Advancements In Predictive Diagnostics
Testing for brain health is becoming more important to patients every day, and while there is a blood test for Alzheimers genes, there are still unknown factors that prevent it from definitively diagnosing Alzheimers or other forms of dementia. However, technologies for assessing and diagnosing neurological diseases are rapidly growing and improving.
Altoida is developing a precision neurology platform to measure and analyze cognitive and functional aspects of brain health. Our unique method for collecting and analyzing nearly 800 active digital biomarkers enables highly specific, accurate, and generalizable data for both cross-sectional and longitudinal analysis of neurocognitive function. This method will serve as the foundation for Altoidas work towards predicting conversion to Alzheimers disease years before symptomatic onset.
To learn more about Atloidas digital platform and how to address the questionis there a blood test for Alzheimers genescontact us today.
Dementia Caused By A Complex Disease
Nearly all cases of dementia are the result of a complex disease. In these cases, genes may increase the risk of developing dementia, but they dont cause it directly.
What causes complex disease dementias?
When dementia is the result of a complex disease, it is likely to be caused by a combination of risk factors. These include:
- non-genetic factors for example, members of the same family may all smoke or have an unhealthy diet, which are both risk factors for dementia.
- genetic factors a person may inherit the same dementia risk variants as other members of their family. This could include variants in genes such as APOE.
These factors are often shared by members of the same family. This is why many people have some family history of dementia for example, they may have a parent who developed dementia in their 90s and a brother who developed dementia in his 70s. This is not the same as familial dementia.
Is it possible to directly inherit dementia when it’s caused by a complex disease?
No. People who are related to each other by birth are more likely to have the same risk variants as each other. However, risk variants for dementia do not directly cause dementia . This means that its not possible to directly inherit dementia through risk variants.
However, a person who has dementia risk variants is at higher risk of developing dementia than someone who does not have risk variants. Despite this higher risk, they still might not develop dementia.
Scientists Have Identified Genetic Mutations That Cause Early
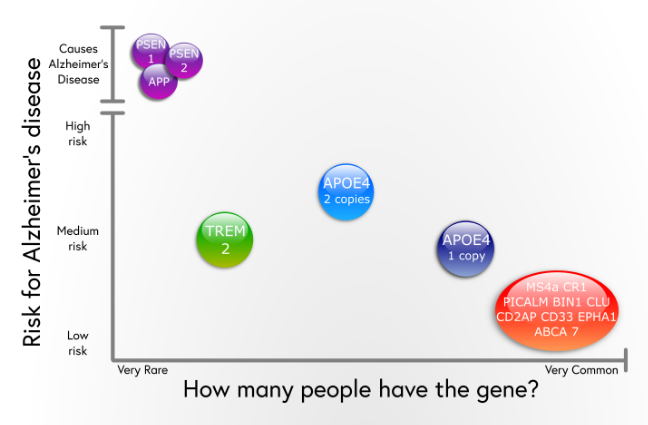
While researchers have found genetic risk factors for late-onset Alzheimers, they have, on the other hand, identified rare genetic mutations that cause early-onset Alzheimers, which affects people younger than the age of 65.
Langbaum said people who carry hereditary genetic mutations in one of three genes PSEN1, PSEN2 and APP nearly always develop the disease. They account for less than 1 percent of all people with Alzheimers.
She noted that the decision to order genetic testing for a genetic mutation that causes early-onset Alzheimers is typically made between the patient, clinician and in many cases, family members.
Meanwhile, there seems to be certain genetic factors that may be protective against Alzheimers. Scientists from the Banner Alzheimers Institute and colleagues found that an extremely rare variant of ApoE, known as the Christchurch mutation, appeared to protect a Colombian woman from the ravages of the disease.
The woman, who carried the PSEN1 mutation and two copies of the Christchurch mutation, had normal cognitive abilities until her 70s when she began to develop memory decline. Even though she had high levels of beta-amyloid plaques in her brain, there were very limited atrophy and tau pathology.
For more guidance on this topic to be the first to know about upcoming live talks with experts on practical topics related to Alzheimers, dementia and caregiving.
