the specific neurotransmitters that it fits into.
After the neurotransmitters play their role, the activity can be stopped through a precise mechanism. The mechanisms include:
Correlation Of Metabolite Abundance And Measures Of Alzheimers Pathology
When examining the relationship between metabolite abundance and measures of pathology and cognitive performance several weak but significant correlations were observed. Of the 15 measured metabolites, all correlated with Braak and CERAD scores in all regions . Correlation analysis to investigate the relationship between metabolite abundance and cognitive performance showed that arginine, aspartate, aminobutanal and guanidobutanoate correlated with MMSE in all regions , GABA, aspartate, tyrosine, DOPA, ornithine, arginine, guanidobutanoate and aminobutanal correlated with Bentons visual retention index and guanidobutanoate correlated with the Boston naming score. Running Title: Neurotransmitter metabolism in Alzheimers brain
Don’t Miss: Can I Buy Jelly Drops For Dementia
Balance Your Blood Sugar And Avoid Stimulants
Your intake of sugar, refined carbohydrates, caffeine, alcohol and cigarettes, as well as stimulant drugs, all affect the ability to keep ones blood sugar level balanced. On top of this common antipsychotic medication may also further disturb blood sugar control. Stimulant drugs, from amphetamines to cocaine, can induce schizophrenia. The incidence of blood sugar problems and diabetes is also much higher in those with schizophrenia.
Therefore it is strongly advisable to reduce, as much as possible, your intake of sugar, refined carbohydrates, caffeine and stimulant drugs and eat a low glycemic load diet.
Neurotransmitters: Functions Types And Examples
The balance of neurotransmitters in our body is the key to proper mood, cognition, energy, and overall health.
Many people know the term neurotransmitters and can even name one or two of them. However, that is where the knowledge ends.
Its highly beneficial to know about each neurotransmitter and what they do for your brain and body.
Neurotransmitters can help us stay calm, improve learning and memory, give us steady energy, manage pain, boost mood, regulate appetite, promote healthy sleep cycles, and contribute to overall health and well-being.
So what exactly are neurotransmitters, and how do they work?
Contents
Read Also: Alzheimer Ribbon
Altered Neurochemistry In Alzheimers Disease: Targeting Neurotransmitter Receptor Mechanisms And Therapeutic Strategy
Neurophysiologyvolume 51, pages 293309 Cite this article
-
290 Accesses
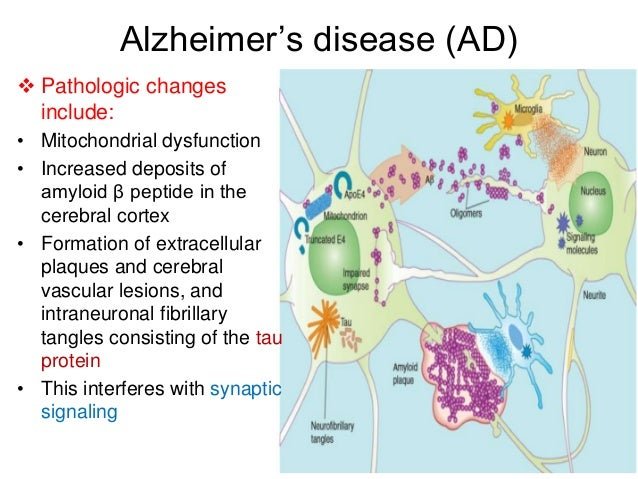
Alzheimers disease is the most common form of dementia characterized by impaired cognitive functions associated with altered neurotransmitter levels in the brain. In AD with advancement of age, symptoms that affect memory, learning ability, language perception, and behavior start to progress. These shifts correlate with accumulations of amyloid beta plaques and neurofibrillary tangles. Such pathological changes are thought to distort synaptic neurotransmitter levels and interrupt neuron-toneuron functioning. The current drugs in AD, like donepezil, rivastigmine, etc., rely to a significant extent on ameliorated cholinergic neurotransmission and utilization of anticholinesterase inhibitors. The recent outbreaks in this disease also target neurotransmitters such as dopamine, serotonin, and their receptor signaling, rather than focusing on cholinergic neurotransmission. This concept emerges due to findings of altered neurotransmitter levels in the post mortem brains of AD patients. The current review summarizes some data underlying mechanisms targeting the role of neurotransmitters and their association with AD, as well as those related to the respective therapeutic aspects. In addition, the review describes advances in recent drugs targeting neurotransmission preclinically and clinically for their neuroprotective role in AD.