You May Like: Grey’s Anatomy Alzheimer’s
Dementia And Down Syndrome
People with Down syndrome are born with an extra piece of DNA. This means they also have an extra copy of the APP gene. This leads to the build-up of amyloid plaques in the brain, which play a role in the development of Alzheimers disease. While not everyone with Down syndrome will go on to develop symptoms of Alzheimers, most people with the condition over the age of 40 will have amyloid build-up. It is estimated that about 50% of people with Down syndrome develop symptoms like memory loss, usually in their 50s and 60s.
To find out more about Down syndrome and dementia you can contact Downs Syndrome Association helpline on 0333 1212 300 or visit their website www.downs-syndrome.org.uk.
Looking Into Ad Pathogenesis With Genome Editing Tools
Another study uncovered the cellular pathways controlling prion-like propagation of Ï aggregation. CRISPR interference screens were performed in a human cell-based model of propagation of Ï aggregation . This revealed that knockdown of several components of the endosomal sorting complexes required for transport machinery, including charged multivesicular body protein 6 , or CHMP2A in combination with CHMP2B , promote propagation of Ï aggregation. These findings suggest that endolysosomal escape is a critical step in Ï propagation in neurodegenerative diseases.
Further support to the idea of impaired endosomal functioning in AD was observed in the study by Kwart et al. who created a panel of isogenic knockin human iPSC lines carrying APP and/or PSEN1 mutations. Global transcriptomic and translatomic profiling revealed that familial AD mutations had overlapping effects on the expression of AD-related and endocytosis-associated genes, including increased Rab5+ early endosome size. These authors found that APP and PSEN1 mutations had discordant effects on Aβ production but similar effects on APP β C-terminal fragments, which accumulated in all mutant neurons. The additional advantage of the genome editing system in this case was the use of a non-overexpression human-based system.
You May Like: Terry Semel Alzheimer’s
Future Of Alzheimers Blood Tests
When blood tests become mainstream, they will significantly impact the way doctors diagnose and even treat the condition.
Beyond the Preclivity AD and Simoa described above, there are multiple other types of blood tests in development some are looking at protective fats in the blood. Research shows that an increase in fatty amide levels indicates more beta amyloid in the brain. Beta amyloid plaques have been found in the brains of people with Alzheimers.
Other blood tests will measure proteins such as tau proteins and the neurofilament light chain protein. Researchers have found that neuron death, characteristic in people with Alzheimers, may be detected by the corresponding increase of these proteins in the blood.
It is also possible that the probable link between insulin resistance, diabetes and Alzheimers means blood tests could show the disease in someone well before symptoms occur. Another protein called IRS-1 is commonly associated with diabetes but has also been linked to Alzheimers. Studies have shown that someone with Alzheimers has more IRS-1 than even someone with diabetes.
What Is At The End Of The Ad Tunnel
AD is a multifactorial disease, the main feature of which is dementia. AD is not a monogenic disease and the same clinical symptoms may be observed as a result of the presence of mutations in many genes. The association of these mutations with the progression of the disease is not fully understood however, the involvement of many genes suggests convergence of their effects in functions. The way from GWAS to the development of new therapies represents a significant challenge for modern medicine. Methods of genome editing, specifically CRISPR/Cas9, may be used for simultaneous correction of multiple gene mutations, although the possibility of the application of this approach to the central nervous system remains uncertain. Figure 2 shows general purposes of application of the CRISPR system for investigation of AD. Firstly, genome editing is used for the correction of well-known mutations, which are responsible for the early-onset familial AD forms, and studies of the consequences of this treatment, as described above. Secondly, CRISPR/Cas9 is a suitable tool to study the consequences of the mutations in genes, which are not causative in order to reveal their role in brain physiology and pathophysiology of the late-onset AD forms. Thus, genome editing can be used to study whether the newly identified mutations can cause or act in combination with others to induce the development of a disease phenotype.
Don’t Miss: What Color Is The Alzheimer’s Ribbon
Cause #: Past Head Trauma
The Alzheimers Association states that scientists have found a link between traumatic brain injury and a greater risk of dementia. After a traumatic injury, your brain creates large amounts of beta amyloid. This is the same protein that develops into the damaging plaques that are a hallmark of Alzheimers.
Theres one difference: After a traumatic brain injury, beta amyloid, although present, doesnt clump into plaques. However, the damage may increase the risk of them doing so later in life.
Gene Editing And Alzheimer’s Disease: Is There Light At The End Of The Tunnel
- Laboratory of Functional Biochemistry of the Nervous System, Institute of Higher Nervous Activity and Neurophysiology, Russian Academy of Sciences, Moscow, Russia
Alzheimer’s disease continues to be a fatal, incurable neurodegenerative disease, despite many years of efforts to find approaches to its treatment. Here we review recent studies on Alzheimer’s disease as a target for gene therapy and specifically, gene editing technology. We also review the opportunities and limitations of modern methods of gene therapy based on the CRISPR editing system. The opportunities of using this approach for modeling, including cellular and animal models, studying on pathogenesis and disease correction mechanisms, as well as limitations for its therapeutic use are discussed.
Functional genomics allowed substantial improvement in specific tools for experiments and gene therapy. In the present review we shall discuss modern studies directed to the application of these new approaches to create new models which allow us to understand the pathogenesis of AD or to correct the known mutated genes associated with AD.
Recommended Reading: What Shampoos Cause Alzheimer’s Disease And Cancer
Designer Nucleases For Directed Genome Editing
Classical gene transfer using viral vectors results in random integration of specific genes into the genome as well as within cell episomes. In the therapeutic field, the replacement of random integration with targeted gene insertion, or targeted correction, has become a commonly accepted potential solution. In principle, genome editing consists of making a break in double-stranded DNA in a locus where the target gene is located, followed by reparation of this break. Endonucleases are enzymes that cleave DNA via catalysis of the disintegration of phosphodiester bonds within polynucleotide chains. At present, three types of nucleases are the main tools used in experimental studies: zinc finger nucleases , transcription activator-like effector nucleases , and clustered, regularly interspaced, short palindromic repeats /CRISPR-associated system , i.e., CRISPR/Cas, system .
The use of sgRNAs is more technologically feasible and cost-efficient as compared to ZFN and TALEN, which require time-consuming customization of DNA binding proteins. CRISPR/Cas9 may be relatively easily packed into one rLV-based vector, and the high efficiency of editing and lower cytotoxicity, at least compared to ZFN , has provided explosive progress in research using genome editing.
What Could These Results Mean For Patients In The Future
Dr. Aubert: In the past, the delivery of therapeutic genes required invasive surgeries for patients with Alzheimers disease. Non-invasive and personalized gene delivery to the brain has the potential to increase beneficial clinical effects for the treatment of Alzheimers disease. Our preclinical study confirms that focused ultrasound and modified gene carriers are feasible strategies for gene delivery to the brain, and that their immunological effects can be controlled.
Also Check: Dementia Ribbon Colour
Am I At Risk For Alzheimer’s
Whether we have seen early signs in ourselves or not, many of us want to know what our chances of getting this disease may be. Research has shown a number of possible factors that can impact your chances of getting Alzheimer’s disease, although none of these are a cause in and of themselves.
Some ages are more at risk
Old age is one of the most obvious risk factors. The vast majority of people develop the disease after the age of 65, and once you reach 65, your risk of getting Alzheimer’s doubles every five years. But Alzheimer’s doesn’t only affect people over 65 it has been known to affect people half that age, although this is much rarer.
Alzheimer’s is hereditary
A family history will also increase your risk of getting the disease. The risk increases even more if you have multiple family members who have suffered from the disease.
Whilst this may be due to the hereditary genetic factors we will look at in more depth later, there may be other factors at play. These could include environmental factors that impact both yourself and your family.
Gender predisposition
Gender is another significant risk factor. The first discovery of the disease back in 1906 was in a woman, and about twice as many women as men over 65 have Alzheimer’s. This may be in part to the fact that women have a longer lifespan or may even possibly be linked to menopause.
Genetic factors of Alzheimer’s
Other risk factors
Blood Tests Available Now
A blood test is available now, through your doctor, that delivers a result in about 10 days and accurately determines whether someone has Alzheimers disease. The Preclivity AD Test from C2N Diagnostics measures proteins in blood plasma that indicate a buildup of plaques known to cause dementia in peoples brains.