Rosmap: Community Studies Fuel Research On Aging And Alzheimers
To understand more about the causes of Alzheimers and related dementias, NIA-funded researchers across the country are making connections between what happens while a person is alive and what can be seen in their brain after death. This approach provides insights about factors that increase the risk of developing dementia as well as what may provide some protection against these diseases.
One such effort is the Religious Orders Study and Rush Memory and Aging Project , referred to collectively as ROSMAP. ROS launched in 1994 and enrolls Catholic nuns, priests, and brothers from across the United States. MAP began in 1997 to study older adults who live in northeastern Illinois and the Chicago metropolitan area. Older adults are recruited primarily from continuous care retirement communities, retirement homes, and senior subsidized housing facilities, as well as through local churches and other social service agencies serving underrepresented communities.
All ROSMAP participants enroll without symptoms of dementia. They agree to annual evaluations and to donate their brain after death, and in some cases, to donate their spinal cord, nerves, and muscles. The researchers meet with the volunteers annually to collect brain scans and blood tests, track any changes in cognition and motor function, and gather information about each participants lifestyle and behaviors.
Also Check: How Long Does A Person Live After Being Diagnosed With Alzheimer’s
Examples Of Important Neurotransmitter Actions
As explained above, the only direct action of a neurotransmitter is to activate a receptor. Therefore, the effects of a neurotransmitter system depend on the connections of the neurons that use the transmitter, and the chemical properties of the receptors that the transmitter binds to.
Here are a few examples of important neurotransmitter actions:
Read Also: Definition Of Phobia
Brain Chemicals And Depression
Researchers have suggested that for some people, having too little of certain substances in the brain could contribute to depression. Restoring the balance of brain chemicals could help alleviate symptomswhich is where the different classes of antidepressant medications may come in.
Even with the help of medications that balance specific neurotransmitters in the brain, depression is a highly complex condition to treat. What proves to be an effective treatment for one person with depression may not work for someone else. Even something that has worked well for someone in the past may become less effective over time, or even stop working, for reasons researchers are still trying to understand.
Researchers continue to try to understand the mechanisms of depression, including brain chemicals, in hopes of finding explanations for these complexities and developing more effective treatments. Depression is a multi-faceted condition, but having an awareness of brain chemistry can be useful for medical and mental health professionals, researchers, and many people who have depression.
Depression Discussion Guide
Dont Miss: Schizophrenia Physiology
Read Also: Bob Knight Alzheimer’s
Icipants Selection And Samples Collection
The study participants were patients from the Neurology Service in the University and Polytechnic Hospital La Fe who previously signed the informed consent. The experimental protocol for the study was approved by the Ethics Committee of the Health Research Institute La Fe , and it is in accordance with the appropriate guidelines .
Saliva samples were obtained from all the participants. They were whole-mouth saliva and collected by spitting into sterile bottles between 10 and 12 a.m. . Participants rinsed their mouth before saliva collection, between 12 mL could be collected across subjects with minor differences between control and case subjects. Then, the samples were aliquoted into 2 mL tubes, and those with visible blood contamination were excluded from the study. Finally, samples were stored at -80ºC until the analysis.
What Happens To The Brain In Alzheimer’s Disease
The healthy human brain contains tens of billions of neuronsspecialized cells that process and transmit information via electrical and chemical signals. They send messages between different parts of the brain, and from the brain to the muscles and organs of the body. Alzheimers disease disrupts this communication among neurons, resulting in loss of function and cell death.
You May Like: At What Age Alzheimer’s Start
Linkage Between Tau And Gabaa Receptors
GABAA receptor is the most well-known pharmacological target for anesthetics including isoflurane, pentobarbital, propofol and chloral hydrate. In recent years, studies indicate that general anesthesia may contribute to the development and exacerbation of AD . Besides A plaques, NFTs composed of hyperphosphorylated tau protein is the most important pathological hallmark of AD. Both pre-clinical and clinical studies have found that anesthesia significantly increase the phosphorylation of tau protein. . Since GABAA receptor is the major pharmacological target of most anesthetics, the activation of GABAA receptors was assumed and later confirmed to participate in the anesthesia-induced tau hyperphosphorylation.