Cost
The cost of this Alzheimers blood test is $1,250, less expensive than a PET scan that looks for the same protein buildups. Unfortunately, the price tag cannot be covered by Medicare or Medicaid, but C2N says people who cant afford the test can qualify for a discount based on their income.
Does it Work?
C2N Diagnostics says they tested Preclivity AD in 686 people and compared it to the results of PET scans. When PET scans showed a high amount of amyloid-beta, a protein that turns to plaques and tangles in the brains of people with Alzheimers, the blood test agreed 92 percent of the time. When the PET scan showed no amyloid, the blood test showed the same about 77 percent of the time.
How to Get the Blood Test
The Preclivity AD Test has not been approved by the Food and Drug Administration. It is currently available only through a doctor, and can be sold by the company under rules for commercial laboratories. The test, in other words, is available despite lacking FDA approval, though C2N has said the FDA is planning on reviewing the test in 2021. The Alzheimers Association says it wont endorse the test until there is FDA approval.
You May Like: What Color Ribbon Is Alzheimer’s
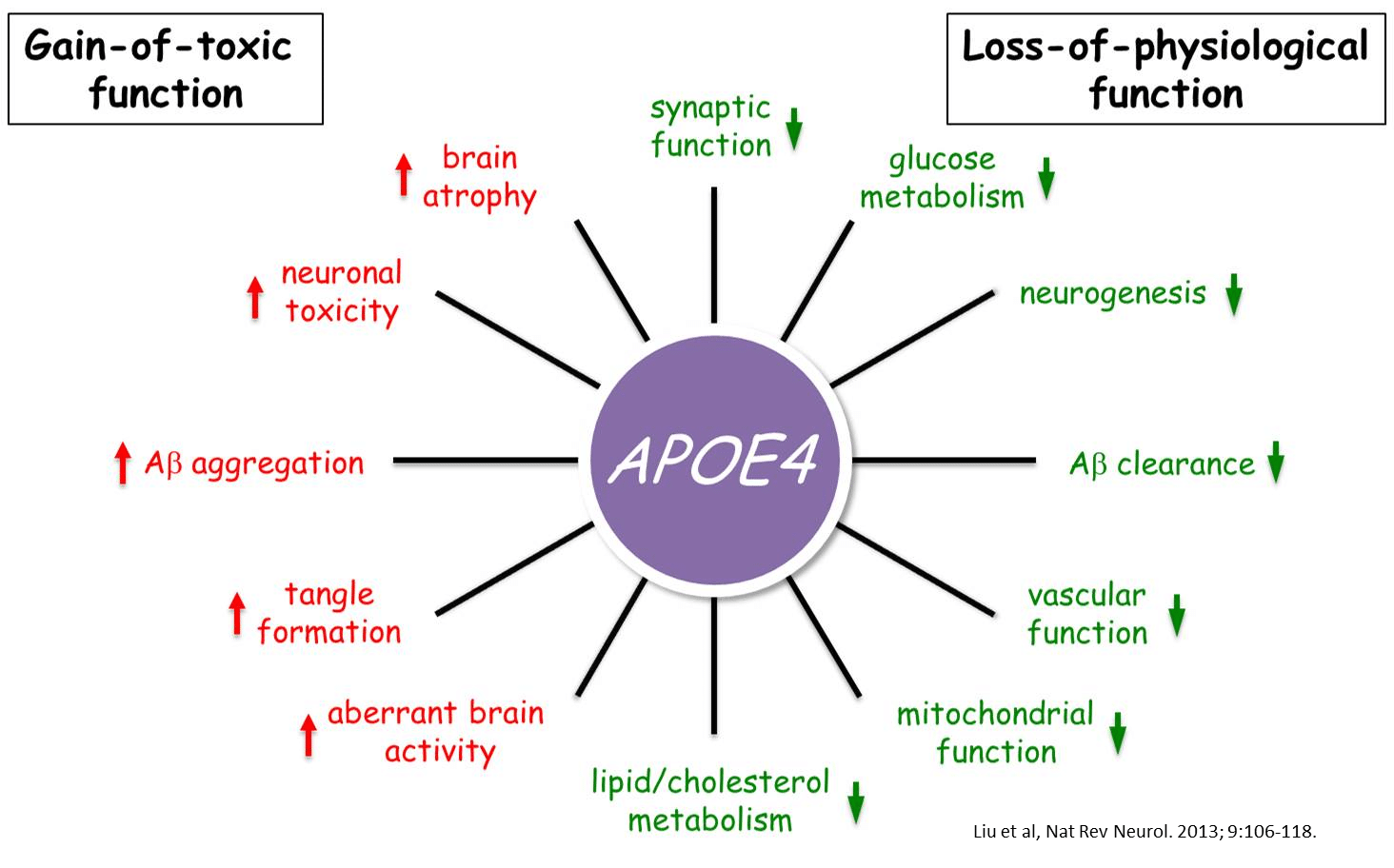
Apoe 4 Does Not Guarantee That Someone Will Develop Alzheimer’s Steps Can Be Taken To Reduce Alzheimer’s Risk
Research has shown us that individuals with the ApoE 4 gene are able to reduce their risk for developing Alzheimers. Individuals with the ApoE 4 gene can reduce their risk by proactively managing other risk factors and by pursuing activities and lifestyles which have been shown to reduce the risk of Alzheimers.
Individuals with the ApoE 4 gene are more vulnerable to certain factors which increase the risk of Alzheimers, risk factors like diabetes or high blood pressure. This makes it even more important than normal for individuals with the ApoE 4 gene to manage their blood pressure and diabetic status.
The chance of developing Alzheimers is influenced by behaviors that occur decades before and leading up to when the symptoms first become noticeable. Individuals with the ApoE 4 gene should begin addressing risk factors sooner, rather than later.
Individuals who have a family history of Alzheimers and individuals who believe they are experiencing memory loss should be tested for the ApoE 4 gene. Other individuals can be tested, but individuals in these two groups are most likely to possess the ApoE 4 gene.