On the other hand, the hyperphosphorylated tau also has influence on GABAergic synapses. In tau P301L transgenic mice, in which the extent of tau phosphorylation was remarkably upregulated, the GABAergic interneurons were observed hyperactivated, leading to increased GABA neurotransmitter level in the brain .
Overall, GABAA receptor activation could enhance tau phosphorylation by reducing the association of PP2A with tau, consequently increase the intracellular NFTs in neurons and contribute to the development of AD. Vice versa, the hyperphosphorylated tau could enhance GABAergic neurotransmission. There might be a feedback loop between GABAA receptor activation and tau phosphorylation in nervous system .
Vascular Disease In Ad
An important cause of the neurological dysfunction in AD is ischemia. This is caused by cerebral amyloid angiopathy , which is found in about 90% of AD cases, and cerebral atherosclerosis and small vessel disease, which is found in the majority of patients. Middle age hypertension is also a risk for AD. Soluble A is a potent vasoconstrictor of cerebral vessels. Amyloid deposition on the vascular wall results in loss of smooth muscle and narrowing of the lumen. With the loss of their smooth muscle, cerebral arterioles lose their ability to constrict and dilate in response to regional brain activation. Narrowing of capillaries decreases cerebral perfusion and impairs the blood brain barrier function. Occluded capillaries form the seed of SPs. Thus, the brain capillary network is diminished and does not regenerate because of senescence. Other consequences of CAA are lobar hemorrhage, cerebral infarcts, and leukoencephlopathy. These changes are superimposed on cerebral atherosclerosis and its complications.
Also Check: Did Ronald Reagan Have Alzheimer In Office
Region Specific Analysis In The Ad Versus Control Group
In the comparison of control versus AD groups, changes were observed mainly in the ITG. In the ITG excitatory neurotransmitters glutamate and aspartate exhibited a lower abundance in AD patients. Also in the ITG, inhibitory neurotransmitters glycine, and serotonin were decreased whilst GABA was increased . A number of neurotransmitter precursors were also increased: ornithine, arginine and tryptophan whilst guanidobutanoate, guanosine, aminobutanal were all significantly decreased in the ITG of AD patients . In the MFG dopamine precursors L-DOPA and tyrosine were the only metabolites to be increased with disease. A significant increase in L-DOPA was also the only significant difference observed in the cerebellum. .
Introduction Of Alzheimers Disease
Alzheimers disease is the most common age-associated neurodegenerative disorder, which is characterized by the deterioration of memory and cognition. About 10% of the population over the age of 65 and 3050% of the population over the age of 85 suffer from AD . Despite significant research and drug development effort in the past decades, currently there are no effective therapies that can prevent, delay or stop the progression of AD, causing a severe burden for the patients, their families and the society.
A small subset of AD cases result from dominantly inherited genetic mutations in genes encoding the -amyloid precursor protein and presenilins . These AD cases usually develop disease before the age of 60, referring as early-onset familial AD . Sporadic or late-onset AD usually develops the disease later in life, representing the majority of AD cases . The pathological hallmarks of AD include widespread neuronal degeneration, senile plaques and intracellular neurofibrillary tangles .
Read Also: Senility Vs Dementia
Inverse Agonists Of Gabaa Receptor 5 Subunit
GABAA receptor is a pentamer containing various allosteric binding sites. It has been suggested that different subunits of GABAA receptor may exert relatively distinct function. For example, 1 subunit is mainly responsible for the sedative action of diazepam, 2 subunit mediates the anxiolytic-like action, whereas 5 subunit may be associated with cognition and memory . Its reported that 5 subunit deficiency enhanced hippocampus-dependent memory and spatial learning ability in mice . In addition, GABAA receptors containing 5 subunit was found upregulated in the dentate gyrus of 5× FAD mice, and has been suggested to mediate the tonic inhibition in CNS. A series of compounds have been developed, serving as inverse agonists of GABAA receptor 5 subunit, which bind to 5 subunit with much higher affinity than other subunits, but negatively regulate receptor activity.
In the early 2000s, the Merck Sharp and Dohme identified a series of benzothiophene derivatives with a notable binding selectivity for GABAA receptor 5 subunit. Among them the ligands MRK-016, 5IA and 5IA-II, all of which displayed encouraging effect on cognition in animal models. The compound 5IA has further advanced to preclinical and clinical studies .