Before being tested it is important to consider the emotional consequences of learning your result and how the result may affect you and your loved ones.
We believe that emotionally-stable individuals willing to take a proactive approach to managing their Alzheimers risk will benefit from genetic testing.
Genetic Testing Explained By An Expert
We asked Dr. G. Allen Power, MD, author of Dementia Beyond Drugs: Changing the Culture of Care and Dementia Beyond Disease: Enhancing Well-Being, for his insights on the Alzheimers genetic test.
First of all, the gene that carries the high risk of younger-onset Alzheimer’s across multiple family members is relatively rare, he explained. Only about 1-2% of people with Alzheimer’s have this type of disorder, and most other forms of dementia do not have a strong genetic component.
He added: Beyond that, some other genes may increase one’s lifetime risk, but not to such a great extent. The risk of getting Alzheimer’s, like most dementias, usually increases with age, meaning it’s mostly caused by a combination of factors that occur cumulatively as we live our lives, not by our genes. Even most of those who develop symptoms before age 65 are simply on the end of the bell curve and not subject to a strong genetic cause.
Like other experts, he strongly cautions those who get the Alzheimers genetic test.
I would recommend gene testing for those who have a very strong family history of younger-onset Alzheimer’s and who want to know if they carry the APOE4 gene, he said. But keep in mind that even having the gene is not a guarantee you will develop dementia.
You May Like: Does Neil Diamond Have Alzheimer’s
People Affected By Dementia Are Often Concerned About Whether The Condition Can Be Passed Along In Families
Here we discuss the role of heredity in Alzheimers disease and other forms of dementia.
Dementia is the term used to describe the symptoms of a large group of illnesses which cause a progressive decline in a persons functioning. It is a broad term to describe a loss of memory, intellect, rationality, social skills and what would be considered normal emotional reactions. Alzheimers disease is the most common form of dementia, accounting for 50% to 70% of all cases of dementia. It occurs relatively frequently in older people, regardless of family history. For females aged 65 to 69 years dementia affects 1 person in 80 compared to 1 person in 60 for males. For both males and females aged 85 and over the rate is approximately 1 person in 4.