Region Specific Analysis In The Asymad Versus Control Group
The abundance of all fifteen metabolites were compared between control and ASYMAD groups . The only neurotransmitter that had an altered abundance was dopamine which was decreased in the MFG . In the ITG aminobutanal and guanosine which are both involved in the metabolism of neurotransmitters were also decreased .
Metabolites significantly increased in abundance and shown as green triangles and metabolites significantly decreased in abundance and shown as red chevrons with the size representing the magnitude of the change. Grey circles represent metabolites that were not significantly associated with disease. A) shifts observed in the cerebellum, B) shifts observed in the inferior temporal gyrus, C) shifts observed in the middle frontal gyrus.
Read Also: Senile Dementia Treatments
Implications Of Gabaergic Neurotransmission In Alzheimers Disease
- 1Fujian Provincial Key Laboratory of Neurodegenerative Disease and Aging Research, Institute of Neuroscience, College of Medicine, Xiamen University, Xiamen, China
- 2Neurodegenerative Disease Research Program, Sanford-Burnham Medical Research Institute, La Jolla, CA, USA
- 3Department of Neuroscience, Mayo Clinic, Jacksonville, FL, USA
- 4The Interdepartmental Program of Translational Biology and Molecular Medicine, Huffington Center on Aging, Baylor College of Medicine, Houston, TX, USA
Current Status Of Neurotransmitters And Alzheimers Disease
Neurotransmitters play a large role in maintaining synaptic and cognitive functions in mammals, including humans, by sending signals across synapses . Neurotransmitters are usually stored in synaptic vesicles, beneath the membrane in the axon terminal, and are released into the synapse with the appropriate signal from neurons. The released neurotransmitters establish connections through the synaptic cleft and bind to their appropriate receptors.
Neurotransmitters with a known role in AD pathogenesis include acetylcholine, which is synthesized from serine dopamine, from L-phenyl alanine/L-tyrosine GABA, from glutamate, a by-product of decarboxylation serotonin, from L-tryptophan histaminergic, from L-histidine and N-methyl-D-aspartate , from D-aspartic acid and arginine. Neurotransmitters without a significant role in AD pathogenesis include glutamate, glycine, norepinephrine, epinephrine, melatonin, gastrin, oxytocin, vasopressin, cholecystokinin, neuropeptide Y, and enkephalins. However, these neurotransmitters may have a role in causing oxidative stress, which is known to be involved in AD pathogenesis.
Fig.1
Neurotransmitters in a healthy synapse and an Alzheimers disease synapse.
Don’t Miss: Does Alzheimer’s Cause Dementia
Other Neurotransmitters In Schizophrenia
Clozapine, which is the most effective medication available for treating the symptoms of Schizophrenia, is a very weak blocker of the D2 dopamine receptors. This suggests that there must be other neurotransmitter systems that are also involved in causing Schizophrenia, though so far not much is known about what are these other factors.
One neurotransmitter that has gotten increased attention in recent years for its role in Schizophrenia in glutamate, which targets NMDA receptors in the brain. Phencyclidine and Ketamine are two drugs that block the actions of glutamate at the NMDA receptor, and these drugs can cause both the positive and negative symptoms of Schizophrenia in people who do not have the condition. Increasing the actions of glutamate by using high doses of precursor molecules that are metabolized into glutamate, such as D-serine, glycine and D-cycloserine, show promise in helping to improve the negative symptoms of Schizophrenia.
Read Also: What Is The Meaning Of Phobia
Sleep And Schizophrenia Are Intimately Linked
Since sleep regulation involves many neurotransmitter systems and brain circuits, it is likely that the mechanisms generating normal sleep overlap with those that maintain mental health. This would explain why disturbed sleep and schizophrenia are so intimately linked. This was the essence of the seminar given at the Lundbeck Institute on 17th April 2015 by Russell Foster, director of the Sleep and Circadian Neuroscience Unit at the University of Oxford, UK.
Sleep disturbance is common in serious mental illness, and schizophrenia is no exception. It is widely accepted that schizophrenia disrupts sleep and circadian rhythms. But it also seems that disturbances of sleep can precede severe mental illness and may even help cause it.1
Recommended Reading: Does Pristiq Help With Anxiety
Also Check: Neurotransmitter Related To Alzheimer’s